Abstract
Purpose
Little is known about the pharmacokinetics (PKs) of oxycodone in patients with advanced cancer. There is considerable reluctance to subject these patients to non-essential tests including repeated venipuncture that has been necessary in PK studies to date. We investigated the possibility of using saliva sampling as a simple non-invasive test to investigate opioid PKs.
Methods
Patients with malignant disease receiving oral sustained release (SR) oxycodone at any dose were asked to provide saliva samples at the same time as blood samples. Samples were not taken within 6 h of a dose of immediate release oxycodone. Plasma and saliva oxycodone and metabolite concentrations were measured using HPLC coupled with tandem mass spectrometric detection.
Results
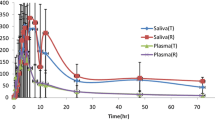
One hundred and thirty-nine paired plasma/saliva samples were collected from 43 cancer patients who had been taking SR oxycodone for more than 5 days at doses ranging from 10 to 600 mg/day (median 40 mg/day). Plasma concentrations of oxycodone and noroxycodone ranged from 1.0 to 256.0 and 0.9–269.4 μg/L, respectively. Salivary concentrations of oxycodone (range 0.93–3,620, mean 336 μg/L) were much higher than plasma concentrations (mean 38.2 μg/L). There was a poor correlation between concentrations of both oxycodone and noroxycodone in plasma and saliva over a range of times following dosing (r 2 = 0.4641 and 0.3891, respectively). No correlation was shown between salivary pH and oxycodone or noroxycodone concentrations. The majority of patients questioned chose saliva sampling over plasma sampling as the preferred method.
Conclusion
High levels of both oxycodone and its major metabolite are present in saliva, but this does not provide a valid substitute for plasma when monitoring oxycodone levels for PK studies or therapeutic monitoring.



Similar content being viewed by others
References
Mullangi R, Agrawal S, Srinivas N (2009) Measurement of xenobiotics in saliva: is saliva an attractive alternative matrix? Case studies and analytical perspectives. Biomed Chromatogr 23:3–25
Michael C, Teichert J, Preiss R (2008) Determination of voriconazole in human plasma and saliva using high performance liquid chromatography with fluorescence detection. J Chromatogr B Analyt Technol Biomed Life Sci 865:74–80
Gallagher P, Leitch M, Massey A, McAllister-Williams R, Young A (2006) Assessing cortisol and dehydroepiandrosterone (DHEA) in saliva: effect of collection method. J Psychopharmacol 20:643–649
Poujol S, Bressolle F, Duffour J, Abderrahim AG, Astre C, Ychou M, Pinguet F (2006) Pharmacokinetics and pharmacodynamics of irinotecan and its metabolites from plasma and saliva data in patients with metastatic digestive cancer receiving Folfiri regimen. Cancer Chemother Pharmacol 58:292–305
Voultsios A, Kennaway D, Dawson D (1997) Salivary melatonin as a circadian phase marker: validation and comparison to plasma melatonin. J Biol Rhythms 12:457–466
Ruiz ME, Conforti P, Fagiolino P, Volonté MG (2010) The use of saliva as a biological fluid in relative bioavailability studies: comparison and correlation with plasma results. Biopharm Drug Dispos 31:476–485
Gubala W, Zuba D (2003) Gender differences in the pharmacokinetics of ethanol in saliva and blood after oral ingestion. Pol J Pharmacol 55:639–644
Gorodischer R, Koren G (1992) Salivary excretion of drugs in children: theoretical and practical issues in therapeutic drug monitoring. Dev Pharmacol Ther 9:161–177
Gorodischer R, Burtin P et al (1997) Is saliva suitable for therapeutic monitoring of anticonvulsants in children: an evaluation in the routine clinical setting. Ther Drug Monit 19(6):637–642
Gandia P, Bareille M, Saivin S, Pavy Le-Traon A, Lavit M, Guell A, Houin G (2003) Influence of simulated weightlessness on the oral pharmacokinetics of acetaminophen as a gastric emptying probe in man: a plasma and a saliva study. J Clin Pharmacol 43:1235–1243
Ritschel WA, Parab PV, Denson DD, Coyle DE, Gregg RV (1987) Absolute bioavailability of hydromorphone after peroral and rectal administration in humans: saliva/plasma ratio and clinical effects. J Clin Pharmacol 27:647–653
Davis M, Varga J, Dickerson D, Walsh D, LeGrand SB, Lagman R (2003) Normal-release and controlled-release oxycodone: pharmacokinetics, pharmacodynamics, and controversy. Support Care Cancer 11:84–92
Grönlund J, Saari TI, Hagelberg NM, Neuvonen PJ, Olkkola KT, Laine K (2010) Exposure to oral oxycodone is increased by concomitant inhibition of CYP2D6 and 3A4 pathways, but not by inhibition of CYP2D6 alone. Br J Clin Pharmacol 70:78–87
Glare P, Davis M (2009) Oxycodone. In: Davis M, Glare P, Hardy JR (eds) Opioids in cancer pain, 2nd edn. Oxford University Press, Oxford, UK
Lalovic B, Kharasch E, Hoffer C, Risler L, Liu-Chen LY, Shen D (2006) Pharmacokinetics and pharmacodynamics of oral oxycodone in healthy human subjects: role of circulating active metabolites. Clin Pharmacol Ther 79:461–479
Andreassen TN, Klepstad P, Davies A, Bjordal K, Lundström S, Kaasa S, Dale O (2011) Influences on the pharmacokinetics of oxycodone: a multicentre cross-sectional study in 439 adult cancer patients. Eur J Clin Pharmacol. doi:10.1007/s00228-010-0948-5
Kirvela M, Lindgren L, Seppala T, Olkkola K (1996) The pharmacokinetics of oxycodone in uremic patients undergoing renal transplantation. J Clin Anesth 8:13–18
Tallgren M, Olkkola KT, Seppala T, Hockerstedt K, Lindgren L (1997) Pharmacokinetics and ventilatory effects of oxycodone before and after liver transplantation. Clin Pharmacol Ther 61:655–661
Lugo RA, Kern SE (2004) The pharmacokinetics of oxycodone. J Pain Palliat Care Pharmacother 18(4):17–28
Edwards SR, Smith MT (2007) Low levels of oxycodone and its oxidative metabolites, noroxycodone, and oxymorphone, in rat plasma by high-performance liquid chromatography-electrospray ionization-tandem mass spectrometry. J Chromatogr 848:264–270
Quigley C, Joel S, Patel N, Baksh A, Slevin M (2003) Plasma concentrations of morphine, morphine-6-glucuronide and morphine-3-glucuronide and their relationship with analgesia and side-effects in patients with cancer-related pain. Palliat Med 17:185–190
Cone E, Clarke J, Tsanaclis L (2007) Prevalence and disposition of drugs of abuse and opioid treatment drugs in oral fluid. J Anal Toxicol 31:424–433
O'Neal C, Crouch D, Rollins D, Fatah AA (1999) Correlation of saliva codeine concentrations with plasma concentrations after oral codeine administration. J Anal Toxicol 23:452–459
Pourzitaki C, Tachou H et al (2006) Heroin metabolites detection in saliva and blood. Rev Clin Pharmacol Pharmacokinet 20:108–109
Shiran M, Hassanzadeh-Khayyat M, Iqbal MZ et al (2005) Can saliva replace plasma for the monitoring of methadone? Ther Drug Monit 27:580–586
Kopecky E, Jacobsen S, Klein J, Bhushan K, Gideon K (1997) Correlation of morphine sulphate in blood plasma and saliva in pediatric patients. Ther Drug Monit 19:530–534
Skopp G, Pötsch L, Klinder K, Richter B, Aderjan R, Mattern R (2001) Saliva testing after single and chronic administration of dihydrocodeine. Int J Leg Med 114:133–140
Okura T, Hattori A, Takano Y, Sato T et al (2008) Involvement of the pyrilamine transporter, a putative organic cation transporter, in blood-brain barrier transport of oxycodone. Drug Metab Dispos 36(10):2005–2013
Yabuuchi H, Tamai I, Nezu J, Sakamoto K et al (1999) Novel membrane transporter OCTN1 mediates multispecific, bidirectional, and pH-dependent transport of organic cation. J Pharmacol Exp Ther 289:768–773
Genter MB, Krishan M, Augustine LM, Cherrington NJ (2010) Drug transporter expression and localization in rat nasal respiratory and olfactory mucosa and olfactory bulb. Drug Metab Dispos 38:1644–1647
Thompson SJ, Koszdin K, Bernards CM (2000) Opiate-induced analgesia is increased and prolonged in mice lacking P-glycoprotein. Anesthesiology 92:1392–1399
Boström E, Simonssen USH, Hammarlund-Udenaes M (2005) Oxycodone pharmacokinetics and pharmacodynamics in the rat in the presence of the P-glycoprotein inhibitor PSC833. J Pharm Sci 94:1060–1066
Hassan HE, Myers AL, Lee IJ, Coop A et al (2007) Oxycodone induces overexpression of P-glycoprotein (ABCB1) and affects paclitaxel's tissue distribution in Sprague Dawley rats. J Pharm Sci 96:2494–2506
Mercer SL, Coop A (2011). Opioid analgesics and P-glycoprotein efflux tranporters: a potential systems-level contribution to analgesic tolerance. Curr Top Med Chem (in press)
Uematsu T, Yamaoka M, Matsuura T, Doto R et al (2001) P-glycoprotein expression in human major and minor salivary glands. Arch Oral Biol 46(6):521–527
Kandimalla KK, Donovan MD (2005) Localization and differential activity of P-glycoprotein in the bovine olfactory and nasal respiratory mucosa. Pharm Res 22(7):1121–1128
Yamahara H, Lee VHL (1993) Drug metabolism in the oral cavity. Adv Drug Deliv Rev 12:25–40
Bosker W, Huestis M (2009) Oral fluid testing for drugs of abuse. Clin Chem 55(11):1910–1931
Acknowledgements
This project was supported by grants from the Australian National Health and Medical Research Council and the Mater Health Services JP Kelly Research Fund.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hardy, J., Norris, R., Anderson, H. et al. Is saliva a valid substitute for plasma in pharmacokinetic studies of oxycodone and its metabolites in patients with cancer?. Support Care Cancer 20, 767–772 (2012). https://doi.org/10.1007/s00520-011-1147-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-011-1147-3