Abstract
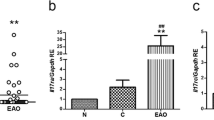
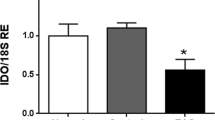
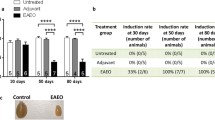
Experimental autoimmune orchitis (EAO) is a chronic inflammatory disorder that causes progressive spermatogenic impairment. EAO is characterized by high intratesticular levels of nitric oxide (NO) and tumor necrosis factor alpha (TNFα) causing germ cell apoptosis and Sertoli cell dysfunction. However, the impact of this inflammatory milieu on the spermatogenic wave is unknown. Therefore, we studied the effect of inflammation on spermatogonia and preleptotene spermatocyte cell cycle progression in an EAO context and through the intratesticular DETA-NO and TNFα injection in the normal rat testes. In EAO, premeiotic germ cell proliferation is limited as a consequence of the undifferentiated spermatogonia (CD9+) cell cycle arrest in G2/M and the reduced number of differentiated spermatogonia (c-kit+) and preleptotene spermatocytes that enter in the meiotic S-phase. Although inflammation disrupts spermatogenesis in EAO, it is maintained in some seminiferous tubules at XIV and VII–VIII stages of the epithelial cell cycle, thereby guaranteeing sperm production. We found that DETA-NO (2 mM) injected in normal testes arrests spermatogonia and preleptotene spermatocyte cell cycle; this effect reduces the number of proliferative spermatogonia and the number of preleptotene spermatocytes in meiosis S-phase (36 h after). The temporal inhibition of spermatogonia clonal amplification delayed progression of the spermatogenic wave (5 days after) finally altering spermatogenesis. TNFα (0.5 and 1 µg) exposure did not affect premeiotic germ cell cycle or spermatogenic wave. Our results show that in EAO the inflammatory microenvironment altered spermatogenesis kinetics through premeiotic germ cell cycle arrest and that NO is a sufficient factor contributing to this phenomenon.













Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
References
Bauché F, Stéphan JP, Touzalin AM, Jégou B (1998) In vitro regulation of an inducible-type NO synthase in the rat seminiferous tubule cells. Biol Reprod 58:431–438. https://doi.org/10.1095/biolreprod58.2.431
Braylan RC, Benson NA, Nourse V, Kruth HS (1982) Correlated analysis of cellular DNA, membrane antigens and light scatter of human lymphoid cells. Cytometry 2:337–343. https://doi.org/10.1002/cyto.990020511
Caneguim BH, Cerri PS, Spolidório LC, Miraglia SM, Sasso-Cerri E (2009) Structural alterations in the seminiferous tubules of rats treated with immunosuppressor tacrolimus. Reprod Biol Endocrinol. https://doi.org/10.1186/1477-7827-7-19
Clermont Y (1972) Kinetics of spermatogenesis in mammals: seminiferous epithelium cycle and spermatogonial renewal. Physiol Rev 52:198–236. https://doi.org/10.1152/physrev.1972.52.1.198
De SK, Chen HL, Pace JL, Hunt JS, Terranova PF, Enders GC (1993) Expression of tumor necrosis factor-alpha in mouse spermatogenic cells. Endocrinology 133:389–396. https://doi.org/10.1210/endo.133.1.8319585
Doncel GF, Di Paola JA, Lustig L (1989) Sequential study of the histopathology and cellular and humoral immune response during the development of an autoimmune orchitis in Wistar rats. Am J Reprod Immunol 20:44–51. https://doi.org/10.1111/j.1600-0897.1989.tb00638.x
Duan YG, Yu CF, Novak N, Bieber T, Zhu CH, Schuppe HC, Haidl G, Allam JP (2011) Immunodeviation towards a Th17 immune response associated with testicular damage in azoospermic men. Int J Androl. https://doi.org/10.1111/j.1365-2605.2010.01137.x
Fayomi A, David S, Doungkamchan C, Orwig KE (2018) Spermatogonial stem cells and sspermatogenesis in mice, monkeys and men. Stem Cell Res 29:107–214. https://doi.org/10.1016/j.scr.2018.04.009
Ferreiro ME, Amarilla MS, Glienke L, Méndez CS, González C, Jacobo PV, Sobarzo CM, De Laurentiis A, Ferraris MJ, Theas MS (2019) The inflammatory mediators TNFα and nitric oxide arrest spermatogonia GC-1 cell cycle. Reprod Biol 19:329–339. https://doi.org/10.1016/j.repbio.2019.11.001
Fijak M, Pilatz A, Hedger MP, Nicolas N, Bhushan S, Michel V, Tung KSK, Schuppe HC, Meinhardt A (2018) Infectious, inflammatory and “autoimmune” male factor infertility: how do rodent models inform clinical practice? Hum Reprod Update 24:416–441. https://doi.org/10.1093/humupd/dmy009
Frungieri MB, Calandra RS, Lustig L, Meineke V, Köhn FM, Vogt HJ, Mayerhofer A (2002) Number, distribution pattern, and identification of macrophages in the testes of infertile men. Fertil Steril 78:298–306. https://doi.org/10.1016/S0015-0282(02)03206-5
Gónzalez L, Amarilla MS, Méndez S, Glienke L, Oxilia H, Fulco MF, Jacobo P, Sobarzo CM, TM (2018) Study of spermatogonia population in infertile patients with immune cells infiltrates. Rev Med 78:245–246
Hakovirta H, Syed V, Jégou B, Parvinen M (1995) Function of interleukin-6 as an inhibitor of meiotic DNA synthesis in the rat seminiferous epithelium. Mol Cell Endocrinol 108:193–198. https://doi.org/10.1016/0303-7207(95)03475-M
Hentrich A, Wolter M, Szardening-Kirchner C, Lüers GH, Bergmann M, Kliesch S, Konrad L (2017) Reduced numbers of Sertoli, germ, and spermatogonial stem cells in impaired spermatogenesis. Mod Pathol 30:311. https://doi.org/10.1038/modpathol.2016.194
Hess RA (1990) Quantitative and qualitative characteristics of the stages and transitions in the cycle of the rat seminiferous epithelium: light microscopic observations of perfusion-fixed and plastic-embedded testes. Biol Reprod 43:525–542. https://doi.org/10.1095/biolreprod43.3.525
Huckins C (1971a) Cell cycle properties of differentiating spermatogonia in adult Sprague- Dawley rats. Cell Tissue Kinet 4:139–154. https://doi.org/10.1111/j.1365-2184.1971.tb01524.x
Huckins C (1971b) The spermatogonial stem cell population. Cell Tissue Kinet 4:313–334. https://doi.org/10.1111/j.1365-2184.1971.tb01543.x
Jarazo-Dietrich S, Jacobo P, Pérez CV, Guazzone VA, Lustig L, Theas MS (2012) Up regulation of nitric oxide synthase-nitric oxide system in the testis of rats undergoing autoimmune orchitis. Immunobiology 217:778–787. https://doi.org/10.1016/j.imbio.2012.04.007
Jarazo-Dietrich S, Fass MI, Jacobo PV, Sobarzo CMA, Lustig L, Theas MS (2015) Inhibition of NOS-NO system prevents autoimmune orchitis development in rats: Relevance of NO released by testicular macrophages in germ cell apoptosis and testosterone secretion. PLoS ONE. https://doi.org/10.1371/journal.pone.0128709
Krishnamurthy H, Danilovich N, Morales CR, Sairam MR (2000) Qualitative and quantitative decline in spermatogenesis of the follicle- stimulating hormone receptor knockout (FORKO) mouse. Biol Reprod 62:1146–1159. https://doi.org/10.1095/biolreprod62.5.1146
Leblond CP, Clermont Y (1952) Definition of the stages of the cycle of the seminiferous epithelium in the rat. Ann N Y Acad Sci 55:548–573. https://doi.org/10.1111/j.1749-6632.1952.tb26576.x
Li MWM, Xia W, Mruk DD, Wang CQF, Yan HHN, Siu MKY, Lui WY, Lee WM, Cheng CY (2006) Tumor necrosis factor α reversibly disrupts the blood-testis barrier and impairs Sertoli-germ cell adhesion in the seminiferous epithelium of adult rat testes. J Endocrinol 190:313–329. https://doi.org/10.1677/joe.1.06781
Linakis JG, Cunningham CL (1979) Effects of concentration of ethanol injected intraperitoneally on taste aversion, body temperature, and activity. Psychopharmacology 64:61–65. https://doi.org/10.1007/BF00427346
Lustig L, Guazzone VA, Theas MS, Pleuger C, Jacobo P, Pérez CV, Meinhardt A, Fijak M (2020) Pathomechanisms of autoimmune based testicular inflammation. Front Immunol 11:1–8. https://doi.org/10.3389/fimmu.2020.583135
Mayerhofer A, Walenta L, Mayer C, Eubler K, Welter H (2018) Human testicular peritubular cells, mast cells and testicular inflammation. Andrologia 50:e13055. https://doi.org/10.1111/and.13055
Meineke V, Frungieri MB, Jessberger B, Vogt HJ, Mayerhofer A (2000) Human testicular mast cells contain tryptase: increased mast cell number and altered distribution in the testes of infertile men. Fertil Steril. https://doi.org/10.1016/S0015-0282(00)00626-9
Muñoz-Velasco I, Ortíz R, Echeverría OM, Escobar ML, Vázquez-Nin GH (2013) Characterization of the pre-meiotic S phase through Incorporation of BrdU during spermatogenesis in the Rat. J Histochem Cytochem 61:680–689. https://doi.org/10.1369/0022155413496639
Pérez CV, Sobarzo CM, Jacobo PV, Pellizzari EH, Cigorraga SB, Denduchis B, Lustig L (2012) Loss of occludin expression and impairment of blood-testis barrier permeability in rats with autoimmune orchitis: effect of interleukin 6 on Sertoli cell tight junctions. Biol Reprod 87:1–12. https://doi.org/10.1095/biolreprod.112.101709
Pilatz A, Kilb J, Kaplan H, Fietz D, Hossain H, Schüttler CG, Diemer T, Bergmann M, Domann E, Weidner W, Wagenlehner F, Schuppe HC (2019) High prevalence of urogenital infection/inflammation in patients with azoospermia does not impede surgical sperm retrieval. Andrologia 51:1–10. https://doi.org/10.1111/and.13401
Riccioli A, Filippini A, De Cesaris P, Barbacci E, Stefanini M, Starace G, Ziparo E (1995) Inflammatory mediators increase surface expression of integrin ligands, adhesion to lymphocytes, and secretion of interleukin 6 in mouse Sertoli cells. Proc Natl Acad Sci U S A 92:5808–5812. https://doi.org/10.1073/pnas.92.13.5808
Rival C, Theas MS, Guazzone VA, Lustig L (2006) Interleukin-6 and IL-6 receptor cell expression in testis of rats with autoimmune orchitis. J Reprod Immunol. https://doi.org/10.1016/j.jri.2005.10.006
Savitt J, Singh D, Zhang C, Chen LC, Folmer J, Shokat KM, Wright WW (2012) The in vivo response of stem and other undifferentiated spermatogonia to the reversible inhibition of glial cell line-derived neurotrophic factor signaling in the adult. Stem Cells 30:732–740. https://doi.org/10.1002/stem.1028
Sezer C, Koksal IT, Usta MF, Gulkesen KH, Erdogru T, Ciftcioglu A, Baykara M (2005) Relationship between mast cell and iNOS expression in testicular tissue associated with infertility. Arch Androl 51:149–158. https://doi.org/10.1080/014850190518161
Sharma M, Braun RE (2018) Cyclical expression of GDNF is required for spermatogonial stem cell homeostasis. Dev. https://doi.org/10.1242/dev.151555
Shiloh Y (2014) ATM: Expanding roles as a chief guardian of genome stability. Exp Cell Res 329:154–161. https://doi.org/10.1016/j.yexcr.2014.09.002
Shiloh Y (2003) ATM and related protein kinases: Safeguarding genome integrity. Nat Rev Cancer 3:155–168. https://doi.org/10.1038/nrc1011
Steger K, Aleithe I, Behre H, Bergmann M (1998) The proliferation of spermatogonia in normal and pathological human seminiferous epithelium: an immunohistochemical study using monoclonal antibodies against Ki-67 protein and proliferating cell nuclear antigen. Mol Hum Reprod 4:227-233. https://doi.org/10.1093/molehr/4.3.227
Suescun MO, Rival C, Theas MS, Calandra RS, Lustig L (2003) Involvement of tumor necrosis factor-α in the pathogenesis of autoimmune orchitis in rats. Biol Reprod 68:2114-2121. https://doi.org/10.1095/biolreprod.102.011189
Takubo K, Ohmura M, Azuma M, Nagamatsu G, Yamada W, Arai F, Hirao A, Suda T (2008) Stem cell defects in ATM-deficient undifferentiated spermatogonia through DNA damage-induced cell-cycle arrest. Cell Stem Cell 2:170–182. https://doi.org/10.1016/j.stem.2007.10.023
Theas MS, Rival C, Dietrich SJ, Guazzone VA, Lustig L (2006) Death receptor and mitochondrial pathways are involved in germ cell apoptosis in an experimental model of autoimmune orchitis. Hum Reprod 21:1734–1742. https://doi.org/10.1093/humrep/del066
Theas MS, Rival C, Jarazo-Dietrich S, Jacobo P, Guazzone VA, Lustig L (2008) Tumour necrosis factor-α released by testicular macrophages induces apoptosis of germ cells in autoimmune orchitis. Hum Reprod 23:1865–1872. https://doi.org/10.1093/humrep/den240
van Kroonenburgh MJ, Beck JL, Scholtz JW, Hacker-Klom U, Herman CJ (1985) DNA analysis and sorting of rat testis cells using two-parameter flow cytometry. Cytometry 6:321–326. https://doi.org/10.1002/cyto.990060408
Wing T-Y, Christensen AK (1982) Morphometric studies on rat seminiferous tubules. Am J Anat 165:13–25. https://doi.org/10.1002/aja.1001650103
Xiong Y, Hales DB (1993) The role of tumor necrosis factor-a in the regulation of mouse leydig cell steroidogenesis. Endocrinology 132:2438–2444. https://doi.org/10.1210/endo.132.6.8504748
Zhang L, Tang J, Haines CJ, Feng H, Lai L, Teng X, Han Y (2011) C-Kit and its related genes in spermatogonial differentiation. Spermatogenesis 1, 186–194. https://doi.org/10.4161/spmg.1.3.17760
Acknowledgements
We thank the Instituto Nacional de Microbiología “A. Malbrán”, División Vacunas Bacterianas for their generous gift of Bordetella pertussis, and M. Imsem, and C. García for their technical assistance.
Funding
Universidad de Buenos Aires (UBA 20020150100104BA) and the Agencia Nacional de Promoción Científica y Tecnológica (ANPCyT PICT 0497).
Author information
Authors and Affiliations
Contributions
María Eugenia Ferreiro, Cinthia Soledad Méndez, and Leilane Glienke: investigation, methodology, formal analysis; Maria Jimena Ferraris and Patricia Verónica Jacobo: conceptualization, review and editing; Cristian Marcelo Sobarzo: visualization; Daniel Pisera and Livia Lustig: supervision, review and editing; Maria Susana Theas: conceptualization supervision, validation, resources, original draft.
Corresponding author
Ethics declarations
Ethics approval
The use of rats followed the National Institutes of Health (NIH) guidelines for care and use of experimental animals, and ethical approval was granted by local the committee: CICUAL-Facultad de Medicina, Universidad de Buenos Aires [RES (CD): 2029/2019].
Informed consent
Not applicable.
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Maria Eugenia Ferreiro and Cinthia Soledad Méndez are co-author.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ferreiro, M.E., Méndez, C.S., Glienke, L. et al. Unraveling the effect of the inflammatory microenvironment in spermatogenesis progression. Cell Tissue Res 392, 581–604 (2023). https://doi.org/10.1007/s00441-022-03703-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-022-03703-z