Abstract
Purpose
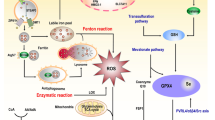
Tumor cells generally exhibit higher levels of reactive oxygen species (ROS), however, when stressed, tumor cells can undergo a process of ‘Redox Resetting’ to acquire a new redox balance with stronger antioxidant systems that enable cancer cells to become resistant to radiation therapy (RT). Here, we describe how RT affects the oxidant/antioxidant balance in human embryonal (RD) and alveolar (RH30) rhabdomyosarcoma (RMS) cell lines, investigating on the molecular mechanisms involved.
Methods
Radiations were delivered using an x-6 MV photon linear accelerator and their effects were assessed by vitality and clonogenic assays. The expression of specific antioxidant-enzymes, such as Superoxide Dismutases (SODs), Catalase (CAT) and Glutathione Peroxidases 4 (GPx4), miRNAs (miR-22, -126, -210, -375, -146a, -34a) and the transcription factor NRF2 was analyzed by quantitative polymerase chain reaction (q-PCR) and western blotting. RNA interference experiments were performed to evaluate the role of NRF2.
Results
Doses of RT higher than 2 Gy significantly affected RMS clonogenic ability by increasing ROS production. RMS rapidly and efficiently brought back ROS levels by up-regulating the gene expression of antioxidant enzymes, miRNAs as well as of NRF2. Silencing of NRF2 restrained the RMS ability to counteract RT-induced ROS accumulation, antioxidant enzyme and miRNA expression and was able to increase the abundance of γ-H2AX, a biomarker of DNA damage, in RT-treated cells.
Conclusions
Taken together, our data suggest the strategic role of oxidant/antioxidant balance in restraining the therapeutic efficiency of RT in RMS treatment and identify NRF2 as a new potential molecular target whose inhibition might represent a novel radiosensitizing therapeutic strategy for RMS clinical management.






Similar content being viewed by others
References
Alexandre J, Batteux F, Nicco C, Chéreau C, Laurent A, Guillevin L, Weill B, Goldwasser F (2006) Accumulation of hydrogen peroxide is an early and crucial step for paclitaxel-induced cancer cell death both in vitro and in vivo. Int J Cancer 119:41–48
Arndt CA, Rose PS, Folpe AL, Laack NN (2012) Common musculoskeletal tumors of childhood and adolescence. Mayo Clin Proc 87:475–487
Azzam EI, Jay-Gerin JP, Pain D (2012) Ionizing radiation-induced metabolic oxidative stress and prolonged cell injury. Cancer Lett 327:48–60
Bairati I, Meyer F, Gélinas M, Fortin A, Nabid A, Brochet F, Mercier JP, Têtu B, Harel F, Abdous B, Vigneault E, Vass S, Del Vecchio P, Roy J (2005) Randomized trial of antioxidant vitamins to prevent acute adverse effects of radiation therapy in head and neck cancer patients. J Clin Oncol 23:5805–5813
Baker JR, Vuppusetty C, Colley T, Papaioannou AI, Fenwick P, Donnelly L, Ito K, Barnes PJ (2016) Oxidative stress dependent microRNA-34a activation via PI3Kα reduces the expression of sirtuin-1 and sirtuin-6 in epithelial cells. Sci Rep 6:35871
Banerjee J, Khanna S, Bhattacharya A (2017) MicroRNA regulation of oxidative stress. Oxid Med Cell Longev 2017:2872156
Barr FG (2001) Gene fusions involving PAX and FOX family members in alveolar rhabdomyosarcoma. Oncogene 20:5736–5746
Cheleschi S, De Palma A, Pascarelli NA, Giordano N, Galeazzi M, Tenti S, Fioravanti A (2017) Could oxidative stress regulate the expression of MicroRNA-146a and MicroRNA34 a in human osteoarthritic chondrocyte cultures? Int J Mol Sci 18(12):2660. https://doi.org/10.3390/ijms18122660
Chen X, Stewart E, Shelat AA, Qu C, Bahrami A, Hatley M, Wu G, Bradley C, McEvoy J, Pappo A, Spunt S, Valentine MB, Valentine V, Krafcik F, Lang WH, Wierdl M, Tsurkan L, Tolleman V, Federico SM, Morton C, Lu C, Ding L, Easton J, Rusch M, Nagahawatte P, Wang J, Parker M, Wei L, Hedlund E, Finkelstein D, Edmonson M, Shurtleff S, Boggs K, Mulder H, Yergeau D, Skapek S, Hawkins DS, Ramirez N, Potter PM, Sandoval JA, Davidoff AM, Mardis ER, Wilson RK, Zhang J, Downing JR, Dyer MA, St. Jude Children’s Research Hospital-Washington University Pediatric Cancer Genome Project (2013) Targeting oxidative stress in embryonal rhabdomyosarcoma. Cancer Cell 24:710–724
Ciccarelli C, Vulcano F, Milazzo L, Gravina GL, Marampon F, Macioce G, Giampaolo A, Tombolini V, Di Paolo V, Hassan HJ, Zani BM (2016) Key role of MEK/ERK pathway in sustaining tumorigenicity and in vitro radioresistance of embryonal rhabdomyosarcoma stem-like cell population. Mol Cancer 15:16
Ciccarelli C, Di Rocco A, Gravina GL, Mauro A, Festuccia C, Del Fattore A, Berardinelli P, De Felice F, Musio D, Bouché M, Tombolini V, Zani BM, Marampon F (2018) Disruption of MEK/ERK/c-Myc signaling radiosensitizes prostate cancer cells in vitro and in vivo. J Cancer Res Clin Oncol 144:1685–1699
Ciccarese F, Ciminale V (2017) Escaping death: mitochondrial redox homeostasis in cancer cells. Front Oncol 7:117
Copin JC, Gasche Y, Chan PH (2000) Overexpression of copper/zinc superoxide dismutase does not prevent neonatal lethality in mutant mice that lack manganese superoxide dismutase. Free Radic Biol Med 28:1571–1576
Desouky O, Ding N, Zhou G (2015) Targeted and non-targeted effects of ionizing radiation. J Radiat Res Appl Sci 8:247–254
Drusco A, Croce CM (2017) MicroRNAs and cancer: a long story for short RNAs. Adv Cancer Res 135:1–24
Fabrizio FP, Sparaneo A, Trombetta D, Muscarella LA (2018) Epigenetic versus genetic deregulation of the KEAP1/NRF2 axis in solid tumors: focus on methylation and noncoding RNAs. Oxid Med Cell Longev 2018:2492063
Faggi F, Chiarelli N, Colombi M, Mitola S, Ronca R, Madaro L, Bouche M, Poliani PL, Vezzoli M, Longhena F, Monti E, Salani B, Maggi D, Keller C, Fanzani A (2015) Cavin-1 and Caveolin-1 are both required to support cell proliferation, migration and anchorage-independent cell growth in rhabdomyosarcoma. Lab Investig 95:585–602
Fanzani A, Poli M (2017) Iron, oxidative damage and ferroptosis in rhabdomyosarcoma. Int J Mol Sci 18:1718
Frezza AM, Lee ATJ, Nizri E, Sbaraglia M, Jones RL, Gronchi A, Dei Tos AP, Casali PG (2018) 2018 ESMO sarcoma and GIST symposium: ‘take-home messages’ in soft tissue sarcoma. ESMO Open 3:e000390
Giannattasio S, Megiorni F, Di Nisio V, Del Fattore A, Fontanella R, Camero S, Antinozzi C, Festuccia C, Gravina GL, Cecconi S, Dominici C, Di Luigi L, Ciccarelli C, De Cesaris P, Riccioli A, Zani BM, Lenzi A, Pestell RG, Filippini A, Crescioli C, Tombolini V, Marampon F (2018) Testosterone-mediated activation of androgenic signalling sustains in vitro the transformed and radioresistant phenotype of rhabdomyosarcoma cell lines. J Endocrinol Investig. https://doi.org/10.1007/s40618-018-0900-6
Gravina GL, Festuccia C, Popov VM, Di Rocco A, Colapietro A, Sanità P, Monache SD, Musio D, De Felice F, Di Cesare E, Tombolini V, Marampon F (2016) c-Myc sustains transformed phenotype and promotes radioresistance of embryonal rhabdomyosarcoma cell lines. Radiat Res 185:411–422
Guo J, Yang C, Zhang S, Liang M, Qi J, Wang Z, Peng Y, Sun B (2018) MiR-375 induces ROS and apoptosis in ST cells by targeting the HIGD1A gene. Gene 685:136–142
Hu H, Gatti RA (2011) MicroRNAs: new players in the DNA damage response. J Mol Cell Biol J 3:151–158
Itoh K, Wakabayashi N, Katoh Y, Ishii T, Igarashi K, Engel JD, Yamamoto M (1999) Keap1 represses nuclear activation of antioxidant responsive elements by Nrf2 through binding to the amino-terminal Neh2 domain. Genes Dev 13:76–86
Je EM, An CH, Yoo NJ, Lee SH (2012) Mutational and expressional analyses of NRF2 and KEAP1 in sarcomas. Tumori 98:510–515
Jothi M, Nishijo K, Keller C, Mal AK (2012) AKT and PAX3-FKHR cooperation enforces myogenic differentiation blockade in alveolar rhabdomyosarcoma cell. Cell Cycle 11:895–908
Kansanen E, Kuosmanen SM, Leinonen H, Levonen AL (2013) The Keap1-Nrf2 pathway: mechanisms of activation and dysregulation in cancer. Redox Biol 1:45–49
Kasinski AL, Slack FJ (2011) Epigenetics and genetics. MicroRNAs en route to the clinic: progress in validating and targeting microRNAs for cancer therapy. Nat Rev Cancer 11:849–864
Kuo LJ, Yang LX (2008) Gamma-H2AX - a novel biomarker for DNA double-strand breaks. In Vivo 22:305–309
Lan J, Huang Z, Han J, Shao J, Huang C (2018) Redox regulation of microRNAs in cancer. Cancer Lett 418:250–259
Li M, Marin-Muller C, Bharadwaj U, Chow KH, Yao Q, Chen C (2009) MicroRNAs: control and loss of control in human physiology and disease. World J Surg 33:667–684
Liou GY, Storz P (2010) Reactive oxygen species in cancer. Free Radic Res 44:479–496
Liu Y, Li Q, Zhou L, Xie N, Nice EC, Zhang H, Huang C, Lei Y (2016) Cancer drug resistance: redox resetting renders a way. Oncotarget 7:42740–42761
Llobet D, Eritja N, Encinas M, Sorolla A, Yeramian A, Schoenenberger JA, Llombart-Cussac A, Marti RM, Matias-Guiu X, Dolcet X (2008) Antioxidants block proteasome inhibitor function in endometrial carcinoma cells. Anticancer Drugs 19:115–124
Lovat F, Valeri N, Croce CM (2011) MicroRNAs in the pathogenesis of cancer. Semin Oncol 38:724–733
Magenta A, Greco S, Gaetano C, Martelli F (2013) Oxidative stress and microRNAs in vascular diseases. Int J Mol Sci 14:17319–17346
Marampon F, Gravina GL, Di Rocco A, Bonfili P, Di Staso M, Fardella C, Polidoro L, Ciccarelli C, Festuccia C, Popov VM, Pestell RG, Tombolini V, Zani BM (2011) MEK/ERK inhibitor U0126 increases the radiosensitivity of rhabdomyosarcoma cells in vitro and in vivo by downregulating growth and DNA repair signals. Mol Cancer Ther 10:159–168
Marampon F, Gravina G, Ju X, Vetuschi A, Sferra R, Casimiro M, Pompili S, Festuccia C, Colapietro A, Gaudio E, Di Cesare E, Tombolini V, Pestell RG (2016) Cyclin D1 silencing suppresses tumorigenicity, impairs DNA double strand break repair and thus radiosensitizes androgen-independent prostate cancer cells to DNA damage. Oncotarget 7:5383–5400
Marampon F, Leoni F, Mancini A, Pietrantoni I, Codenotti S, Letizia F, Megiorni F, Porro G, Galbiati E, Pozzi P, Mascagni P, Budillon A, Maggio R, Tombolini V, Fanzani A, Gravina GL, Festuccia C (2018) Histone deacetylase inhibitor ITF2357 (givinostat) reverts transformed phenotype and counteracts stemness in in vitro and in vivo models of human glioblastoma. J Cancer Res Clin Oncol. https://doi.org/10.1007/s00432-018-2800-8
Mates JM, Sanchez-Jimenez FM (2000) Role of reactive oxygen species in apoptosis: implications for cancer therapy. Int J Biochem Cell Biol 32:157–170
McDonald JT, Kim K, Norris AJ, Vlashi E, Phillips TM, Lagadec C, Della Donna L, Ratikan J, Szelag H, Hlatky L, McBride WH (2010) Ionizing radiation activates the Nrf2 antioxidant response. Cancer Res 70:8886–8895
Megiorni F, Gravina GL, Camero S, Ceccarelli S, Del Fattore A, Desiderio V, Papaccio F, McDowell HP, Shukla R, Pizzuti A, Beirinckx F, Pujuguet P, Saniere L, der Aar EV, Maggio R, De Felice F, Marchese C, Dominici C, Tombolini V, Festuccia C, Marampon F (2017) Pharmacological targeting of the ephrin receptor kinase signalling by GLPG1790 in vitro and in vivo reverts oncophenotype, induces myogenic differentiation and radiosensitizes embryonal rhabdomyosarcoma cells. J Hematol Oncol 10:161
Menegon S, Columbano A, Giordano S (2016) The dual roles of NRF2 in cancer. Trends Mol Med 22:578–593
Meza JL, Anderson J, Pappo AS, Meyer WH (2006) Children’s Oncology Group. Analysis of prognostic factors in patients with nonmetastatic rhabdomyosarcoma treated on intergroup rhabdomyosarcoma studies III and IV: the Children’s Oncology Group. J Clin Oncol 24:3844–3851
Moldogazieva NT, Lutsenko SV, Terentiev AA (2018) Reactive oxygen and nitrogen species-induced protein modifications: implication in carcinogenesis and anticancer therapy. Cancer Res 78:6040–6047
Newton WA Jr, Soule EH, Hamoudi AB, Reiman HM, Shimada H, Beltangady M, Maurer H (1988) Histopathology of childhood sarcomas, intergroup rhabdomyosarcoma studies I and II: clinicopathologic correlation. J Clin Oncol 6:67–75
Oberley TD, Oberley LW (1997) Antioxidant enzyme levels in cancer. Histol Histopathol 12:525–535
Pelosi L, Forcina L, Nicoletti C, Scicchitano BM, Musarò A (2017) Increased circulating levels of interleukin-6 induce perturbation in redox-regulated signaling cascades in muscle of dystrophic mice. Oxid Med Cell Longev 2017:1987218
Romania P, Bertaina A, Bracaglia G, Locatelli F, Fruci D, Rota R (2012) Epigenetic deregulation of microRNAs in rhabdomyosarcoma and neuroblastoma and translational perspectives. Int J Mol Sci 13(12):16554–16579
Scicchitano BM, Sorrentino S, Proietti G, Lama G, Dobrowolny G, Catizone A, Binda E, Larocca LM, Sica G (2018) Levetiracetam enhances the temozolomide effect on glioblastoma stem cell proliferation and apoptosis. Cancer Cell Int 18:136
Smith MA, Altekruse SF, Adamson PC, Reaman GH, Seibel NL (2014) Declining childhood and adolescent cancer mortality. Cancer 120:2497–2506
Smolle MA, Leithner A, Posch F, Szkandera J, Liegl-Atzwanger B, Pichler M (2017) MicroRNAs in different histologies of soft tissue sarcoma: a comprehensive review. Int J Mol Sci 18(9):1960. https://doi.org/10.3390/ijms18091960
Sosa V, Moliné T, Somoza R, Paciucci R, Kondoh H, LLeonart ME (2013) Oxidative stress and cancer: an overview. Ageing Res Rev 12:376–390
Spencer RM, Aguiar Junior S, Ferreira FO, Stevanato Filho PR, Kupper BE, Silva ML, Mello CA, Bezerra TS, Lopes A (2017) Neoadjuvant hypofractionated radiotherapy and chemotherapy in high-grade extremity soft tissue sarcomas: phase 2 clinical trial protocol. JMIR Res Protoc 6:e97
Storz P (2005) Reactive oxygen species in tumor progression. Front Biosci 10:1881–1896
Szatrowski TP, Nathan CF (1991) Production of large amounts of hydrogen peroxide by human tumor cells. Cancer Res 51:794–798
Taguchi K, Yamamoto M (2017) The KEAP1-NRF2 system in cancer. Front Oncol 7:85
Taguchi K, Motohashi H, Yamamoto M (2011) Molecular mechanisms of the Keap1–Nrf2 pathway in stress response and cancer evolution. Genes Cells 16:123–140
Tang Q, Len Q, Liu Z, Wang W (2018) Overexpression of miR-22 attenuates oxidative stress injury in diabetic cardiomyopathy via Sirt 1. Cardiovasc Ther. https://doi.org/10.1111/1755-5922.12318
Trachootham D, Alexandre J, Huang P (2009) Targeting cancer cells by ROS-mediated mechanisms: a radical therapeutic approach? Nat Rev Drug Discov 8:579–591
van Kouwenhove M. Kedde M, Agami R (2011) MicroRNA regulation by RNA-binding proteins and its implications for cancer. Nat Rev Cancer 11:644–656
von Mehren M, Randall RL, Benjamin RS, Boles S, Bui MM, Ganjoo KN, George S, Gonzalez RJ, Heslin MJ, Kane JM 3rd, Keedy V, Kim E, Koon H, Mayerson J, McCarter M, McGarry SV, Meyer C, Morris ZS, O’Donnell RJ, Pappo AS, Paz IB, Petersen IA, Pfeifer JD, Riedel RF, Ruo B, Schuetze S, Tap WD, Wayne JD, Bergman MA, Scavone JL (2018) Soft tissue sarcoma, version 2.2018, NCCN clinical practice guidelines in oncology. J Natl Compr Cancer Netw 16:536–563
Wan RJ, Li YH (2018) MicroRNA-146a/NAPDH oxidase4 decreases reactive oxygen species generation and inflammation in a diabetic nephropathy model. Mol Med Rep 17:4759–4766
Williams JR, Zhang Y, Zhou H, Gridley DS, Koch CJ, Slater JM, Little JB (2008) Overview of radiosensitivity of human tumor cells to low-dose-rate irradiation. Int J Radiat Oncol Biol Phys 72:909–917
Wolden SL, Alektiar KM (2010) Sarcomas across the age spectrum. Semin Radiat Oncol 20:45–51
Yang HH, Chen Y, Gao CY, Cui ZT, Yao JM (2017) Protective effects of MicroRNA-126 on human cardiac microvascular endothelial cells against hypoxia/reoxygenation-induced injury and inflammatory response by activating PI3K/Akt/eNOS signaling pathway. Cell Physiol Biochem 42:506–518
Zhang B, Pan X, Cobb GP, Anderson TA (2007) microRNAs as oncogenes and tumor suppressors. Dev Biol 302:1–12
Zhao Q, Mao A, Yan J, Sun C, Di C, Zhou X, Li H, Guo R, Zhang H (2016) Downregulation of Nrf2 promotes radiation-induced apoptosis through Nrf2 mediated Notch signaling in non-small cell lung cancer cells. Int J Oncol 48:765–773
Zhou S, Ye W, Shao Q, Zhang M, Liang J (2013) Nrf2 is a potential therapeutic target in radioresistance in human cancer. Crit Rev Oncol Hematol 88:706–715
Acknowledgements
We are grateful to the Umberto Veronesi Foundation for awarding a post-doctoral fellowship to Francesco Marampon for the year 2018 and “FIVA Confcommercio” for supporting part of our work.
Author information
Authors and Affiliations
Contributions
FMa, SC, FMe and ADF planned experiments; SC, GLG, CF, DM, FDF, VN and ANS performed experiments; CD, LP, AF VT and AF analyze data; SC and PT wrote the paper.
Corresponding author
Ethics declarations
Conflict of interest
The other authors declare that they have no competing interests.
Ethical approval
Studies on animal models were not performed. This article does not contain any studies with human participants performed by any of the authors.
Informed consent
This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Marampon, F., Codenotti, S., Megiorni, F. et al. NRF2 orchestrates the redox regulation induced by radiation therapy, sustaining embryonal and alveolar rhabdomyosarcoma cells radioresistance. J Cancer Res Clin Oncol 145, 881–893 (2019). https://doi.org/10.1007/s00432-019-02851-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-019-02851-0