Abstract
Background
This study was conducted to evaluate the efficacy of dietary n-6 and n-3 polyunsaturated fatty acids (PUFAs) in dry eye in a rat model.
Methods
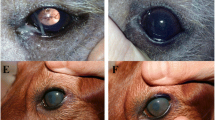
Female Lewis rats were fed with diets containing (1) γ-linolenic acid (GLA), (2) eicosapentaenoic acid (EPA) + docosahexaenoic acid (DHA), or (3) GLA + EPA + DHA, for 2 months before the induction of dry eye using a continuous delivery of scopolamine and during scopolamine treatment. Two, 10 and 28 days after dry-eye induction, clinical signs of corneal dryness were evaluated in vivo using fluorescein staining. MHC II expression and mucin rMuc5AC production in the conjunctival epithelium were evaluated by immunostaining. Lipids and prostaglandins (PGs) E1 and E2 were analysed from the exorbital lacrimal gland (LG).
Results
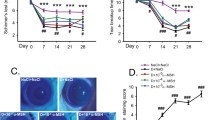
Dietary PUFAs minimised the occurrence of corneal keratitis 28 days after induction of dry eye. The decrease in mucin production observed on the conjunctival epithelium was partially prevented by EPA + DHA supplementation after 2 days of scopolamine treatment, as well as by GLA and GLA + EPA + DHA diets after 10 days of treatment. The overexpression of MHC II in the conjunctival epithelium caused by dry eye induction was significantly reduced only with the GLA + EPA + DHA diet after 28 days of treatment. Dietary PUFAs were incorporated into phospholipids of the exorbital LG. Induction of dry eye was associated with a significant increase in PGE1 and PGE2 levels in the exorbital LG, which was inhibited by dietary EPA + DHA at 10 days (for PGE2) and 28 days (for PGE1).
Conclusions
Dietary GLA, EPA and DHA significantly interfered with lipid homeostasis in the exorbital LG and partially prevented the course of dry eye. In particular, our results demonstrate the efficacy of the combination of n-6 and n-3 PUFAs.





Similar content being viewed by others
References
Moss SE, Klein R, Klein BE (2000) Prevalence of and risk factors for dry eye syndrome. Arch Ophthalmol 118(9):1264–1268
Creuzot-Garcher C et al (2004) Treating severe dry eye syndromes with autologous serum. J Fr Ophtalmol 27(4):346–351, doi:10.1016/S0181-5512(04)96139-6
Kojima T et al (2005) The effect of autologous serum eyedrops in the treatment of severe dry eye disease: a prospective randomized case-control study. Am J Ophthalmol 139(2):242–246, doi:10.1016/j.ajo.2004.08.040
Noble BA et al (2004) Comparison of autologous serum eye drops with conventional therapy in a randomised controlled crossover trial for ocular surface disease. Br J Ophthalmol 88(5):647–652, doi:10.1136/bjo.2003.026211
Tsubota K et al (1999) Treatment of dry eye by autologous serum application in Sjogren's syndrome. Br J Ophthalmol 83(4):390–395, doi:10.1136/bjo.83.4.390
Calonge M (2001) The treatment of dry eye. Surv Ophthalmol 45(Suppl 2):S227–S239, doi:10.1016/S0039-6257(00)00205-8
Pflugfelder SC (2004) Antiinflammatory therapy for dry eye. Am J Ophthalmol 137(2):337–342, doi:10.1016/j.ajo.2003.10.036
Management and Therapy Subcommittee of the International Dry Eye WorkShop (2007) Management and therapy of dry eye disease: Report of the Management and Therapy Subcommittee of the International Dry Eye WorkShop 2007. Ocul Surf 5(2):163–178
Brignole F et al (2001) Flow cytometric analysis of inflammatory markers in KCS: 6-month treatment with topical cyclosporin A. Invest Ophthalmol Vis Sci 42(1):90–95
Kunert KS et al (2000) Analysis of topical cyclosporine treatment of patients with dry eye syndrome: effect on conjunctival lymphocytes. Arch Ophthalmol 118(11):1489–1496
Kunert KS, Tisdale AS, Gipson IK (2002) Goblet cell numbers and epithelial proliferation in the conjunctiva of patients with dry eye syndrome treated with cyclosporine. Arch Ophthalmol 120(3):330–337
Sall K et al (2000) Two multicenter, randomized studies of the efficacy and safety of cyclosporine ophthalmic emulsion in moderate to severe dry eye disease. CsA Phase 3 Study Group. Ophthalmology 107(4):631–639, doi:10.1016/S0161-6420(99)00176-1
Stevenson D, Tauber J, Reis BL (2000) Efficacy and safety of cyclosporin A ophthalmic emulsion in the treatment of moderate-to-severe dry eye disease: a dose-ranging, randomized trial. The Cyclosporin A Phase 2 Study Group. Ophthalmology 107(5):967–974, doi:10.1016/S0161-6420(00)00035-X
James MJ, Gibson RA, Cleland LG (2000) Dietary polyunsaturated fatty acids and inflammatory mediator production. Am J Clin Nutr 71(1 Suppl):343S–348S
Calder PC (2002) Dietary modification of inflammation with lipids. Proc Nutr Soc 61(3):345–358, doi:10.1079/PNS2002166
Tocher DR et al (1997) Effects of dietary gamma-linolenic acid-rich borage oil combined with marine fish oils on tissue phospholipid fatty acid composition and production of prostaglandins E and F of the 1-, 2- and 3-series in a marine fish deficient in delta5 fatty acyl desaturase. Prostaglandins Leukot Essent Fatty Acids 57(2):125–134, doi:10.1016/S0952-3278(97)90002-8
Kast RE (2001) Borage oil reduction of rheumatoid arthritis activity may be mediated by increased cAMP that suppresses tumor necrosis factor-alpha. Int Immunopharmacol 1(12):2197–2199, doi:10.1016/S1567-5769(01)00146-1
Leventhal LJ, Boyce EG, Zurier RB (1993) Treatment of rheumatoid arthritis with gammalinolenic acid. Ann Intern Med 119(9):867–873
Espersen GT et al (1992) Decreased interleukin-1 beta levels in plasma from rheumatoid arthritis patients after dietary supplementation with n-3 polyunsaturated fatty acids. Clin Rheumatol 11(3):393–395, doi:10.1007/BF02207200
Aragona P et al (2005) Systemic omega-6 essential fatty acid treatment and pge1 tear content in Sjogren's syndrome patients. Invest Ophthalmol Vis Sci 46(12):4474–4479, doi:10.1167/iovs.04-1394
Barabino S et al (2003) Systemic linoleic and gamma-linolenic acid therapy in dry eye syndrome with an inflammatory component. Cornea 22(2):97–101, doi:10.1097/00003226-200303000-00002
Macri A et al (2003) Effect of linoleic acid and gamma-linolenic acid on tear production, tear clearance and on the ocular surface after photorefractive keratectomy. Graefes Arch Clin Exp Ophthalmol 241(7):561–566, doi:10.1007/s00417-003-0685-x
Cermak JM et al (2003) Nutrient intake in women with primary and secondary Sjogren's syndrome. Eur J Clin Nutr 57(2):328–334, doi:10.1038/sj.ejcn.1601543
Miljanovic B et al (2005) Relation between dietary n-3 and n-6 fatty acids and clinically diagnosed dry eye syndrome in women. Am J Clin Nutr 82(4):887–893
Oxholm P et al (1998) Essential fatty acid status in cell membranes and plasma of patients with primary Sjogren's syndrome. Correlations to clinical and immunologic variables using a new model for classification and assessment of disease manifestations. Prostaglandins Leukot Essent Fatty Acids 59(4):239–245, doi:10.1016/S0952-3278(98)90136-3
Creuzot C et al (2006) Improvement of dry eye symptoms with polyunsaturated fatty acids. J Fr Ophtalmol 29(8):868–873, doi:10.1016/S0181-5512(06)70106-1
Viau S et al (2008) Time course of ocular surface and lacrimal gland changes in a new scopolamine-induced dry eye model. Graefes Arch Clin Exp Ophthalmol 246(6):857–867, doi:10.1007/s00417-008-0784-9
Inatomi T et al (1996) Expression of secretory mucin genes by human conjunctival epithelia. Invest Ophthalmol Vis Sci 37(8):1684–1692
Inatomi T et al (1997) Cloning of rat Muc5AC mucin gene: comparison of its structure and tissue distribution to that of human and mouse homologues. Biochem Biophys Res Commun 236(3):789–797, doi:10.1006/bbrc.1997.7051
McKenzie RW, Jumblatt JE, Jumblatt MM (2000) Quantification of MUC2 and MUC5AC transcripts in human conjunctiva. Invest Ophthalmol Vis Sci 41(3):703–708
Huang AJ, Tseng SC, Kenyon KR (1988) Morphogenesis of rat conjunctival goblet cells. Invest Ophthalmol Vis Sci 29(6):969–975
Folch J, Lees M, Sloane Stanley GH (1957) A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem 226(1):497–509
Juaneda P, Rocquelin G (1985) Rapid and convenient separation of phospholipids and non phosphorus lipids from rat heart using silica cartridges. Lipids 20(1):40–41, doi:10.1007/BF02534360
Morrison WR, Smith LM (1964) Preparation of fatty acid methyl esters and dimethylacetals from lipids with boron fluoride-methanol. J Lipid Res 5:600–608
Johnson MM et al (1997) Dietary supplementation with gamma-linolenic acid alters fatty acid content and eicosanoid production in healthy humans. J Nutr 127(8):1435–1444
Baudouin C et al (2005) CCR 4 and CCR 5 expression in conjunctival specimens as differential markers of T(H) 1/ T(H) 2 in ocular surface disorders. J Allergy Clin Immunol 116(3):614–619, doi:10.1016/j.jaci.2005.05.033
Pflugfelder SC et al (1999) Altered cytokine balance in the tear fluid and conjunctiva of patients with Sjogren's syndrome keratoconjunctivitis sicca. Curr Eye Res 19(3):201–211, doi:10.1076/ceyr.19.3.201.5309
Solomon A et al (2001) Pro- and anti-inflammatory forms of interleukin-1 in the tear fluid and conjunctiva of patients with dry-eye disease. Invest Ophthalmol Vis Sci 42(10):2283–2292
Furse RK, Rossetti RG, Zurier RB (2001) Gammalinolenic acid, an unsaturated fatty acid with anti-inflammatory properties, blocks amplification of IL-1 beta production by human monocytes. J Immunol 167(1):490–496
Hughes DA et al (1996) Fish oil supplementation inhibits the expression of major histocompatibility complex class II molecules and adhesion molecules on human monocytes. Am J Clin Nutr 63(2):267–272
Miles EA et al (2001) Influence of age and dietary fish oil on plasma soluble adhesion molecule concentrations. Clin Sci (Lond) 100(1):91–100, doi:10.1042/CS20000198
Fan YY, Ramos KS, Chapkin RS (1997) Dietary gamma-linolenic acid enhances mouse macrophage-derived prostaglandin E1 which inhibits vascular smooth muscle cell proliferation. J Nutr 127(9):1765–1771
Rashid S et al (2008) Topical omega−3 and omega−6 fatty acids for treatment of dry eye. Arch Ophthalmol 126(2):219–225, doi:10.1001/archophthalmol.2007.61
Guesnet P et al (2005) [Les rôles physiologiques majeurs exercés par les acides gras polyinsaturés (AGPI)]. Oléagineux, Corps gras. Lipides 12(5–6):333–343
Schnebelen C et al (2009) Nutrition for the eye: different susceptibility of the retina and the lacrimal gland to dietary omega-6 and omega-3 polyunsaturated fatty acid incorporation. Ophthalmic Res (in press).
Barham JB et al (2000) Addition of eicosapentaenoic acid to gamma-linolenic acid-supplemented diets prevents serum arachidonic acid accumulation in humans. J Nutr 130(8):1925–1931
Herman CA, Hamberg M, Granstrom E (1987) Quantitative determination of prostaglandins E1, E2 and E3 in frog tissue. J Chromatogr A 394(2):353–362, doi:10.1016/S0021-9673(01)94187-1
Rubin D, Laposata M (1991) Regulation of agonist-induced prostaglandin E1 versus prostaglandin E2 production. A mass analysis. J Biol Chem 266(35):23618–23623
Levin G et al (2002) Differential metabolism of dihomo-gamma-linolenic acid and arachidonic acid by cyclo-oxygenase-1 and cyclo-oxygenase-2: implications for cellular synthesis of prostaglandin E1 and prostaglandin E2. Biochem J 365(Pt 2):489–496, doi:10.1042/BJ20011798
Zhao Y et al (2004) Eicosapentaenoic acid prevents LPS-induced TNF-alpha expression by preventing NF-kappaB activation. J Am Coll Nutr 23(1):71–78
Forman BM, Chen J, Evans RM (1997) Hypolipidemic drugs, polyunsaturated fatty acids, and eicosanoids are ligands for peroxisome proliferator-activated receptors alpha and delta. Proc Natl Acad Sci USA 94(9):4312–4317, doi:10.1073/pnas.94.9.4312
Kliewer SA et al (1997) Fatty acids and eicosanoids regulate gene expression through direct interactions with peroxisome proliferator-activated receptors alpha and gamma. Proc Natl Acad Sci USA 94(9):4318–4323, doi:10.1073/pnas.94.9.4318
Christman JW, Lancaster LH, Blackwell TS (1998) Nuclear factor kappa B: a pivotal role in the systemic inflammatory response syndrome and new target for therapy. Intensive Care Med 24(11):1131–1138, doi:10.1007/s001340050735
Clark RB (2002) The role of PPARs in inflammation and immunity. J Leukoc Biol 71(3):388–400
Acknowledgements
The technical assistance of F. Charpentier (University Claude Bernard, Lyon, France) and C. Fournier (University of Burgundy, Dijon, France), the technical support of C. Schneider (UMR LEG, INRA, Epoisses, France) in the analysis of rMuc5AC immunostaining, J. Bara (U-673, INSERM, St-Antoine Hospital, Paris, France) for providing the anti-rMuc5AC antibody, Lesieur (Asnières, France) for providing some of the dietary oils, UPAE (Unité de Préparation des Aliments Expérimentaux, INRA, Jouy-en-Josas, France) for preparing the diets, and Linda Northrup (English Solutions, Voiron, France) for the English language editing of the manuscript are acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Support: Regional Council of Burgundy (Dijon, France) for S. Viau (PhD fellowship) and C. Joffre (Grant no 29000026) The Nutrition, Chemical Food Safety and Consumer Behaviour Division from INRA (Dijon, France) for S. Viau (BTH01698) Horus Pharma (Saint Laurent du Var, France) for C. Joffre (research contract no 29000144)
Rights and permissions
About this article
Cite this article
Viau, S., Maire, MA., Pasquis, B. et al. Efficacy of a 2-month dietary supplementation with polyunsaturated fatty acids in dry eye induced by scopolamine in a rat model. Graefes Arch Clin Exp Ophthalmol 247, 1039–1050 (2009). https://doi.org/10.1007/s00417-009-1080-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-009-1080-z