Abstract
Purpose
Histologic classification of invasive lung adenocarcinomas by predominant subtype has prognostic value. Papillary predominant adenocarcinoma (PPA) reportedly shows poorer prognosis than lepidic predominant adenocarcinoma (LPA); however, biological differences between PPA and LPA are unclear. The purpose of this study was to clarify biological differences between PPA and LPA.
Methods
Clinicopathological characteristics of invasive 62 PPAs and 117 LPAs smaller than 30 mm were investigated. Furthermore, we compared immunochemical staining scores of 9 molecular markers (E-cadherin, S100A4, fibronectin, integrinβ1, ezrin, GLUT1, ALDH1, SOX2 and Nanog) between PPA and LPA. We performed Western blot analysis using ezrin shRNA-knockdown lung adenocarcinoma cell lines to examine whether molecules that are highly expressed in PPA, such as ezrin, affect pAkt. Finally, we performed immunochemical staining to compare pAkt expression level in PPA and LPA.
Results
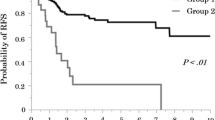
Lymphovascular and pleural invasion and lymph node metastasis were significantly more often detected in PPA than in LPA (lymphatic permeation: 31 vs 3 %, vascular invasion: 35 vs 3 %, pleural invasion: 29 vs 5 %, lymph node metastasis: 18 vs 1 %; all P < 0.01). Immunohistochemical (IHC) study revealed that expression score of ezrin was significantly higher in PPA than in LPA (38.3 vs 15.0; P < 0.01). The level of pAkt decreased in shEzrin-induced PC-9 and A549 cancer cells. Moreover, the IHC staining score of pAkt was significantly higher in PPA than in LPA (13.3 vs 0.0; P < 0.01).
Conclusions
Our results show that the activation of the ezrin–pAkt signaling axis is associated with the more aggressive clinicopathological features of PPA compared with LPA.





Similar content being viewed by others
Abbreviations
- ALDH1:
-
Aldehyde dehydrogenase-1
- EGFR:
-
Epidermal growth factor receptor
- EMT:
-
Epithelial–mesenchymal transition
- GLUT1:
-
Glucose transporter-1
- IHC:
-
Immunohistochemical
- LPA:
-
Lepidic predominant adenocarcinoma
- mTOR:
-
Mammalian target of rapamycin
- pAkt:
-
Phospho-Akt
- PI3K:
-
Phosphoinositide 3-kinase
- PPA:
-
Papillary predominant adenocarcinoma
- RNA:
-
Ribo nucleic acid
- shRNA:
-
Short hairpin RNA
References
Arslan AA, Silvera D, Arju R et al (2012) Atypical ezrin localization as a marker of locally advanced breast cancer. Breast Cancer Res Treat 134:981–988. doi:10.1007/s10549-012-2017-5
Barthel A, Okino ST, Liao J et al (1999) Regulation of GLUT1 gene transcription by the serine/threonine kinase Akt1. J Biol Chem 274:20281–20286. doi:10.1074/jbc.274.29.20281
Bretscher A, Reczek D, Berryman M (1997) Ezrin: a protein requiring conformational activation to link microfilaments to the plasma membrane in the assembly of cell surface structures. J Cell Sci 110(Pt 2):3011–3018
Chadha KS, Khoury T, Yu J et al (2006) Activated Akt and Erk expression and survival after surgery in pancreatic carcinoma. Ann Surg Oncol 13:933–939. doi:10.1245/ASO.2006.07.011
Devesa SS, Bray F, Vizcaino AP, Parkin DM (2005) International lung cancer trends by histologic type: male:female differences diminishing and adenocarcinoma rates rising. Int J Cancer 117:294–299. doi:10.1002/ijc.21183
Di Cristofano C, Leopizzi M, Miraglia A et al (2010) Phosphorylated ezrin is located in the nucleus of the osteosarcoma cell. Mod Pathol 23:1012–1020. doi:10.1038/modpathol.2010.77
Gautreau A, Poullet P, Louvard D, Arpin M (1999) Ezrin, a plasma membrane-microfilament linker, signals cell survival through the phosphatidylinositol 3-kinase/Akt pathway. Proc Natl Acad Sci USA 96:7300–7305. doi:10.1073/pnas.96.13.7300
Hiramatsu M, Ninomiya H, Inamura K et al (2010) Activation status of receptor tyrosine kinase downstream pathways in primary lung adenocarcinoma with reference of KRAS and EGFR mutations. Lung Cancer 70:94–102. doi:10.1016/j.lungcan.2010.01.001
Imamichi Y, Menke A (2007) Signaling pathways involved in collagen-induced disruption of the E-cadherin complex during epithelial-mesenchymal transition. Cells Tissues Organs 185:180–190. doi:10.1159/000101319
Jian Z, Tomizawa Y, Yanagitani N et al (2005) Papillary adenocarcinoma of the lung is a more advanced adenocarcinoma than bronchioloalveolar carcinoma that is composed of two distinct histological subtypes. Pathol Int 55:619–625. doi:10.1111/j.1440-1827.2005.01879.x
Karmakar S, Mukherjee R (2003) Integrin receptors and ECM proteins involved in preferential adhesion of colon carcinoma cells to lung cells. Cancer Lett 196:217–227. doi:10.1016/S0304-3835(03)00208-8
Khanna C, Wan X, Bose S et al (2004) The membrane-cytoskeleton linker ezrin is necessary for osteosarcoma metastasis. Nat Med 10:182–186. doi:10.1038/nm982
Makinoshima H, Takita M, Saruwatari K et al (2015) Signaling through the phosphatidylinositol 3-Kinase (PI3K)/mammalian target of rapamycin (mTOR) axis is responsible for aerobic glycolysis mediated by glucose transporter in epidermal growth factor receptor (EGFR)-mutated lung adenocarcinoma. J Biol Chem. doi:10.1074/jbc.M115.660498
Mcclatchey AI (2003) Merlin and ERM proteins: unappreciated roles in cancer development? Nat Rev Cancer 3:877–883
Motoi N, Szoke J, Riely GJ et al (2008) Lung adenocarcinoma: modification of the 2004 WHO mixed subtype to include the major histologic subtype suggests correlations between papillary and micropapillary adenocarcinoma subtypes, EGFR mutations and gene expression analysis. Am J Surg Pathol 32:810–827. doi:10.1097/PAS.0b013e31815cb162
Pujuguet P, Del Maestro L, Gautreau A et al (2003) Ezrin regulates E-cadherin-dependent adherens junction assembly through Rac1 activation. Mol Biol Cell 14:2181–2191. doi:10.1091/mbc.E02
Russell PA, Wainer Z, Wright GM et al (2011) Does lung adenocarcinoma subtype predict patient survival? J Thorac Oncol 6:1496–1504. doi:10.1097/JTO.0b013e318221f701
Saruwatari K, Ikemura S, Sekihara K, Kuwata T (2016) Aggressive tumor microenvironment of solid predominant lung adenocarcinoma subtype harboring with epidermal growth factor receptor mutations. Lung Cancer 91:7–14
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675. doi:10.1038/nmeth.2089
Sica G, Yoshizawa A, Sima CS et al (2010) A grading system of lung adenocarcinomas based on histologic pattern is predictive of disease recurrence in stage I tumors. Am J Surg Pathol 34:1155–1162. doi:10.1097/PAS.0b013e3181e4ee32
Song G-B, Qin J, Luo Q et al (2005) Adhesion of different cell cycle human hepatoma cells to endothelial cells and roles of integrin beta1. World J Gastroenterol 11:212–215
Sousa V, Bastos B, Silva M, et al (2015) Bronchial-pulmonary adenocarcinoma subtyping relates with different molecular pathways. Rev Port Pneumol 21(5):259–270
Suzuki S, Ishii G, Matsuwaki R et al (2014) Ezrin-expressing lung adenocarcinoma cells and podoplanin-positive fibroblasts form a malignant microenvironment. J Cancer Res Clin Oncol 141:475–484. doi:10.1007/s00432-014-1851-8
Takahashi Y, Ishii G, Aokage K et al (2013) Distinctive histopathological features of lepidic growth predominant node-negative adenocarcinomas 3–5 cm in size. Lung Cancer 79:118–124. doi:10.1016/j.lungcan.2012.10.013
Takeuchi K, Naruki S, Hideko K et al (1994) Perturbation of cell adhesion and microvilli formation by antisense oligonucleotides to ERM family members. J Cell Biol 125:1371–1384
Takuwa T, Ishii G, Nagai K et al (2012) Characteristic immunophenotype of solid subtype component in lung adenocarcinoma. Ann Surg Oncol 19:3943–3952. doi:10.1245/s10434-012-2428-x
Travis WD, Brambilla E, Noguchi M et al (2011) Art: Concise Review International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society international multidisciplinary classification of lung adenocarcinoma. J Thorac Oncol 6:244–285
Travis WD, Brambilla E, Nicholson AG et al (2015) The 2015 World Health Organization classification of lung tumors. J Thorac Oncol 10:1. doi:10.1097/JTO.0000000000000630
Tsao M-S, Marguet S, Le Teuff G et al (2015) Subtype classification of lung adenocarcinoma predicts benefit from adjuvant chemotherapy in patients undergoing complete resection. J Clin Oncol. doi:10.1200/JCO.2014.58.8335
Tynninen O, Carpén O, Jääskeläinen J et al (2004) Ezrin expression in tissue microarray of primary and recurrent gliomas. Neuropathol Appl Neurobiol 30:472–477. doi:10.1111/j.1365-2990.2004.00562.x
Uddin S, Hussain AR, Siraj AK et al (2006) Role of phosphatidylinositol 3′-kinase/AKT pathway in diffuse large B-cell lymphoma survival. Blood 108:4178–4186. doi:10.1182/blood-2006-04-016907
Yoshida J, Ishii G, Yokose T et al (2010) Possible delayed cut-end recurrence after limited resection for ground-glass opacity adenocarcinoma, intraoperatively diagnosed as Noguchi type B, in three patients. J Thorac Oncol 5:546–550. doi:10.1097/JTO.0b013e3181d0a480
Yoshizawa A, Motoi N, Riely GJ et al (2011) Impact of proposed IASLC/ATS/ERS classification of lung adenocarcinoma: prognostic subgroups and implications for further revision of staging based on analysis of 514 stage I cases. Mod Pathol 24:653–664. doi:10.1038/modpathol.2010.232
Zhang CJ, Shen ZJ, Pan CW et al (2012a) Engagement of integrinβ1 induces resistance of bladder cancer cells to mitomycin-C. Urology 79:638–643. doi:10.1016/j.urology.2011.12.012
Zhang X, Chen G, Wu T et al (2012b) Expression and clinical significance of Ezrin in non-small-cell lung cancer. Clin Lung Cancer. doi:10.1016/j.cllc.2011.04.002
Acknowledgments
This work was supported by the National Cancer Center Research and Development Fund (23-A-16). We are grateful to Ms. Yuka Nakamura for her technical support.
Author contribution
KH, JY and GI conceived the study. KH, GI, HU and HH carried out the experiments. KH and GI analyzed the data. KH, JY and GI were involved in writing the paper. All authors had final approval of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interests to disclose.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
432_2016_2154_MOESM2_ESM.docx
Supplementary material 2 Figure S2: IHC staining for GLUT1 of LPA and PPA. Upper panel: LPA cancer cells show negative staining for GLUT1, while PPA cancer cells show positive staining for GLUT1 in both periphery and center of the tumor; lower panel: summary of GLUT1 staining scores for LPA and PPA (DOCX 857 kb)
432_2016_2154_MOESM3_ESM.docx
Supplementary material 3 Figure S3: Correlation between ezrin and pAkt expressions in each cases. Ezrin and pAkt expressions in each case tend to have positive correlation by using Spearman’s rank correlation coefficients (DOCX 94 kb)
Rights and permissions
About this article
Cite this article
Hata, K., Yoshida, J., Udagawa, H. et al. The difference in Ezrin–pAkt signaling axis between lepidic and papillary predominant invasive adenocarcinomas of the lung. J Cancer Res Clin Oncol 142, 1421–1430 (2016). https://doi.org/10.1007/s00432-016-2154-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-016-2154-z