Abstract
Objective
To determine frequency and type of cognitive disorders in cross-sectional analysis of a Parkinson’s disease (PD) cohort, and explore its relations to motor symptoms, modifiable vascular risk factors and white matter lesions (WML) volume.
Methods
In a group of 133 PD patients, mild cognitive impairment (PD-MCI) and dementia (PDD) were diagnosed according to Movement Disorders Society Task Force criteria (level 2 for PD-MCI). Detailed motor measurements were applied, including rigidity, axial, bradykinesia, tremor and postural instability gait disorders (PIGD) scores. Vascular risk was estimated by the Framingham General Cardiovascular Disease risk scoring algorithm and WML volume was measured for whole brain and frontal lobe.
Results
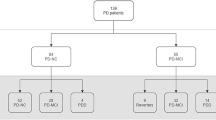
Sixty-one (46.9%) patients fulfilled criteria for PD-MCI, and 23 (17.7%) for PDD. Non-amnestic multiple domain MCI was most frequent (52% of PD-MCI patients). Motor scores were significantly higher in cognitively impaired patients, but only axial score discriminated between MCI and dementia. High vascular risk was related to impaired cognition, bradykinesia, axial, PIGD and freezing of gait (FOG) score, while whole brain WML volume was associated with PDD, FOG and attention deficits. Furthermore, high vascular risk was identified as a potential predictor of both MCI and dementia in PD. Additionally, age and bradykinesia score were independently associated with PD-MCI and age, axial score and whole brain WML volume with PDD.
Conclusion
Cognitive disorders in PD are associated with more severe, predominantly axial motor deficits and increased, but partly modifiable vascular burden, thus opening a possibility for development of preventive strategies in PD.


Similar content being viewed by others
References
Aarsland D, Creese B, Politis M, Chaudhuri KR, Ffytche DH, Weintraub D, Ballard C (2017) Cognitive decline in Parkinson disease. Nat Rev Neurol 13(4):217–231
Winter Y, von Campenhausen S, Arend M, Longo K, Boetzel K, Eggert K, Oertel WH, Dodel R, Barone P (2011) Health-related quality of life and its determinants in Parkinson’s disease, results of an Italian cohort study. Parkinsonism Relat Disord 17:265–269
Rosenthal E, Brennan L, Xie S, Hurtig H, Milber J, Weintraub D, Karlawish J, Siderowf A (2010) Association between cognition and function in patients with Parkinson disease with and without dementia. Mov Disord 25:1170–1176
Lo RY, Tanner CM, Albers KB, Leimpeter AD, Fross RD, Bernstein AL, McGuire V, Quesenberry CP, Nelson LM, Van Den Eeden SK (2009) Clinical features in early Parkinson disease and survival. Arch Neurol 66:1353–1358
Kehagia AA, Barker RA, Robbins TW (2013) Cognitive impairment in Parkinson’s disease the dual syndrome hypothesis. Neurodegener Dis 11:79–92
Kelly VE, Johnson CO, McGough EL, Shumway-Cook A, Horak FB, Chung KA, Espay AJ, Revilla FJ, Devoto J, Wood-Siverio C, Factor SA, Cholerton B, Edwards KL, Peterson AL, Quinn JF, Montine TJ, Zabetian CP, Leverenz JB (2015) Association of cognitive domains with postural instability/gait disturbance in Parkinson’s disease. Parkinsonism Relat Disord 21:692–697
Veselý B, Rektor I (2016) The contribution of white matter lesions (WML) to Parkinson’s disease cognitive impairment symptoms: a critical review of the literature. Parkinsonism Relat Disord 22:66–70
Veselý B, Antonini A, Rektor I (2016) The contribution of white matter lesions to Parkinson’s disease motor and gait symptoms: a critical review of the literature. J Neural Transm 123(3):241–250
Malek N, Lawton MA, Swallow DM, Grosset KA, Marrinan SL, Bajaj N, Barker RA, Burn DJ, Hardy J, Morris HR, Williams NM, Wood N, Ben-Shlomo Y, Grosset DG, PRoBaND Clinical Consortium (2016) Vascular disease and vascular risk factors in relation to motor features and cognition in early Parkinson’s disease. Mov Disord 31(10):1518–1526
Kotagal V, Albin RL, Müller ML, Koeppe RA, Frey KA, Bohnen NI (2014) Modifiable cardiovascular risk factors and axial motor impairments in Parkinson disease. Neurology 82:1514–1520
Hughes AJ, Daniel SE, Kilford L, Lees AJ (1992) Accuracy of clinical diagnosis of idiopathic Parkinson’s disease a clinico-pathological study of 100 cases. J Neurol Neurosurg Psychiatry 55:181–184
Zijlmans JC, Daniel SE, Hughes AJ, Revesz T, Lees AJ (2004) Clinicopathological investigation of vascular parkinsonism, including clinical criteria for diagnosis. Mov Disord 19:630–640
Hoehn MM, Yahr MD (1967) Parkinsonism onset, progression and mortality. Neurology 15:427–442
Movement Disorder Society Task Force on Rating Scales for Parkinson’s Disease (2003) The Unified Parkinson’s disease rating scale (UPDRS): status and recommendations. Mov Disord 18:738–750
Jankovic J, McDermott M, Carter J, Gauthier S, Goetz C, Golbe L, Huber S, Koller W, Olanow C, Shoulson I et al (1990) Variable expression of Parkinson’s disease: a base-line analysis of the DATATOP cohort. The Parkinson Study Group. Neurology 40:1529–1534
Levy G, Tang MX, Cote LJ, Louis ED, Alfaro B, Mejia H, Stern Y, Marder K (2000) Motor impairment in PD relationship to incident dementia and age. Neurology 55:539–544
Khoo T, Tilley B, Giladi N, Shabtai H, Simon ES, Biran S, Tal J, Korczyn AD (2000) Construction of freezing of gait questionnaire for patients with Parkinsonism. Parkinsonism Relat Disord 6:165–170
Folstein MF, Folstein SE, McHugh PR (1975) ‘Mini-mental state’, a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198
Mioshi E, Dawson K, Mitchell J, Arnold R, Hodges JR (2006) The Addenbrooke’s Cognitive Examination Revised (ACE-R) a brief cognitive test battery for dementia screening. Int J Geriatr Psychiatry 21:1078–1085
Nasreddine ZS, Phillips NA, Bédirian V, Charbonneau S, Whitehead V, Collin I, Cummings JL, Chertkow H (2005) The Montreal Cognitive Assessment (MoCA) a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 53:695–699
Hamilton M (1960) A rating scale for depression. J Neurol Neurosurg Psychiatry 23:56–62
Hamilton M (1959) Diagnosis and rating of anxiety. Br J Med Psychol 32:50–55
Marin RS (1996) Apathy, concept, syndrome, mechanisms and treatment. Neural Clin Neuropsychiatry 1:303–314
Lezak M (1983) Neuropsychological assessment, 2nd edn. Oxford University Press, New York
Robbins TW, James M, Owen AM, Sahakian BJ, McInnes L, Rabbitt P (1994) Cambridge Neuropsychological Test Automated Battery (CANTAB): a factor analytic study of a large sample of normal elderly volunteers. Dementia 5:266–281
Stefanova E, Žiropadja L, Stojković T, Stanković I, Tomić A, Ječmenica-Lukić M, Petrović I, Kostić V (2015) Mild cognitive impairment in early Parkinson’s disease using the Movement Disorder Society Task Force criteria: cross-sectional study in Hoehn and Yahr stage 1. Dement Geriatr Cogn Disord 40:199–209
Litvan I, Goldman JG, Tröster AI, Schmand BA, Weintraub D, Petersen RC, Mollenhauer B, Adler CH, Marder K, Williams-Gray CH, Aarsland D, Kulisevsky J, Rodriguez-Oroz MC, Burn DJ, Barker RA, Emre M (2012) Diagnostic criteria for mild cognitive impairment in Parkinson’s disease. Mov Disord Soc Task Force Guidel Mov Disord 27:349–356
Dubois B, Burn D, Goetz C, Aarsland D, Brown RG, Broe GA, Dickson D, Duyckaerts C, Cummings J, Gauthier S, Korczyn A, Lees A, Levy R, Litvan I, Mizuno Y, McKeith IG, Olanow CW, Poewe W, Sampaio C, Tolosa E, Emre M (2007) Diagnostic procedures for Parkinson’s disease dementia: recommendations from the movement disorder society task force. Mov Disord 22:2314–2324
Biundo R, Weis L, Antonini A (2016) Cognitive decline in Parkinson’s disease the complex picture, npj. Parkinson’s Dis 2:1–7
Braak H, Rüb U, Steur EJ, Del Tredici K, de Vos RA (2005) Cognitive status correlates with neuropathologic stage in Parkinson disease. Neurology 64(8):1404–1411
Yarnall AJ, Rochester L, Burn DJ (2013) Mild cognitive impairment in Parkinson’s disease. Age Aging 42:567–576
Wills AM, Li R, Pérez A, Ren X, Boyd J, Investigators NINDSNET-PD (2017) Predictors of weight loss in early treated Parkinson’s disease from the NET-PD LS-1 cohort. J Neurol 264(8):1746–1753
Lee PC, Ahmed I, Loriot MA, Mulot C, Paul KC, Bronstein JM, Ritz B, Elbaz A (2018) Smoking and Parkinson disease: evidence for gene-by-smoking interactions. Neurology 90(7):e583–e592
Román GC, Boller F (2014) Vascular factors in neurodegenerative diseases a path towards treatment and prevention. Funct Neurol 29:85–86
Sławek J, Wieczorek D, Derejko M, Dubaniewicz M, Brockhuis B, Sitek E, Wilczewska L, Roszmann A, Lass P (2010) Vascular risk factors do not contribute to motor and cognitive impairment in Parkinson’s disease. Parkinsonism Relat Disord 16(1):73–74 (author reply 75–6)
Moore SF, Barker RA (2014) Predictors of Parkinson’s disease dementia towards targeted therapies for a heterogeneous disease. Parkinsonism Relat Disord 20(Suppl 1):S104–S107
Kivipelto M, Solomon A, Ahtiluoto S, Ngandu T, Lehtisalo J, Antikainen R et al (2013) The Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER): study design and progress. Alzheimers Dement 9:657–665
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
T. Stojkovic has received speaker honoraria from Actavis and research grant from the Ministry of Education and Science, Republic of Serbia, as well as Alzheimer’s Association International Research Grant. E. Stefanova has received speaker honoraria from Novartis, Glaxo Smith Kline and Boehringer Ingelheim, research grant from the Ministry of Education and Science, Republic of Serbia, as well as Alzheimer’s Association International Research Grant. I. Soldatovic. Declarations of interest: none. V. Markovic has received research grants from the Ministry of Education and Science, Republic of Serbia. I. Stankovic has received research grants from the Ministry of Education and Science, Republic of Serbia. I Petrovic has received speaker honoraria from Boehringer Ingelheim, GSK, El pharma, Roche, Actavis and research grants from the Ministry of Education and Science, Republic of Serbia. F. Agosta is Section Editor of NeuroImage: Clinical; has received speaker honoraria from Biogen Idec, Novartis, and ExceMED—Excellence in Medical Education; and receives or has received research supports from the Italian Ministry of Health, AriSLA (Fondazione Italiana di Ricerca per la SLA), and the European Research Council. S. Galantucci. Declarations of interest: none. M. Filippi is Editor-in-Chief of the Journal of Neurology; serves on a scientific advisory board for Teva Pharmaceutical Industries; has received compensation for consulting services and/or speaking activities from Biogen Idec, ExceMED, Novartis, and Teva Pharmaceutical Industries; and receives research support from Biogen Idec, Teva Pharmaceutical Industries, Novartis, Italian Ministry of Health, Fondazione Italiana Sclerosi Multipla, Cure PSP, Alzheimer’s Drug Discovery Foundation (ADDF), the Jacques and Gloria Gossweiler Foundation (Switzerland), and ARiSLA (Fondazione Italiana di Ricerca per la SLA). V. Kostic serves on the editorial board of the Journal of Neurology; has received research grants from Ministry of Education and Science, Republic of Serbia, and the Serbian Academy of Science and Arts; and receives research support from Valeant, Stada, Novartis, and Boehringer Ingelheim, and speaker honoraria from Novartis and Boehringer Ingelheim.
Ethical standards
The local Ethical Committee (School of Medicine, University of Belgrade, Serbia) approved the study protocol and all participants signed written informed consent.
Rights and permissions
About this article
Cite this article
Stojkovic, T., Stefanova, E., Soldatovic, I. et al. Exploring the relationship between motor impairment, vascular burden and cognition in Parkinson’s disease. J Neurol 265, 1320–1327 (2018). https://doi.org/10.1007/s00415-018-8838-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-018-8838-3