Abstract
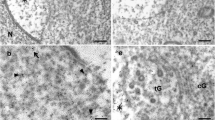
The role of microtubule-based trafficking in acrosomal biogenesis was examined by studying the effects of colchicine on spermiogenesis. In electron micrographs of untreated cap-phase mouse spermatids, coated vesicles were always seen on the apex and caudal margins of the developing acrosomal cap. The increase in volume and the accumulation of materials in the acrosome during the Golgi and cap phases were observed to occur via fusion of vesicles at various sites on the growing acrosome. By studying the acid phosphatase localization pattern and colchicine-treated spermatids, the role of clathrin-coated vesicles became clear. Coated vesicle formation at the caudal margin of the acrosome appeared to be responsible for the spreading and shaping of the acrosome over the surface of the nucleus and also established distinct regional differences in the acrosome. In colchicine-treated spermatids, the Golgi apparatus lost its typical membranous stack conformation and disintegrated into many small vesicles. Acrosome formation was retarded, and there was discordance of the spread of the acrosomal cap with that of the modified nuclear envelope. Many symplasts were also found because of the breakdown of intercellular bridges. Colchicine treatment thus indicated that microtubule-dependent trafficking of transport vesicles between the Golgi apparatus and the acrosome plays a vital role in acrosomal biogenesis. In addition, both anterograde and retrograde vesicle trafficking are extensively involved and seem to be equally important in acrosome formation.





Similar content being viewed by others
References
Bonifacino JS, Glick BS (2004) The mechanisms of vesicle budding and fusion. Cell 116:153–166
Bozzola JJ, Polakoski K, Haas N, Russell LD, Campbell P, Peterson RN (1991) Localization of boar sperm proacrosin during spermatogenesis and during sperm maturation in the epididymis. Am J Anat 192:129–141
Clermont Y, Leblond CP (1955) Spermiogenesis of man, monkey, ram and other mammals as shown by the periodic acid-Schiff technique. Am J Anat 96:229–253
Dell KR (2003) Dynactin polices two-way organelle traffic. J Cell Biol 160:291–293
Fouquet JP, Valentin A, Kann ML (1992) Perinuclear cytoskeleton of acrosome-less spermatids in the blind sterile mutant mouse. Tissue Cell 24:655–665
Friend DS, Fawcett DW (1974) Membrane differentiations in freeze-fractured mammalian sperm. J Cell Biol 63:641–664
Galli T, Haucke V (2004) Cycling of synaptic vesicles: how far? How fast! Sci STKE 264:1–12
Griffiths G, Warren G, Stuhlfauth I, Jockusch BM (1981) The role of clathrin-coated vesicles in acrosome formation. Eur J Cell Biol 26:52–60
Griffiths GW (1979) Transport of glial cell acid phosphatase by endoplasmic reticulum into damaged axons. J Cell Sci 36:361–389
Handel MA (1979) Effects of colchicine on spermiogenesis in the mouse. J Embryol Exp Morphol 51:73–83
Ho HC, Tang CY, Suarez SS (1999) Three-dimensional structure of the Golgi apparatus in mouse spermatids: a scanning electron microscopic study. Anat Rec 256:189–194
Holt WV (1979) Development and maturation of the mammalian acrosome. A cytochemical study using phosphotungstic acid staining. J Ultrastruct Res 68:58–71
Jordens I, Fernandez-Borja M, Marsman M, Dusseljee S, Janssen L, Calafat J, Janssen H, Wubbolts R, Neefjes J (2001) The Rab7 effector protein RILP controls lysosomal transport by inducing the recruitment of dynein-dynactin motors. Curr Biol 11:1680–1685
Kierszenbaum AL, Rivkin E, Tres LL (2003) Acroplaxome, an F—actin—keratin—containing plate, anchors the acrosome to the nucleus during shaping of the spermatid head. Mol Biol Cell 14:4628–4640
Kierszenbaum AL, Tres LL (2004) The acrosome—acroplaxome—manchette complex and the shaping of the spermatid head. Arch Histol Cytol 67:271–284
Kraemer J, Schmitz F, Drenckhahn D (1999) Cytoplasmic dynein and dynactin as likely candidates for microtubule-dependent apical targeting of pancreatic zymogen granules. Eur J Cell Biol 78:265–277
Leblond CP, Clermont Y (1952) Spermiogenesis of rat, mouse, hamster and guinea pig as revealed by the “periodic acid-fuchsin sulfurous acid” technique. Am J Anat 90:167–215
Moreno RD, Ramalho-Santos J, Sutovsky P, Chan EK, Schatten G (2000) Vesicular traffic and Golgi apparatus dynamics during mammalian spermatogenesis: implications for acrosome architecture. Biol Reprod 63:89–98
Moreno RD, Schatten G (2000) Microtubule configurations and post-translational alpha-tubulin modifications during mammalian spermatogenesis. Cell Motil Cytoskeleton 46:235–246
Parvinen M, Wright WW, Phillips DM, Mather JP, Musto NA, Bardin CW (1983) Spermatogenesis in vitro: completion of meiosis and early spermiogenesis. Endocrinology 112:1150–1152
Pelletier RM, Friend DS (1983) Development of membrane differentiations in the guinea pig spermatid during spermiogenesis. Am J Anat 167:119–141
Peterson RN, Bozzola J, Polakoski K (1992) Protein transport and organization of the developing mammalian sperm acrosome. Tissue Cell 24:1–15
Ramalho-Santos J, Moreno RD, Wessel GM, Chan EK, Schatten G (2001) Membrane trafficking machinery components associated with the mammalian acrosome during spermiogenesis. Exp Cell Res 267:45–60
Rindler MJ, Ivanov IE, Sabatini DD (1987) Microtubule-acting drugs lead to the nonpolarized delivery of the influenza hemagglutinin to the cell surface of polarized Madin-Darby canine kidney cells. J Cell Biol 104:231–241
Russell LD, Lee IP, Ettlin R, Peterson RN (1983) Development of the acrosome and alignment, elongation and entrenchment of spermatids in procarbazine-treated rats. Tissue Cell 15:615–626
Russell LD, Malone JP, MacCurdy DS (1981) Effect of the microtubule disrupting agents, colchicine and vinblastine, on seminiferous tubule structure in the rat. Tissue Cell 13:349–367
Russell LD, Russell JA, MacGregor GR, Meistrich ML (1991) Linkage of manchette microtubules to the nuclear envelope and observations of the role of the manchette in nuclear shaping during spermiogenesis in rodents. Am J Anat 192:97–120
Russell LD, Ying L, Overbeek PA,(1994) Insertional mutation that causes acrosomal hypo-development: its relationship to sperm head shaping. Anat Rec 238:437–453
Schnapp BJ, Vale RD, Sheetz MP, Reese TS (1985) Single microtubules from squid axoplasm support bidirectional movement of organelles. Cell 40:455–462
Sheetz MP, Vale R, Schnapp B, Schroer T, Reese T (1987) Movements of vesicles on microtubules. Ann N Y Acad Sci 493:409–416
Sinowatz F, Wrobel KH (1981) Development of the bovine acrosome. An ultrastructural and cytochemical study. Cell Tissue Res 219:511–524
Sotomayor RE, Handel MA (1986) Failure of acrosome assembly in a male sterile mouse mutant. Biol Reprod 34:171–182
Stackpole CW, Devorkin D (1974) Membrane organization in mouse spermatozoa revealed by freeze-etching. J Ultrastruct Res 49:167–187
Tanaka K (1989) High resolution scanning electron microscopy of the cell. Biol Cell 65:89–98
Tang XM, Lalli MF, Clermont Y (1982) A cytochemical study of the Golgi apparatus of the spermatid during spermiogenesis in the rat. Am J Anat 163:283–294
Tanii I, Yoshinaga K, Toshimori K (1998) The effects of brefeldin A on acrosome formation and protein transport to the acrosome in organ cultures of rat seminiferous tubules. J Electron Microsc 47:161–167
Thyberg J, Moskalewski S (1985) Microtubules and the organization of the Golgi complex. Exp Cell Res 159:1–16
Ventela S, Mulari M, Okabe M, Tanaka H, Nishimune Y, Toppari J, Parvinen M (2000) Regulation of acrosome formation in mice expressing green fluorescent protein as a marker. Tissue Cell 32:501–507
Weber JE, Russell LD (1987) A study of intercellular bridges during spermatogenesis in the rat. Am J Anat 180:1–24
Yamaguchi N, Takanezawa Y, Koizumi H, Umezu-Goto M, Aoki J, Arai H, (2004) Expression of NUDEL in manchette and its implication in spermatogenesis. FEBS Lett 566:71–76
Yanagimachi R (1994) Mammalian fertilization. In: Knobil E, Neill J (eds) The physiology of reproduction. Raven, New York, pp 189–317
Yang WX, Sperry AO (2003) C-terminal kinesin motor KIFC1 participates in acrosome biogenesis and vesicle transport. Biol Reprod 69:1719–1729
Acknowledgements
We express our gratitude to our mentors Jia-Chyuan Chen and Hsiao-Hsu Jen for introducing us to the world of electron microscopy. We also thank Dr. Susan S. Suarez for critical review of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by grants 83-0211-B-002-184 and 93-2320-B-320-012 from the National Science Council, Taiwan, Republic of China.
Rights and permissions
About this article
Cite this article
Huang, WP., Ho, HC. Role of microtubule-dependent membrane trafficking in acrosomal biogenesis. Cell Tissue Res 323, 495–503 (2006). https://doi.org/10.1007/s00441-005-0097-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-005-0097-9