Abstract
Purpose
To predict short-term anti-vascular endothelial growth factor (anti-VEGF) treatment responder/non-responder for neovascular age-related macular degeneration (nAMD) patients based on optical coherence tomography (OCT) images.
Methods
A total of 4944 OCT scans from 206 patients with nAMD were involved to develop and evaluate a responder/non-responder prediction method for the short-term effect of anti-VEGF therapy. A deep learning architecture named sensitive structure guided network (SSG-Net) was proposed to make the prediction leveraging a sensitive structure guidance module trained from pre- and post-treatment images. To verify its clinical efficiency, other 2 deep learning methods and 4 experienced ophthalmologists were involved to evaluate the performance of the developed model.
Results
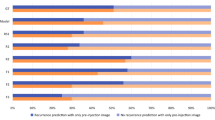
For the testing dataset, SSG-Net could predict the response by an accuracy of 84.6% and an area under the receiver curve (AUC) of 0.83, with a sensitivity of 0.692 and specificity of 1. In contrast, the 2 compared deep learning methods achieved an accuracy of 65.4% with a sensitivity of 0.461 and specificity of 0.846, and an accuracy of 73.1% with a sensitivity of 0.692 and specificity of 0.846, respectively. The predicted accuracy for 4 experienced ophthalmologists was 53.8 to 76.9%, with sensitivity of 0.538 to 0.923 and specificity of 0.385 to 0.846, respectively.
Conclusion
Our proposed SSG-Net shows effective prediction on the short-term efficacy of anti-VEGF treatment for nAMD patients. This technique could potentially help clinicians explain the necessity of anti-VEGF treatment to the potential responder and avoid unnecessary treatment for the non-responder.





Similar content being viewed by others
Data availability
All relevant data was shown in the manuscript.
Code availability
Acquired by contacting the corresponding author.
References
Quartilho A, Simkiss P, Zekite A, Xing W, Wormald R, Bunce C (2016) Leading causes of certifiable visual loss in England and Wales during the year ending 31 March 2013. Eye 30(4):602–607
Ferrara N, Adamis AP (2016) Ten years of anti-vascular endothelial growth factor therapy. Nat Rev Drug Discov 15(6):385–403
Hodgson N, Wu F, Zhu J, Wang W, Ferreyra H, Zhang K, Wang J (2016) Economic and quality of life benefits of anti-VEGF therapy. Mol Pharm 13(9):2877–2880
Sadda SR, Guymer R, Holz FG, Schmitz-Valckenberg S, Curcio CA, Bird AC, Blodi BA, Bottoni F, Chakravarthy U, Chew EY (2018) Consensus definition for atrophy associated with age-related macular degeneration on OCT: classification of atrophy report 3. Ophthalmology 125(4):537–548
Keane PA, Sadda SR (2014) Retinal imaging in the twenty-first century: state of the art and future directions. Ophthalmology 121(12):2489–2500
Rohm M, Tresp V, Müller M, Kern C, Manakov I, Weiss M, Sim DA, Priglinger S, Keane PA, Kortuem K (2018) Predicting visual acuity by using machine learning in patients treated for neovascular age-related macular degeneration. Ophthalmology 125(7):1028–1036
Bogunović H, Waldstein SM, Schlegl T, Langs G, Sadeghipour A, Liu X, Gerendas BS, Osborne A, Schmidt-Erfurth U (2017) Prediction of anti-VEGF treatment requirements in neovascular AMD using a machine learning approach. Invest Ophthalmol Vis Sci 58(7):3240–3248
Yim J, Chopra R, Spitz T, Winkens J, Obika A, Kelly C, Askham H, Lukic M, Huemer J, Fasler K (2020) Predicting conversion to wet age-related macular degeneration using deep learning. Nat Med 26(6):892–899
Cao J, You K, Jin K, Lou L, Wang Y, Chen M, Pan X, Shao J, Su Z, Wu J (2021) Prediction of response to anti-vascular endothelial growth factor treatment in diabetic macular oedema using an optical coherence tomography-based machine learning method. Acta Ophthalmol 99(1):e19–e27. https://doi.org/10.1111/aos.14514
Rasti R, Allingham MJ, Mettu PS, Kavusi S, Govind K, Cousins SW, Farsiu S (2020) Deep learning-based single-shot prediction of differential effects of anti-VEGF treatment in patients with diabetic macular edema. Biomed Opt Express 11(2):1139–1152
Kawczynski MG, Bengtsson T, Dai J, Hopkins JJ, Gao SS, Willis JR (2020) Development of deep learning models to predict best-corrected visual acuity from optical coherence tomography. Transl Vis Sci Technol 9(2):51–51
Liu Y, Yang J, Zhou Y, Wang W, Zhao J, Yu W, Zhang D, Ding D, Li X, Chen Y (2020) Prediction of OCT images of short-term response to anti-VEGF treatment for neovascular age-related macular degeneration using generative adversarial network. Br J Ophthalmol 104(12):1735–1740
Ou WC, Brown DM, Payne JF, Wykoff CC (2017) Relationship between visual acuity and retinal thickness during anti–vascular endothelial growth factor therapy for retinal diseases. Am J Ophthalmol 180:8–17
Lee J, Moon BG, Cho AR, Yoon YH (2016) Optical coherence tomography angiography of DME and its association with anti-VEGF treatment response. Ophthalmology 123(11):2368–2375
Vujosevic S, Torresin T, Bini S, Convento E, Pilotto E, Parrozzani R, Midena E (2017) Imaging retinal inflammatory biomarkers after intravitreal steroid and anti-VEGF treatment in diabetic macular oedema. Acta Ophthalmol 95(5):464–471
Jaffe GJ, Kaiser PK, Thompson D, Gibson A, Saroj N, Vitti R, Berliner AJ, Heier JS (2016) Differential response to anti-VEGF regimens in age-related macular degeneration patients with early persistent retinal fluid. Ophthalmology 123(9):1856–1864
Hwang HS, Chae JB, Kim JY, Kim DY (2017) Association between hyperreflective dots on spectral-domain optical coherence tomography in macular edema and response to treatment. Invest Ophthalmol Vis Sci 58(13):5958–5967
Schreur V, Altay L, van Asten F, Groenewoud JM, Fauser S, Klevering BJ, Hoyng CB, de Jong EK (2018) Hyperreflective foci on optical coherence tomography associate with treatment outcome for anti-VEGF in patients with diabetic macular edema. PLoS One 13(10):e0206482
Liu TA, Farsiu S, Ting DS (2020) Generative adversarial networks to predict treatment response for neovascular age-related macular degeneration: interesting, but is it useful? Br J Ophthalmol 104(12):1629–1630
Ronneberger O, Fischer P, Brox T (2015) U-net: convolutional networks for biomedical image segmentation. https://arxiv.org/pdf/1505.04597v1.pdf. Accessed 8 Dec 2020
Hu J, Shen L, Albanie S, Sun G, Wu E (2018) Squeeze-and-excitation networks. Proceedings of the IEEE conference on computer vision and pattern recognition: 7132–7141
Krizhevsky A, Sutskever I, Hinton GE (2017) Imagenet classification with deep convolutional neural networks. Commun ACM 60(6):84–90
Fawcett T (2006) An introduction to ROC analysis. Pattern Recogn Lett 27(8):861–874
Zhou B, Khosla A, Lapedriza A, Oliva A, Torralba A (2016) Learning deep features for discriminative localization. IEEE Comput Soc. https://doi.org/10.1109/CVPR.2016.319
Kermany DS, Goldbaum M, Cai W, Valentim CCS, Liang H, Baxter SL, McKeown A, Yang G, Wu X, Yan F, Dong J, Prasadha MK, Pei J, Ting MYL, Zhu J, Li C, Hewett S, Dong J, Ziyar I, Shi A, Zhang R, Zheng L, Hou R, Shi W, Fu X, Duan Y, Huu VAN, Wen C, Zhang ED, Zhang CL, Li O, Wang X, Singer MA, Sun X, Xu J, Tafreshi A, Lewis MA, Xia H, Zhang K (2018) Identifying medical diagnoses and treatable diseases by image-based deep learning. Cell 172(5):1122–1131.e9
Jager RD, Mieler WF, Miller JW (2008) Age-related macular degeneration. N Engl J Med 358(24):2606–2617
Rim TH, Lee AY, Ting DS, Teo K, Betzler BK, Teo ZL, Yoo TK, Lee G, Kim Y, Lin AC, Kim SE, Tham YC, Kim SS, Cheng CY, Wong TY, Cheung CMG (2020) Detection of features associated with neovascular age-related macular degeneration in ethnically distinct data sets by an optical coherence tomography: trained deep learning algorithm. Br J Ophthalmol. https://doi.org/10.1136/bjophthalmol-2020-316984
Ting DS, Peng L, Varadarajan AV, Keane PA, Burlina PM, Chiang MF, Schmetterer L, Pasquale LR, Bressler NM, Webster DR (2019) Deep learning in ophthalmology: the technical and clinical considerations. Prog Retin Eye Res 72:100759
Arichika S, Hangai M, Yoshimura N (2010) Correlation between thickening of the inner and outer retina and visual acuity in patients with epiretinal membrane. Retina 30(3):503–508
Acknowledgements
We would like to thank the editor and anonymous reviewers for the valuable comments and suggestions.
Funding
No funding existed for this study.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics approval
The study was approved by the Ethics Committee of Peking Union Medical College Hospital, Chinese Academy of Medical Sciences (No. S-K1611). The whole process adhered to the tenets of the Declaration of Helsinki.
Consent to participate
A written informed consent was obtained from each participant before treatment.
Consent for publication
A written informed consent for publication was obtained from each participant before treatment.
Conflict of interest
All authors have contributed significantly and are in agreement with the content of the manuscript. All authors have no relevant financial relationships to disclose.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Figure S1.
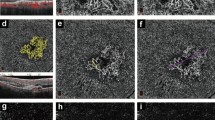
For the encoder, we utilized SE-ResNet blocks to achieve a channel-wise attention and consistent training. The encoder is then connected by global average pooling and fully connected layers to output classification results. The decoder is a top-down architecture with lateral connections and outputs spatial transform field. Then the image of pre-treatment is warped with spatial transform field using a spatial transformer function and transformed to an artificial OCT image of post-treatment. Encoders could get the back propagation by skip connection from decoders, absorbing more guiding information from the back propagation and concentrate more on the structural changes from semantic feature maps at all scales when outputting results. (PNG 1075 kb)
Rights and permissions
About this article
Cite this article
Zhao, X., Zhang, X., Lv, B. et al. Optical coherence tomography-based short-term effect prediction of anti-vascular endothelial growth factor treatment in neovascular age-related macular degeneration using sensitive structure guided network. Graefes Arch Clin Exp Ophthalmol 259, 3261–3269 (2021). https://doi.org/10.1007/s00417-021-05247-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-021-05247-4