Abstract
Purpose
High myopia can lead to blindness. Genipin is a collagen cross-linking agent that may be used to treat myopia. However, the mechanism of action of genipin for the treatment of myopia is unclear. This study investigated the effect of genipin on the scleral expression of the miR-29 cluster, matrix metalloproteinase 2 (MMP2), and collagen alpha1 chain of type I (COL1A1) in a guinea pig model of myopia.
Methods
The model of myopia was established by treating guinea pigs with a − 8D lens on both eyes for 21 days, and eyes with a refractive error of − 6D or greater were included. Quantitative reverse transcription–polymerase chain reaction (RT-PCR) and western blot were used to examine the mRNA and protein expression, respectively. A dual-luciferase assay was used to determine the direct targeting of the miR-29 cluster on the 3′-untranslated region (UTR) of the COL1A1 gene.
Results
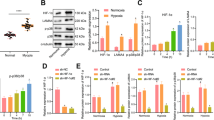
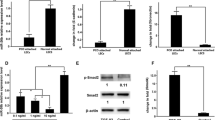
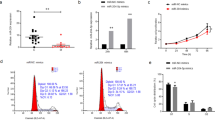
The scleral expression of miR-29a, miR-29b, and miR-29c as well as MMP2 was significantly increased, and the scleral expression of COL1A1 was significantly decreased in the myopia group. Genipin treatment reversed these effects in myopic eyes. The dual-luciferase assay showed that the luciferase activities were significantly decreased in human embryonic kidney (HEK) cells transfected with miR-29a and miR-29b, but not miR-29c, compared with those transfected with control miRNAs.
Conclusions
Genipin inhibits the scleral expression of the miR-29 cluster and MMP2 and promotes COL1A1 expression in a guinea pig model of myopia. Thus, genipin may promote COL1A1 expression by reducing the expression of the miR-29 cluster.



Similar content being viewed by others
Availability of data and materials
The datasets generated and analyzed during the present study are available from the corresponding author on reasonable request.
Abbreviations
- COL1A1:
-
Collagen alpha1 chain of type I
- RT-PCR:
-
Reverse transcription–polymerase chain reaction
- UTR:
-
Untranslated region
- HEK:
-
Human embryonic kidney
- MMP2:
-
Matrix metalloproteinase 2
- miRNAs:
-
MicroRNAs
References
McBrien NA, Gentle A (2003) Role of the sclera in the development and pathological complications of myopia. Prog Retin Eye Res 22:307–338
Li J, Zhang Q (2017) Insight into the molecular genetics of myopia. Mol Vis 23:1048–1080
Leo SW, Young TL (2011) An evidence-based update on myopia and interventions to retard its progression. J AAPOS 15:181–189. https://doi.org/10.1016/j.jaapos.2010.09.020
Norton TT, Rada JA (1995) Reduced extracellular matrix in mammalian sclera with induced myopia. Vis Res 35:1271–1281
Harper AR, Summers JA (2015) The dynamic sclera: extracellular matrix remodeling in normal ocular growth and myopia development. Exp Eye Res 133:100–111. https://doi.org/10.1016/j.exer.2014.07.015
Liu TX, Wang Z (2017) Biomechanics of sclera crosslinked using genipin in rabbit. Int J Ophthalmol 10:355–360. https://doi.org/10.18240/ijo.2017.03.05
Campbell IC, Hannon BG, Read AT, Sherwood JM, Schwaner SA, Ethier CR (2017) Quantification of the efficacy of collagen cross-linking agents to induce stiffening of rat sclera. J R Soc Interface 14. https://doi.org/10.1098/rsif.2017.0014
Liu TX, Wang Z (2013) Collagen crosslinking of porcine sclera using genipin. Acta Ophthalmol 91:e253–e257. https://doi.org/10.1111/aos.12172
Zhu SQ, Zheng LY, Pan AP, Yu AY, Wang QM, Xue AQ (2016) The efficacy and safety of posterior scleral reinforcement using genipin cross-linked sclera for macular detachment and retinoschisis in highly myopic eyes. Br J Ophthalmol 100:1470–1475. https://doi.org/10.1136/bjophthalmol-2015-308087
Wang M, Corpuz CC (2015) Effects of scleral cross-linking using genipin on the process of form-deprivation myopia in the Guinea pig: a randomized controlled experimental study. BMC Ophthalmol 15:89. https://doi.org/10.1186/s12886-015-0086-z
Wang Q, Zhao G, Xing S, Zhang L, Yang X (2011) Role of bone morphogenetic proteins in form-deprivation myopia sclera. Mol Vis 17:647–657
Frost MR, Norton TT (2007) Differential protein expression in tree shrew sclera during development of lens-induced myopia and recovery. Mol Vis 13:1580–1588
Gong B, Qu C, Huang XF, Ye ZM, Zhang DD, Shi Y, Chen R, Liu YP, Shuai P (2016) Genetic association of COL1A1 polymorphisms with high myopia in Asian population: a meta-analysis. Int J Ophthalmol 9:1187–1193. https://doi.org/10.18240/ijo.2016.08.16
Zhang X, Zhou X, Qu X (2015) Association between COL1A1 polymorphisms and high myopia: a meta-analysis. Int J Clin Exp Med 8:5862–5868
Yu L, Li ZK, Gao JR, Liu JR, Xu CT (2011) Epidemiology, genetics and treatments for myopia. Int J Ophthalmol 4:658–669. https://doi.org/10.3980/j.issn.2222-3959.2011.06.17
Rada JA, Brenza HL (1995) Increased latent gelatinase activity in the sclera of visually deprived chicks. Invest Ophthalmol Vis Sci 36:1555–1565
Rada JA, Perry CA, Slover ML, Achen VR (1999) Gelatinase a and TIMP-2 expression in the fibrous sclera of myopic and recovering chick eyes. Invest Ophthalmol Vis Sci 40:3091–3099
Guggenheim JA, McBrien NA (1996) Form-deprivation myopia induces activation of scleral matrix metalloproteinase-2 in tree shrew. Invest Ophthalmol Vis Sci 37:1380–1395
Liu Z, Qiu F, Li J, Zhu Z, Yang W, Zhou X, An J, Huang F, Wang Q, Reinach PS, Li W, Chen W, Liu Z (2015) Adenomatous polyposis coli mutation leads to myopia development in mice. PLoS One 10:e0141144. https://doi.org/10.1371/journal.pone.0141144
Zhou X, Ji F, An J, Zhao F, Shi F, Huang F, Li Y, Jiao S, Yan D, Chen X, Chen J, Qu J (2012) Experimental murine myopia induces collagen type Ialpha1 (COL1A1) DNA methylation and altered COL1A1 messenger RNA expression in sclera. Mol Vis 18:1312–1324
Jiang B, Huo Y, Gu Y, Wang J (2017) The role of microRNAs in myopia. Graefes Arch Clin Exp Ophthalmol 255:7–13. https://doi.org/10.1007/s00417-016-3532-6
Luna C, Li G, Qiu J, Epstein DL, Gonzalez P (2009) Role of miR-29b on the regulation of the extracellular matrix in human trabecular meshwork cells under chronic oxidative stress. Mol Vis 15:2488–2497
Xie M, Li Y, Wu J, Wu J (2016) Genetic variants in MiR-29a associated with high myopia. Ophthalmic Genet 37:456–458. https://doi.org/10.3109/13816810.2015.1101776
Wang M, Yang ZK, Li RQ, Fan H, Liu Y (2019) Comparison of the biomechanics and fixation index of crosslinking between lysyl oxidase and genipin on guinea pig sclera, an animal model of defocus-induced high myopia. Proc Est Acad Sci 2:214–221
McBrien NA, Cornell LM, Gentle A (2001) Structural and ultrastructural changes to the sclera in a mammalian model of high myopia. Invest Ophthalmol Vis Sci 42:2179–2187
Vannini I, Fanini F, Fabbri M (2018) Emerging roles of microRNAs in cancer. Curr Opin Genet Dev. https://doi.org/10.1016/j.gde.2018.01.001
Maurer B, Stanczyk J, Jungel A, Akhmetshina A, Trenkmann M, Brock M, Kowal-Bielecka O, Gay RE, Michel BA, Distler JH, Gay S, Distler O (2010) MicroRNA-29, a key regulator of collagen expression in systemic sclerosis. Arthritis Rheum 62:1733–1743. https://doi.org/10.1002/art.27443
Kim JW, Lindsey JD, Wang N, Weinreb RN (2001) Increased human scleral permeability with prostaglandin exposure. Invest Ophthalmol Vis Sci 42:1514–1521
Wojciechowski R, Bailey-Wilson JE, Stambolian D (2010) Association of matrix metalloproteinase gene polymorphisms with refractive error in Amish and Ashkenazi families. Invest Ophthalmol Vis Sci 51:4989–4995. https://doi.org/10.1167/iovs.10-5474
Nakanishi H, Hayashi H, Yamada R, Yamashiro K, Nakata I, Shimada N, Ohno-Matsui K, Mochizuki M, Ozaki M, Yoshitake S, Kuriyama S, Saito M, Iida T, Matsuo K, Matsuda F, Yoshimura N (2010) Single-nucleotide polymorphisms in the promoter region of matrix metalloproteinase-1, −2, and −3 in Japanese with high myopia. Invest Ophthalmol Vis Sci 51:4432–4436. https://doi.org/10.1167/iovs.09-4871
Leung KH, Yiu WC, Yap MK, Ng PW, Fung WY, Sham PC, Yip SP (2011) Systematic investigation of the relationship between high myopia and polymorphisms of the MMP2, TIMP2, and TIMP3 genes by a DNA pooling approach. Invest Ophthalmol Vis Sci 52:3893–3900. https://doi.org/10.1167/iovs.11-7286
Gong B, Liu X, Zhang D, Wang P, Huang L, Lin Y, Lu F, Ma S, Cheng J, Chen R, Li X, Lin H, Zeng G, Zhu X, Hu J, Yang Z, Shi Y (2013) Evaluation of MMP2 as a candidate gene for high myopia. Mol Vis 19:121–127
Siegwart JT Jr, Norton TT (2005) Selective regulation of MMP and TIMP mRNA levels in tree shrew sclera during minus lens compensation and recovery. Invest Ophthalmol Vis Sci 46:3484–3492. https://doi.org/10.1167/iovs.05-0194
Funding
Supported by National Natural Science Foundation of China (No.81770958).
Author information
Authors and Affiliations
Contributions
Min Wang and Zhi-Kuan Yang designed most of the investigation, performed data analysis, and wrote the manuscript; Hong Liu provided pathological and experimental assistance; Rui-qin Li contributed to interpretation of the data and analyses. Yu Liu and Wen-Jun Zhong provided help with retinoscopy.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Research involving animals
All experimental protocols were approved by the Ethics Committee of the Shanghai Experimental Animal Society (Approval number: SZY201704007).
Informed consent
Not applicable.
Patient consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, M., Yang, ZK., Liu, H. et al. Genipin inhibits the scleral expression of miR-29 and MMP2 and promotes COL1A1 expression in myopic eyes of guinea pigs. Graefes Arch Clin Exp Ophthalmol 258, 1031–1038 (2020). https://doi.org/10.1007/s00417-020-04634-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-020-04634-7