Abstract
Background
Numerous studies have reported the preventive and protective effects of aspirin in patients with rectal cancer. However, it is not clear whether aspirin can be used as an assistance drug in preoperative neoadjuvant chemoradiotherapy. Therefore, this study will explore the efficacy of aspirin as an adjuvant agent in rectal cancer neoadjuvant chemoradiotherapy.
Methods
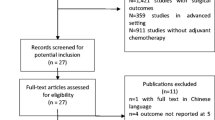
A literature search was performed using the electronic platforms to obtain relevant research studies published up to Jan 2020. The search was limited to papers published in English or Chinese language. Confidence intervals of research endpoints in each study were extracted and merged. The meta-analysis was performed using Stata12.0 software. Furthermore, we performed trial sequential analysis (TSA) to evaluate the robustness of our findings and to obtain a more conservative estimation.
Results
A total of 5 studies including 977 patients were identified to be eligible for this meta-analysis. Compared with control group, aspirin group significantly increased pathologic complete response rate from 16.5 to 22.3% (RR 1.41, 95% CI 1.01–1.96, P = 0.041), partial remission rate from 21.8 to 45.7% (RR 1.87, 95% CI 1.37–2.54, P < 0.001), and tumor down-staging rate from 44.4 to 63.8% (RR 1.43, 95% CI 1.17–1.75, P = 0.001). Moreover, aspirin group can reduce local recurrence rate (RR 0.37, 95% CI 0.17–0.84, P = 0.017), improve 3-year survival rate (RR 1.24, 95% CI 1.12–1.36, P < 0.001), and 5-year survival rate (RR 1.29, 95% CI 1.14–1.46, P < 0.001). TSA shows that the meta-analysis results of pathologic complete response rate and local recurrence rate may be a false positive. Furthermore, the meta-analysis results of other study endpoints were further confirmed by TSA.
Conclusion
Aspirin, as an adjuvant agent, can enhance the effect of neoadjuvant chemoradiotherapy and improve the prognosis of patients with rectal cancer. Neoadjuvant therapy combined with aspirin may be considered a better option for preoperative rectal cancer patients.





Similar content being viewed by others
References
Ashktorab H, Dawkins FW, Mohamed R, Larbi D, Smoot DT (2005) Apoptosis induced by aspirin and 5-fluorouracil in human colonic adenocarcinoma cells. Dig Dis Sci 50:1025–1032. https://doi.org/10.1007/s10620-005-2698-2
Baron JA et al (2003) A randomized trial of aspirin to prevent colorectal adenomas. NEJM 348:891–899
Bastiaannet E et al (2012) Use of aspirin postdiagnosis improves survival for colon cancer patients. Br J Cancer 106:1564–1570. https://doi.org/10.1038/bjc.2012.101
Chan AT, Ogino S, Fuchs CS (2009) Aspirin use and survival after diagnosis of colorectal cancer. JAMA 302:649–658. https://doi.org/10.1001/jama.2009.1112
Eberhart CE, Coffey RJ, Radhika A, Giardiello FM, Ferrenbach S, DuBois RN (1994) Up-regulation of cyclooxygenase 2 gene expression in human colorectal adenomas and adenocarcinomas. Gastroenterology 107:1183–1188. https://doi.org/10.1016/0016-5085(94)90246-1
Fu J, Xu Y, Yang Y, Liu Y, Ma L, Zhang Y (2019) Aspirin suppresses chemoresistance and enhances antitumor activity of 5-Fu in 5-Fu-resistant colorectal cancer by abolishing 5-Fu-induced NF-kappaB activation. Sci Rep 9:16937. https://doi.org/10.1038/s41598-019-53276-1
Fuchs C, Meyerhardt JA, Heseltine DL, Niedzwiecki D, Mayer RJ (2005) Influence of regular aspirin use on survival for patients with stage III colon cancer: findings from intergroup trial CALGB 89803. J Clin Oncol 23:3530–3530
Gash KJ, Chambers AC, Cotton DE, Williams AC, Thomas MG (2017) Potentiating the effects of radiotherapy in rectal cancer: the role of aspirin, statins and metformin as adjuncts to therapy. Br J Cancer 117:210–219. https://doi.org/10.1038/bjc.2017.175
Gay LJ, Felding-Habermann B (2011) Contribution of platelets to tumour metastasis. Nat Rev Cancer 11:123–134. https://doi.org/10.1038/nrc3004
Gonzalez I, Bauer PS, Chapman WC Jr, Alipour Z, Rais R, Liu J, Chatterjee D (2019) Clinicopathologic determinants of pathologic treatment response in neoadjuvant treated rectal adenocarcinoma. Ann Diagn Pathol 45:151452. https://doi.org/10.1016/j.anndiagpath.2019.151452
Guo X, Ni B, Chen R, Zhu Z, Lin L (2018) Clinical study of aspirin as adjuvant agent during neoadjuvant chemradiotherapy for rectal cancer Lab. Med Clin 15:106–109
Hamada T et al (2017) Aspirin use and colorectal cancer survival according to tumor CD274 (programmed cell death 1 ligand 1) expression status. J Clin Oncol 35:1836–1844
Hardie C, Jung Y, Jameson M (2016) Effect of statin and aspirin use on toxicity and pathological complete response rate of neo-adjuvant chemoradiation for rectal cancer. Asia Pac J Clin Oncol 12:167–173. https://doi.org/10.1111/ajco.12468
Higgins JP, Whitehead A, Simmonds M (2011) Sequential methods for random-effects meta-analysis. Stat Med 30:903–921. https://doi.org/10.1002/sim.4088
Hua X et al (2017) Timing of aspirin and other nonsteroidal anti-inflammatory drug use among patients with colorectal cancer in relation to tumor markers and survival. J Clin Oncol Off J Am Soc Clin Oncol 35:2806–2813
Iqbal U, Yang HC, Jian WS, Yen Y, Li YJ (2017) Does aspirin use reduce the risk for cancer? J Investig Med Off Publ Am Fed Clin Res 65:391–392. https://doi.org/10.1136/jim-2016-000275
Katz MS, Minsky BD, Saltz LB, Riedel E, Chessin DB, Guillem JG (2005) Association of statin use with a pathologic complete response to neoadjuvant chemoradiation for rectal cancer. Int J Radiat Oncol Biol Phys 62:1363–1370. https://doi.org/10.1016/j.ijrobp.2004.12.033
Kune GA, Kune S, Watson LF (1988) Colorectal cancer risk, chronic illnesses, operations, and medications: case control results from the Melbourne Colorectal Cancer Study. Cancer Res 48:4399–4404
Latkauskas T, Paskauskas S, Dambrauskas Z, Gudaityte J, Saladzinskas S, Tamelis A, Pavalkis D (2010) Preoperative chemoradiation vs radiation alone for stage II and III resectable rectal cancer: a meta-analysis. Colorectal Dis Off J Assoc Coloproctol G B Irel 12:1075–1083. https://doi.org/10.1111/j.1463-1318.2009.02015.x
Li P et al (2015) Aspirin use after diagnosis but not prediagnosis improves established colorectal cancer survival: a meta-analysis. Gut 64:1419–1425. https://doi.org/10.1136/gutjnl-2014-308260
Liao X, Lochhead P, Nishihara R, Morikawa T, Ogino S (2012) Aspirin use, tumor PIK3CA mutation, and colorectal-cancer survival. N Engl J Med 367:1596–1606
Liu Y, Gao D, Zhong J, Wan Y, Li X, Jin G, Wang X (2014) Synergistic anti-proliferation effect of aspirin and 5-fluorouracil on colon cancer cells and its mechanism. Chin J Pathophysiol 30:988–993
Ludmir EB, Palta M, Willett CG, Czito BG (2017) Total neoadjuvant therapy for rectal cancer: an emerging option. Cancer 123:1497–1506. https://doi.org/10.1002/cncr.30600
Martin ST, Heneghan HM, Winter DC (2012) Systematic review and meta-analysis of outcomes following pathological complete response to neoadjuvant chemoradiotherapy for rectal cancer. Br J Surg 99:918–928. https://doi.org/10.1002/bjs.8702
Morris ZS et al (2016) Increased tumor response to neoadjuvant therapy among rectal cancer patients taking angiotensin-converting enzyme inhibitors or angiotensin receptor blockers. Cancer 122:2487–2495. https://doi.org/10.1002/cncr.30079
Nishihara R et al (2013) Aspirin use and risk of colorectal cancer according to BRAF mutation status. JAMA 309:2563–2571. https://doi.org/10.1001/jama.2013.6599
Restivo A, Cocco IM, Casula G, Scintu F, Cabras F, Scartozzi M, Zorcolo L (2015) Aspirin as a neoadjuvant agent during preoperative chemoradiation for rectal cancer. Br J Cancer 113:1133–1139. https://doi.org/10.1038/bjc.2015.336
Rothwell PM, Wilson M, Elwin CE, Norrving B, Algra A, Warlow CP, Meade TW (2010) Long-term effect of aspirin on colorectal cancer incidence and mortality: 20-year follow-up of five randomised trials. Lancet 376:1741–1750. https://doi.org/10.1016/S0140-6736(10)61543-7
Sauer R et al (2012) Preoperative versus postoperative chemoradiotherapy for locally advanced rectal cancer: results of the German CAO/ARO/AIO-94 randomized phase III trial after a median follow-up of 11 years. J Clin Oncol Off J Am Soc Clin Oncol 30:1926–1933. https://doi.org/10.1200/JCO.2011.40.1836
Siegel RL, Miller KD, Fedewa SA, Ahnen DJ, Meester RGS, Barzi A, Jemal A (2017) Colorectal cancer statistics, 2017. CA Cancer J Clin 67:177–193. https://doi.org/10.3322/caac.21395
Siegel RL, Miller KD, Jemal A (2020) Cancer statistics, 2020. CA Cancer J Clin 70:7–30. https://doi.org/10.3322/caac.21590
Skelin M, Javor E, Lucijanic M (2019) Aspirin use and the risk of cancer. JAMA Oncol 5:912–913. https://doi.org/10.1001/jamaoncol.2019.0611
Smith CA, Kachnic LA (2018) Evolving treatment paradigm in the treatment of locally advanced rectal cancer. J Natl Compr Cancer Net JNCCN 16:909–915. https://doi.org/10.6004/jnccn.2018.7032
Stang A (2010) Critical evaluation of the Newcastle–Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 25:603–605. https://doi.org/10.1007/s10654-010-9491-z
Wetterslev J, Thorlund K, Brok J, Gluud C (2009) Estimating required information size by quantifying diversity in random-effects model meta-analyses. BMC Med Res Methodol 9:86. https://doi.org/10.1186/1471-2288-9-86
Ye XF, Wang J, Shi WT, He J (2014) Relationship between aspirin use after diagnosis of colorectal cancer and patient survival: a meta-analysis of observational studies. Br J Cancer 111:2172–2179. https://doi.org/10.1038/bjc.2014.481
Zaborowski A, Stakelum A, Winter DC (2019) Systematic review of outcomes after total neoadjuvant therapy for locally advanced rectal cancer. Br J Surg 106:979–987. https://doi.org/10.1002/bjs.11171
Zorcolo L, Rosman AS, Restivo A, Pisano M, Nigri GR, Fancellu A, Melis M (2012) Complete pathologic response after combined modality treatment for rectal cancer and long-term survival: a meta-analysis. Ann Surg Oncol 19:2822–2832. https://doi.org/10.1245/s10434-011-2209-y
Funding
No sources of funding were used to conduct this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, B., Huang, Y. Effect of aspirin use on neoadjuvant chemoradiotherapy for rectal cancer: a meta-analysis with trial sequential analysis. J Cancer Res Clin Oncol 146, 2161–2171 (2020). https://doi.org/10.1007/s00432-020-03222-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-020-03222-w