Abstract
Key message
Root morphological responses of neotropical tree seedlings to nitrate and ammonium nutrition vary according to the successional group, but may also depend on each species.
Abstract
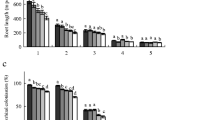
Neotropical tree seedlings from contrasting successional groups have different nitrogen (N) use strategies, as well as they exhibit striking differences in root morphology. Here, we assessed the effects of hydroponic cultivation with nitrate or ammonium on the growth and root morphological traits of seedlings of pioneer (Cecropia pachystachya, Croton floribundus, Schinus terebinthifolius, Solanum mauritianum, and Trema micrantha) and non-pioneer (Cabralea canjerana, Campomanesia xanthocarpa, Cariniana estrellensis, Guarea kunthiana, and Poecilanthe parviflora) tree species native to Brazilian Atlantic Forest. Nitrate-grown seedlings of pioneer species showed higher shoot and root dry matters and total root length compared to those cultivated with ammonium, while non-pioneer trees showed smaller or no difference in these traits when comparing N sources. These results agree with the decrease of nitrate availability in the soil relative to that of ammonium over an Atlantic Forest succession. Regardless of the successional group, nitrate treatment resulted in longer main and lateral roots in most species. The effects of nitrate and ammonium on specific root length, lateral-root density, and fine-root diameter could be associated with successional groups and N source preference. However, root hair responses varied according to each species. Cecropia pachystachya and C. canjerana roots had morphological responses to N source intermediaries between the successional groups, which are coherent with their ecological niches. These results demonstrate that the effects of N source on the growth and root morphology of neotropical tree seedlings could be associated with their successional group, although species-specific responses were also observed.



Similar content being viewed by others
Code availability
Not applicable.
References
Addo-Danso SD, Defrenne CE, McCormack ML et al (2020) Fine-root morphological trait variation in tropical forest ecosystems: an evidence synthesis. Plant Ecol 221:1–13
Aidar MPM, Schmidt S, Moss G, Stewart GR, Joly CA (2003) Nitrogen use strategies of neotropical rainforest trees in threatened Atlantic Forest. Plant Cell Environ 26:389–399
Andrews M, Raven JA, Lea PJ (2013) Do plants need nitrate? The mechanisms by which nitrogen form affects plants. Ann Appl Biol 163:174–199
Araya T, Kubo T, von Wirén N, Takahashi H (2016) Statistical modeling of nitrogen-dependent modulation of root system architecture in Arabidopsis thaliana. J Integr Plant Biol 58:254–265
Armstrong W (1982) Waterlogged soils. In: Etherighton JR (ed) Environment and plant ecology, 2nd edn. Wiley, London, pp 290–330
Bastin J, Finegold Y, Garcia C et al (2019) The global tree restoration potential. Science 365:76–79
Batista CUN, Medri ME, Bianchini E, Medri C, Pimenta JA (2008) Flood tolerance in Cecropia pachystachya Trec. (Cecropiaceae): ecophysiological and morpho-anatomical aspects. Acta Bot Brasil 22:91–98
Bittsánszky A, Pillinszky K, Gyulai G, Komives T (2015) Overcoming ammonium toxicity. Plant Sci 231:184–190
Boudsocq S, Niboyet A, Lata JC et al (2012) Plant preference for ammonium versus nitrate: a neglected determinant of ecosystem functioning? Am Nat 180(1):60–69
Bouguyon E, Brun F, Meynard D, et al. (2015) Multiple mechanisms of nitrate sensing by Arabidopsis nitrate transceptor NRT1.1. Nat. Plants 1:15015.
Bouguyon E, Perrine-Walker F, Pervent M et al (2016) Nitrate controls root development through posttranscriptional regulation of the NRT1.1/ NPF6.3 transporter/sensor. Plant Physiol 172:1237–1248
Britto DT, Kronzucker HJ (2002) NH4+ toxicity in higher plants: a critical review. J Plant Physiol 159:567–584
Britto DT, Kronzucker HJ (2013) Ecological significance and complexity of N-source preference in plants. Ann Bot 112:957–963
Caldeira MVW, Gomes DR, Gonçalves EDO, Delarmelina WM, Sperandio HV, Trazzi PA (2012) Biossólido como substrato para produção de mudas de Toona ciliata var. australis (Biosolid as a soil substrate for Toona ciliata var. australis. seedlings). Rev Árv 36:1009–1018
Chandler G (1981) Physiological aspects of rainforest regeneration: I. effects of light and nitrogen source on growth and ammonium assimilating enzymes of Solanum mauritianum and Syzygium floribundum. New Phytol 87:301–313
Debiasi TV, Calzavara AK, da Silva LM et al (2019) Nitrogen metabolism of Neotropical tree seedlings with contrasting ecological characteristics. Acta Physiol Plant 41:131
Dias J, Surian T, Mantoani MC, Pereira LCDSM, Torezan JMD (2016) Crescimento de Megathyrsus maximus (capim-colonião) e duas espécies nativas arbóreas em diferentes condições ambientais (Growth of Megathyrsus maximus and of two native tree species in different environmental conditions). Floresta 46:325–334
Dunbabin V, Diggle A, Rengel Z (2003) Is there an optimal root architecture for nitrate capture in leaching environments? Plant Cell Environ 26:835–844
Durigan G, Gurgel-Garrido LMA, Garrido MAO (1999) Desenvolvimento de Plathymenia reticulata Benth. em plantio puro e em consorciação com espécies de diferentes estágios sucessionais (The growth of Plathymenia reticulata Benth. in pure and mixed stands with species from different successional stages). Revista do Instituto Florestal (RIF) São Paulo 11:131–136.
Esteban R, Royo B, Urarte E, Zamarreño ÁM, Garcia-Mina JM, Moran JF (2016) Both free indole-3-acetic acid and photosynthetic performance are important players in the response of Medicago truncatula to urea and ammonium nutrition under axenic conditions. Front Plant Sci 7:140
FAO (1994) Soil map of the world. FAO-UNESCO, Roma
Foehse D, Jungk A (1983) Influence of phosphate and nitrate supply on root hair formation of rape, spinach and tomato plants. Plant Soil 74:359–368
Gandolfi S, Joly CA, Leitão-Filho HDF (2009) “Gaps of deciduousness”: cyclical gaps in tropical forests. Sci Agric 66:280–284
Giam X (2017) Global biodiversity loss from tropical deforestation. Proc Natl Acad Sci USA 114:5775–5777
Guan P, Wang R, Nacry P et al (2014) Nitrate foraging by Arabidopsis roots is mediated by the transcription factor TCP20 through the systemic signaling pathway. Proc Natl Acad Sci USA 111:15267–15272
Hawkesford M, Horst W, Kichey T et al (2012) Functions of macronutrients. In: Marschner H (ed) Marschner’s Mineral Nutrition of Higher Plants, 3rd edn. Academic Press, London, pp 135–189
Helali SMR, Nebli H, Kaddour R, Mahmoudi H, Lachaâl M, Ouerghi Z (2010) Influence of nitrate—ammonium ratio on growth and nutrition of Arabidopsis thaliana. Plant Soil 336:65–74
Hochholdinger F, Yu P, Marcon C (2018) Genetic control of root system development in maize. Trends in Plant Sci 23:79–88
Hodge A (2004) The plastic plant: root responses to heterogeneous supplies of nutrients. New Phytol 162:9–24
Keeney DR, Nelson DW (1982) Nitrogen inorganic forms. In: Page AL, Miller RH, Keeney DR (eds) Methods of soil analysis, part 2: Chemical and microbiological properties. American Society of Agronomy, Madison, pp 643–698
Kraiser T, Gras DE, Gutiérrez AG, González B, Gutiérrez RA (2011) A holistic view of nitrogen acquisition in plants. J Exp Bot 62:1455–1466
Kronzucker HJ, Siddiqi MY, Glass ADM, Britto DT (2003) Root ammonium transport efficiency as a determinant in forest colonization patterns: an hypothesis. Physiol Plant 117:164–170
Krouk G, Lacombe B, Bielach A et al (2010) Nitrate-regulated auxin transport by NRT1.1 defines a mechanism for nutrient sensing in plants. Dev Cell 18:927–937
Li Q, Li BH, Kronzucker HJ, Shi WM (2010) Root growth inhibition by NH4+ in Arabidopsis is mediated by the root tip and is linked to NH4+ efflux and GMPase activity. Plant Cell Environ 33:1529–1542
Li C, Li Q, Qiao N, Xu X, Li Q, Wang H (2015) Inorganic and organic nitrogen uptake by nine dominant subtropical tree species. iForest 9:253–258
Lima JE, Kojima S, Takahashi H, von Wirén N (2010) Ammonium triggers lateral root branching in Arabidopsis in an AMMONIUM TRANSPORTER1; 3-dependent manner. Plant Cell 22:3621–3633
Liu Y, Von Wirén N (2017) Ammonium as a signal for physiological and morphological responses in plants. J Exp Bot 68:2581–2592
Liu Y, Lai N, Gao K, Chen F, Yuan L, Mi G (2013) Ammonium inhibits primary root growth by reducing the length of meristem and elongation zone and decreasing elemental expansion rate in the root apex in Arabidopsis thaliana. PLoS ONE 8:e61031
Liu M, Li C, Xu X, Wanek W, Jiang N, Wang H, Yang X (2017) Organic and inorganic nitrogen uptake by 21 dominant tree species in temperate and tropical forests. Tree Physiol 37:1515–1526
Lorenzi H (2008) Árvores Brasileiras: Manual de Identificação e Cultivo de Plantas Arbóreas Nativas do Brasil (Trees of Brazil: Manual of identification and cultivation of native tree species of Brazil). Instituto Plantarum de Estudos da Flora Ltda, SP
Marino D, Moran JF (2019) Can ammonium stress be positive for plant performance? Front Plant Sci 10:1103
Mian IA, Riaz M, Cresser MS (2009) The importance of ammonium mobility in nitrogen-impacted unfertilized grasslands: a critical reassessment. Environ Poll 157:1287–1293
Mielke MS, De Almeida AAF, Gomes FP, Mangabeira PAO, Silva DDC (2005) Effects of soil flooding on leaf gas exchange and growth of two neotropical pioneer tree species. New For 29:161–168
Morais WWC, Susin F, Vivian MA, Araújo MM (2012) Influência da irrigação no crescimento de mudas de Schinus terebinthifolius (Influence of irrigation on Schinus terebinthifolius seedlings growth). Pesq Flor Br 32:23
Mounier E, Pervent M, Ljung K, Gojon A, Nacry P (2014) Auxin mediated nitrate signalling by NRT1.1 participates in the adaptive response of Arabidopsis root architecture to the spatial heterogeneity of nitrate availability. Plant Cell Environ 37:162–174
Na L, Li Z, Xiangxiang M, Ara N, Jinghua Y, Mingfang Z (2014) Effect of nitrate/ammonium ratios on growth, root morphology and nutrient elements uptake of watermelon (Citrullus lanatus) seedlings. J Plant Nutr 37:1859–1872
Naulin PA, Armijo GI, Vega AS, Tamayo KP, Gras DE, de la Cruz J, Gutiérrez RA (2020) Nitrate induction of primary root growth requires cytokinin signaling in Arabidopsis thaliana. Plant Cell Physiol 61:342–352
Nolan RH, Drew DM, O’Grady AP et al (2018) Safeguarding reforestation efforts against changes in climate and disturbance regimes. For Ecol Manag 424:458–467
Oliveira HC, da Silva LMI, de Freitas LD et al (2017) Nitrogen use strategies of seedlings from neotropical tree species of distinct successional groups. Plant Physiol Biochem 114:119–127
Pilbeam DJ (2015) Essential Elements: Macronutrients - Nitrogen. In: Barker AV Pilbeam DJ (ed) Handbook of Plant Nutrition. 2nd edn. CRC press: Boca Raton, 17–64.
Pinheiro KAO, de Carvalho JOP, Quanz B, de Barros Francez LM, Schwartz G (2007) Fitossociologia de uma área de preservação permanente no leste da Amazônia: indicação de espécies para recuperação de áreas alteradas (Phytosociology of a permanent preservation area in Eastern Amazon: plant species recommendations for the recovery of a degraded land). Floresta 37:175–187
R Core Team (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL http://www.R-project.org/
Rondina ABL, Tonon BC, Lescano LEAM, Hungria M, Nogueira MA, Zangaro W (2019) Plants of distinct successional stages have different strategies for nutrient acquisition in an Atlantic Rain Forest ecosystem. Int J Plant Sci 180:186–199
Ruffel S, Krouk G, Ristova D, Shasha D, Birnbaum KD, Coruzzi GM (2011) Nitrogen economics of root foraging: transitive closure of the nitrate– cytokinin relay and distinct systemic signaling for N supply vs. demand. Proc Natl Acad Sci USA 108:18524–18529
Sousa-Silva JC, Salgado MAS, Felfil JM, Rezende AV, Franco AC (1999) Desenvolvimento inicial de Cabralea canjerana Saldanha em diferentes condições de luz (Early stage development of Cabralea canjerana Saldanha in different light conditions). Boletim do Herbario Ezechias Paulo Heringer 4:80–89
Sun CH, Yu JQ, Hu DG (2017) Nitrate: a crucial signal during lateral roots development. Front Plant Sci 8:485
Szczerba MW, Britto DT, Ali SA, Balkos KD, Kronzucker HJ (2008) NH4+-stimulated and-inhibited components of K+ transport in rice (Oryza sativa L.). J Exp Bot 59:3415–3423
Tennant D (1975) A test of a modified line intersect method of estimating root length. J Ecol 63:995–1001
Tian D, Niu S (2015) A global analysis of soil acidification caused by nitrogen addition. Environ Res Lett 10:024019
Vatter T, Neuhäuser B, Stetter M, Ludewig U (2015) Regulation of length and density of Arabidopsis root hairs by ammonium and nitrate. J Plant Res 128:839–848
Wang H, Gao JE, Li XH, Zhang SL, Wang HI (2015) Nitrate accumulation and leaching in surface and ground water based on simulated rainfall experiments. PLoS ONE 10:e0136274
Wang J, Sun Z, Hui D et al (2018) Responses of seedling performance to altered seasonal precipitation in a secondary tropical forest, southern China. For Ecol Manag 410:27–34
Wu Q, Pagès L, Wu J (2016) Relationships between root diameter, root length and root branching along lateral roots in adult, field-grown maize. Ann Bot 117:379–390
Yu P, Hochholdinger F, Li C (2015) Root-type-specific plasticity in response to localized high nitrate supply in maize (Zea mays). Ann Bot 116:751–762
Zangaro W, Nishidate FR, Camargo FRS, Romagnoli GG, Vandressen J (2005) Relationships among arbuscular mycorrhizas, root morphology and seedling growth of tropical native woody species in southern Brazil. J Trop Ecol 21:529–540
Zangaro W, Nishidate FR, Vandresen J, Andrade G, Nogueira MA (2007) Root mycorrhizal colonization and plant responsiveness are related to root plasticity, soil fertility and successional status of native woody species in southern Brazil. J Trop Ecol 23:53–62
Zangaro W, Alves RA, Lescano LE, Ansanelo AP, Nogueira MA (2012) Investment in fine roots and arbuscular mycorrhizal fungi decrease during succession in three Brazilian ecosystems. Biotropica 44:141–150
Zangaro W, Alves RA, Souza PB, Rostirola LV, Lescano LEAM, Rondina ABL, Nogueira MA (2014) Succession and environmental variation influence soil exploration potential by fine roots and mycorrhizal fungi in an Atlantic ecosystem in southern Brazil. J Trop Ecol 30:237–248
Zangaro W, Lescano LEAM, Matsuura EM, Rondina ABL, Nogueira MA (2016) Differences between root traits of early-and late-successional trees influence below-ground competition and seedling establishment. J Trop Ecol 32:300–313
Zhang HM, Rong HL, Pilbeam D (2007) Signalling mechanisms underlying the morphological responses of the root system to nitrogen in Arabidopsis thaliana. J Exp Bot 58:2329–2338
Zhao DY, Tian QY, Li LH, Zhang WH (2007) Nitric oxide is involved in nitrate-induced inhibition of root elongation in Zea mays. Ann Bot 100:497–503
Zhu C, Yang N, Guo Z, Qian M, Gan L (2016) An ethylene and ROS-dependent pathway is involved in low ammonium-induced root hair elongation in Arabidopsis seedlings. Plant Physiol Biochem 105:37–44
Zimmermann APL, Fleig FD, Tabaldi LA, Aimi SC (2019) Morphological and physiological plasticity of saplings of Cabralea canjerana (Vell.) Mart. in different light conditions. Rev. Árv. 43:e430103.
Acknowledgements
The authors are grateful to Diego Genuário Gomes and Angélica Nunes Tiepo for helping with the statistical analysis and to the Laboratory of Biodiversity and Ecosystem Restoration of the State University of Londrina for making the seeds available.
Funding
This study was financed in part by the Coordination for the Improvement of Higher Education Personnel—Brazil (CAPES)—Finance Code 001 (RCS) National Council for Scientific and Technological Development (CNPq) (HCO, grant 306583/2017–8), and CNPq/Araucaria Foundation (WZ and HCO, grant PELD 441540/2016–3).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and material
The authors declare that all data supporting the findings of this study are available within the paper.
Additional information
Communicated by A. DesRochers.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
da Silva, R.C., Rondina, A.B.L., Zangaro, W. et al. Inorganic nitrogen sources alter the root morphology of neotropical tree seedlings from different successional groups. Trees 35, 875–887 (2021). https://doi.org/10.1007/s00468-021-02087-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-021-02087-x