Abstract
Background
Glucocorticoid discontinuation, a challenge in systemic lupus erythematosus (SLE), might be achievable with the advent of new therapeutic options.
Methods
This single-center study included 31 children with newly diagnosed pediatric SLE between 2002 and 2021, after the exclusion of patients who were followed for less than 1 year after treatment initiation and those lost to follow-up. Patient characteristics, clinical course including flares, treatment, glucocorticoid discontinuation, and outcomes were retrospectively analyzed.
Results
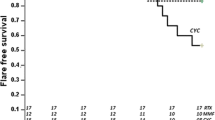
Glucocorticoids could be discontinued in 19 (61%) patients during a median observation period of 105.5 (range, 17–221) months. Of these, 5 (26%), 12 (63%), and 18 (95%) patients could discontinue glucocorticoids in 3, 5, and 10 years from treatment initiation, respectively. Additionally, 18 of the 19 patients did not experience flares after glucocorticoid discontinuation during a median duration of 37.2 (7.2–106.8) months. Three of the nineteen patients achieved drug-free remission. At last follow-up, all patients achieved low disease activity with or without glucocorticoids and 19, 8, and 1 patient were receiving mycophenolate mofetil (MMF), MMF plus tacrolimus, and MMF plus ciclosporin A, respectively. Flares were observed in 15 patients during the observation period. MMF as initial immunosuppressant (P = 0.01) and shorter interval between therapy initiation and achieving maintenance prednisolone dose of 0.1–0.15 mg/kg/day (P = 0.001) were associated with significantly reduced flare risk. Femoral head necrosis was observed in two patients.
Conclusion
Despite the small sample size, these results support glucocorticoid discontinuation as a therapeutic target in pediatric SLE.


Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Levy DM, Kamphuis S (2012) Systemic lupus erythematosus in children and adolescents. Pediatr Clin North Am 59:345–364. https://doi.org/10.1016/j.pcl.2012.03.007
Mackie FE, Kainer G, Adib N, Boros C, Elliott EJ, Fahy R, Munro J, Murray K, Rosenberg A, Wainstein B, Ziegler JB, Singh-Grewal D (2015) The national incidence and clinical picture of SLE in children in Australia - a report from the Australian Paediatric Surveillance Unit. Lupus 24:66–73. https://doi.org/10.1177/0961203314552118
Hiraki LT, Feldman CH, Liu J, Alarcón GS, Fischer MA, Winkelmayer WC, Costenbader KH (2012) Prevalence, incidence, and demographics of systemic lupus erythematosus and lupus nephritis from 2000 to 2004 among children in the US Medicaid beneficiary population. Arthritis Rheum 64:2669–2676. https://doi.org/10.1002/art.34472
Hersh AO, von Scheven E, Yazdany J, Panopalis P, Trupin L, Julian L, Katz P, Criswell LA, Yelin E (2009) Arthritis Rheum 61:13–20. https://doi.org/10.1002/art.24091
Urowitz MB, Gladman DD, Tom BD, Ibañez D, Farewell VT (2008) Changing patterns in mortality and disease outcomes for patients with systemic lupus erythematosus. J Rheumatol 35:2152–2158. https://doi.org/10.3899/jrheum.080214
Felten R, Scher F, Sibilia J, Chasset F, Arnaud L (2019) Advances in the treatment of systemic lupus erythematosus: from back to the future, to the future and beyond. Joint Bone Spine 86:429–436. https://doi.org/10.1016/j.jbspin.2018.09.004
Moroni G, Gatto M, Raffiotta F, Binda V, Frangou E, Lightstone L, Boumpas DT (2018) Can we withdraw immunosuppressants in patients with lupus nephritis in remission? An expert debate. Autoimmun Rev 17:11–18. https://doi.org/10.1016/j.autrev.2017.11.003
van Vollenhoven RF, Mosca M, Bertsias G, Isenberg D, Kuhn A, Lerstrøm K, Aringer M, Bootsma H, Boumpas D, Bruce IN, Cervera R, Clarke A, Costedoat-Chalumeau N, Czirják L, Derksen R, Dörner T, Gordon C, Graninger W, Houssiau F, Inanc M, Jacobsen S, Jayne D, Jedryka-Goral A, Levitsky A, Levy R, Mariette X, Morand E, Navarra S, Neumann I, Rahman A, Rovensky J, Smolen J, Vasconcelos C, Voskuyl A, Voss A, Zakharova H, Zoma A, Schneider M (2014) Treat-to-target in systemic lupus erythematosus: recommendations from an international task force. Ann Rheum Dis 73:958–967. https://doi.org/10.1136/annrheumdis-2013-205139
Walsh M, Jayne D, Moist L, Tonelli M, Pannu N, Manns B (2010) Practice pattern variation in oral glucocorticoid therapy after the induction of response in proliferative lupus nephritis. Lupus 19:628–633. https://doi.org/10.1177/0961203309356292
Moroni G, Gallelli B, Quaglini S, Banfi G, Rivolta E, Messa P, Ponticelli C (2006) Withdrawal of therapy in patients with proliferative lupus nephritis: long-term follow-up. Nephrol Dial Transplant 21:1541–1548. https://doi.org/10.1093/ndt/gfk073
Basu B, Roy B, Babu BG (2017) Efficacy and safety of rituximab in comparison with common induction therapies in pediatric active lupus nephritis. Pediatr Nephrol 32:1013–1021. https://doi.org/10.1007/s00467-017-3583-x
Rygg M, Pistorio A, Ravelli A, Maghnie M, Di Iorgi N, Bader-Meunier B, Da Silva C, Roldan-Molina R, Barash J, Dracou C, Laloum SG, Jarosova K, Deslandre CJ, Koné-Paut I, Garofalo F, Press J, Sengler C, Tauber T, Martini A, Ruperto N; Paediatric Rheumatology International Trials Organisation (PRINTO) (2012) A longitudinal PRINTO study on growth and puberty in juvenile systemic lupus erythematosus. Ann Rheum Dis 71:511-517https://doi.org/10.1136/annrheumdis-2011-200106
Hochberg MC (1997) Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 40:1725. https://doi.org/10.1002/art.1780400928
Weening JJ, D'Agati VD, Schwartz MM, Seshan SV, Alpers CE, Appel GB, Balow JE, Bruijn JA, Cook T, Ferrario F, Fogo AB, Ginzler EM, Hebert L, Hill G, Hill P, Jennette JC, Kong NC, Lesavre P, Lockshin M, Looi LM, Makino H, Moura LA, Nagata M; International Society of Nephrology Working Group on the Classification of Lupus Nephritis; Renal Pathology Society Working Group on the Classification of Lupus Nephritis (2004) The classification of glomerulonephritis in systemic lupus erythematosus revisited. Kidney Int 65:521-530. https://doi.org/10.1111/j.1523-1755.2004.00443.x
Bombardier C, Gladman DD, Urowitz MB, Caron D, Chang CH (1992) Derivation of the SLEDAI. A disease activity index for lupus patients. The Committee on Prognosis Studies in SLE. Arthritis Rheum 35:630–640. https://doi.org/10.1002/art.1780350606
Fanouriakis A, Kostopoulou M, Alunno A, Aringer M, Bajema I, Boletis JN, Cervera R, Doria A, Gordon C, Govoni M, Houssiau F, Jayne D, Kouloumas M, Kuhn A, Larsen JL, Lerstrøm K, Moroni G, Mosca M, Schneider M, Smolen JS, Svenungsson E, Tesar V, Tincani A, Troldborg A, van Vollenhoven R, Wenzel J, Bertsias G, Boumpas DT (2019) 2019 update of the EULAR recommendations for the management of systemic lupus erythematosus. Ann Rheum Dis 78:736–745. https://doi.org/10.1136/annrheumdis-2019-215089
Kizawa T, Nozawa T, Kikuchi M, Nagahama K, Okudela K, Miyamae T, Imagawa T, Nakamura T, Mori M, Yokota S, Tsutsumi H (2015) Mycophenolate mofetil as maintenance therapy for childhood-onset systemic lupus erythematosus patients with severe lupus nephritis. Mod Rheumatol 25:210–214. https://doi.org/10.3109/14397595.2014.950810
Steiman AJ, Urowitz MB, Ibañez D, Papneja A, Gladman DD (2014) Prolonged clinical remission in patients with systemic lupus erythematosus. J Rheumatol 41:1808–1816. https://doi.org/10.3899/jrheum.131137
Zen M, Iaccarino L, Gatto M, Bettio S, Nalotto L, Ghirardello A, Punzi L, Doria A (2015) Prolonged remission in Caucasian patients with SLE: prevalence and outcomes. Ann Rheum Dis 74:2117–2122. https://doi.org/10.1136/annrheumdis-2015-207347
Otten MH, Cransberg K, van Rossum MA, Groothoff JW, Kist-van Holthe JE, Ten Cate R, Van Suijlekom-Smit LW (2010) Disease activity patterns in juvenile systemic lupus erythematosus and its relation to early aggressive treatment. Lupus 19:1550–1556. https://doi.org/10.1177/0961203310374485
Dooley MA, Jayne D, Ginzler EM, Isenberg D, Olsen NJ, Wofsy D, Eitner F, Appel GB, Contreras G, Lisk L, Solomons N; ALMS Group (2011) Mycophenolate versus azathioprine as maintenance therapy for lupus nephritis. N Engl J Med 365:1886-1895. https://doi.org/10.1056/NEJMoa1014460
Li X, Ren H, Zhang Q, Zhang W, Wu X, Xu Y, Shen P, Chen N (2012) Mycophenolate mofetil or tacrolimus compared with intravenous cyclophosphamide in the induction treatment for active lupus nephritis. Nephrol Dial Transplant 27:1467–1472. https://doi.org/10.1093/ndt/gfr484
Jiang YP, Zhao XX, Chen RR, Xu ZH, Wen CP, Yu J (2020) Comparative efficacy and safety of mycophenolate mofetil and cyclophosphamide in the induction treatment of lupus nephritis: a systematic review and meta-analysis. Medicine 99:e22328. https://doi.org/10.1097/MD.0000000000022328
Bengtsson C, Bengtsson A, Costenbader Kh, Jönsen A, Rantapää-Dahlqvist S, Sturfelt G, Nived O (2011) Systemic lupus erythematosus and cardiac risk factors: medical record documentation and patient adherence. Lupus 20:1057–1062. https://doi.org/10.1177/0961203311403639
Yen EY, Shaheen M, Woo JMP, Mercer N, Li N, McCurdy DK, Karlamangla A, Singh RR (2017) 46-year trends in systemic lupus erythematosus mortality in the United States, 1968 to 2013: a nationwide population-based study. Ann Intern Med 167:777–785. https://doi.org/10.7326/M17-0102
Acknowledgements
The authors would like to thank Drs. Kentaro Ogata and Kentaro Matsuoka for their academic contribution and pathological diagnosis. The authors would also like to thank Mr. James R. Valera for his assistance with editing of the manuscript.
Author information
Authors and Affiliations
Contributions
KN prepared the first draft of the manuscript and performed data collection and analysis. MOg, TK, SIs, MOk, SY, TN, and MS edited and reviewed the manuscript. SIt supervised and revised the manuscript. KI and KK revised and oversaw the work. All authors contributed to the study conception and design and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study (No. 1868) was approved by the Ethics Committee of the National Center for Child Health and Development.
Consent to participant
Informed consent for participating in this study was not required in accordance with the Declaration of Helsinki and the Ethical Guidelines for Medical and Health Research Involving Human Subjects of the Ministry of Health, Labor, and Welfare.
Consent for publication
Consent for publication was waived in accordance with the guidelines.
Conflict of interest
Koichi Kamei has received research funding from the Terumo Foundation for Life Sciences and Arts, Public Foundation of Vaccination Research Center, and Taiju Life Social Welfare Foundation and donations from Ono Pharmaceutical Co., Ltd. Kenji Ishikura has received lecture fees from Asahi Kasei Pharma, Chugai Pharmaceutical Co., Ltd, Zenyaku Kogyo Co., Ltd, and Novartis Pharma K.K. and grants from Asahi Kasei Pharma, Chugai Pharmaceutical Co., Ltd., Novartis Pharma, and Zenyaku Kogyo Co., Ltd. Shuichi Ito has received honoraria and research funding from Asahi Kasei Pharma, Chugai Pharmaceutical Co., Ltd, Zenyaku Kogyo Co., Ltd, and Astellas PHARMA Co., Ltd. Other authors have no potential conflicts of interest to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nishi, K., Ogura, M., Ishiwa, S. et al. Glucocorticoid discontinuation in pediatric-onset systemic lupus erythematosus: a single-center experience. Pediatr Nephrol 37, 2131–2139 (2022). https://doi.org/10.1007/s00467-021-05350-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-021-05350-y