Abstract
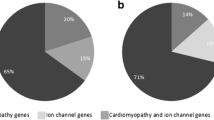
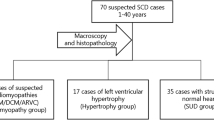
Sudden cardiac death (SCD) is responsible for a large proportion of sudden deaths in young individuals. In forensic medicine, many cases remain unexplained after routine postmortem autopsy and conventional investigations. These cases are called sudden unexplained deaths (SUD). Genetic testing has been suggested useful in forensic medicine, although in general with a significantly lower success rate compared to the clinical setting. The purpose of the study was to estimate the frequency of pathogenic variants in the genes most frequently associated with SCD in SUD cases and compare the frequency to that in patients with inherited cardiac channelopathies. Fifteen forensic SUD cases and 29 patients with channelopathies were investigated. DNA from 34 of the genes most frequently associated with SCD were captured using NimbleGen SeqCap EZ library build and were sequenced with next-generation sequencing (NGS) on an Illumina MiSeq. Likely pathogenic variants were identified in three out of 15 (20 %) forensic SUD cases compared to 12 out of 29 (41 %) patients with channelopathies. The difference was not statistically significant (p = 0.1). Additionally, two larger deletions of entire exons were identified in two of the patients (7 %). The frequency of likely pathogenic variants was >2-fold higher in the clinical setting as compared to SUD cases. However, the demonstration of likely pathogenic variants in three out of 15 forensic SUD cases indicates that NGS investigations will contribute to the clinical investigations. Hence, this has the potential to increase the diagnostic rate significantly in the forensic as well as in the clinical setting.

Similar content being viewed by others
References
Winkel BG, Holst AG, Theilade J, Kristensen IB, Thomsen JL, Ottesen GL, Bundgaard H, Svendsen JH, Haunsø S, Tfelt-Hansen J (2011) Nationwide study of sudden cardiac death in persons aged 1–35 years. Eur Heart J 32(8):983–990. doi:10.1093/eurheartj/ehq428
Wisten A, Forsberg H, Krantz P, Messner T (2002) Sudden cardiac death in 15–35-year olds in Sweden during 1992–99. J Intern Med 252(6):529–536
Puranik R, Chow CK, Duflou JA, Kilborn MJ, McGuire MA (2005) Sudden death in the young. Heart Rhythm 2(12):1277–1282. doi:10.1016/j.hrthm.2005.09.008
Tester DJ, Medeiros-Domingo A, Will ML, Haglund CM, Ackerman MJ (2012) Cardiac channel molecular autopsy: insights from 173 consecutive cases of autopsy-negative sudden unexplained death referred for postmortem genetic testing. Mayo Clin Proc 87(6):524–539. doi:10.1016/j.mayocp.2012.02.017
Tester DJ, Ackerman MJ (2009) Cardiomyopathic and channelopathic causes of sudden unexplained death in infants and children. Annu Rev Med 60:69–84. doi:10.1146/annurev.med.60.052907.103838
Rodríguez-Calvo MS, Ma B, Allegue C, Concheiro L, Carracedo A (2008) Molecular genetics of sudden cardiac death. Forensic Sci Int 182(1–3):1–12. doi:10.1016/j.forsciint.2008.09.013
Hedley PL, Jørgensen P, Schlamowitz S, Moolman‐Smook J, Kanters JK, Corfield VA, Christiansen M (2009) The genetic basis of Brugada syndrome: a mutation update. Hum Mutat 30(9):1256–1266. doi:10.1002/humu.21066
Hedley PL, Jorgensen P, Schlamowitz S, Wangari R, Moolman-Smook J, Brink PA, Kanters JK, Corfield VA, Christiansen M (2009) The genetic basis of long QT and short QT syndromes: a mutation update. Hum Mutat 30(11):1486–1511. doi:10.1002/humu.21106
Online Mendelian Inheritance in Man, OMIM. McKusick-Nathans Institute of Genetic Medicine, John Hopkins University (Baltimore, MD)
MRC-Holland. http://mlpa.com/WebForms/WebFormMain.aspx?Tag=_fNPBLedDVp38p-CxU2h0mQ
Kent WJSC, Furey TS, Roskin KM, Pringle TH, Zahler AM, Haussler D (2002) The human genome browser at USCS. Genome Res 12(6):996–1006
Li HDR (2009) Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25:1754–1760
Van der Auwera GA, Carneiro MO, Hartl C, Poplin R, del Angel G, Levy-Moonshine A, Jordan T, Shakir K, Roazen D, Thibault J, Banks E, Garimella KV, Altshuler D, Gabriel S, DePristo MA (2002) From FastQ data to high-confidence variant calls: the Genome Analysis Toolkit Best Practices Pipeline. In: Current Protocols in Bioinformatics. Wiley
Exome Variant Server. http://evs.gs.washington.edu/EVS/.
National Center for Biotechnology Information. http://www.ncbi.nlm.nih.gov/.
Stenson PDBE, Mort M, Phillips AD, Shiel JA, Thomas NS, Abeysinghe S, Krawczak M, Copper DN (2003) The Human Gene Mutation Database (HGMD). Hum Mutat 21:577–581
Spaendonck-Zwarts KY, Rijsingen IAW, Berg MP, Deprez RHL, Post JG, Mil AM, Asselbergs FW, Christiaans I, Langen IM, Wilde AAM, Boer RA, Jongbloed JDH, Pinto YM, Tintelen JP (2013) Genetic analysis in 418 index patients with idiopathic dilated cardiomyopathy: overview of 10 years’ experience. Eur J Heart Fail 15(6):628–636. doi:10.1093/eurjhf/hft013
Team RC (2014) R: A language and environment for statistical computing. Available via R Foundation for Statistical Computing. http://www.R-project.org
Hertz CL F-ML, Frank-Hansen R, Morling N, Bundgaard H (2014) A comparison of genetic findings in sudden cardiac death victims and cardiac patients: the importance of phenotypic classification. Europace In press. doi:10.1093/europace/euu210
Chugh SS, Senashova O, Watts A, Tran PT, Zhou Z, Gong Q, Titus JL, Hayflick SJ (2004) Postmortem molecular screening in unexplained sudden death. J Am Coll Cardiol 43(9):1625–1629. doi:10.1016/j.jacc.2003.11.052
Skinner JR, Crawford J, Smith W, Aitken A, Heaven D, Evans C-A, Hayes I, Neas KR, Stables S, Koelmeyer T, Denmark L, Vuletic J, Maxwell F, White K, Yang T, Roden DM, Leren TP, Shelling A, Love DR (2011) Prospective, population-based long QT molecular autopsy study of postmortem negative sudden death in 1 to 40 year olds. Heart Rhythm 8(3):412–419. doi:10.1016/j.hrthm.2010.11.016
Winkel BG, Larsen MK, Berge KE, Leren TP, Nissen PH, Olesen MS, Hollegaard MV, Jespersen T, Yuan L, Nielsen N, Haunsø S, Svendsen JH, Wang Y, Kristensen IB, Jensen HK, Tfelt-Hansen J, Banner J (2012) The prevalence of mutations in KCNQ1, KCNH2, and SCN5A in an unselected national cohort of young sudden unexplained death cases. J Cardiovasc Electrophysiol 23(10):1092–1098. doi:10.1111/j.1540-8167.2012.02371.x
Tester DJ, Will ML, Haglund CM, Ackerman MJ (2005) Compendium of cardiac channel mutations in 541 consecutive unrelated patients referred for long QT syndrome genetic testing. Heart Rhythm 2(5):507–517. doi:10.1016/j.hrthm.2005.01.020
Berge KE, Haugaa KH, Früh A, Anfinsen OG, Gjesdal K, Siem G, Øyen N, Greve G, Carlsson A, Rognum TO, Hallerud M, Kongsgård E, Amlie JP, Leren TP (2008) Molecular genetic analysis of long QT syndrome in Norway indicating a high prevalence of heterozygous mutation carriers. Scand J Clin Lab Investig 68(5):362–368. doi:10.1080/00365510701765643
Nakajima T, Kaneko Y, Saito A, Irie T, Tange S, Iso T, Kurabayashi M (2011) Identification of six novel SCN5A mutations in Japanese patients with Brugada syndrome. Int Heart J 52(1):27–31
Liang P, Liu W, Li C, Tao W, Li L, Hu D (2010) Genetic analysis of Brugada syndrome and congenital long-QT syndrome type 3 in the Chinese. J Cardiovasc Dis Res 1(2):69–74. doi:10.4103/0975-3583.64437
Priori SG, Napolitano C, Memmi M, Colombi B, Drago F, Gasparini M, DeSimone L, Coltorti F, Bloise R, Keegan R, Cruz Filho FES, Vignati G, Benatar A, DeLogu A (2002) Clinical and molecular characterization of patients with catecholaminergic polymorphic ventricular tachycardia. Circulation 106(1):69–74. doi:10.1161/01.CIR.0000020013.73106.D8
Van Driest SL, Jaeger MA, Ommen SR, Will ML, Gersh BJ, Tajik AJ, Ackerman MJ (2004) Comprehensive analysis of the beta-myosin heavy chain gene in 389 unrelated patients with hypertrophic cardiomyopathy. J Am Coll Cardiol 44(3):602–610. doi:10.1016/j.jacc.2004.04.039
Richard P, Charron P, Carrier L, Ledeuil C, Cheav T, Pichereau C, Benaiche A, Isnard R, Dubourg O, Burban M, Gueffet J-P, Millaire A, Desnos M, Schwartz K, Hainque B, Komajda M, for the EHFP (2003) Hypertrophic cardiomyopathy. Circulation 107(17):2227–2232. doi:10.1161/01.CIR.0000066323.15244.54
Papadakis M, Raju H, Behr ER, Noronha SVD, Spath N, Kouloubinis A, Sheppard MN, Sharma S (2013) Sudden cardiac death with autopsy findings of uncertain significance potential for erroneous interpretation. Circ Arrhythm Electrophysiol 6(3):588–596. doi:10.1161/CIRCEP.113.000111
Andreasen C, Refsgaard L, Nielsen JB, Sajadieh A, Winkel BG, Tfelt-Hansen J, Haunso S, Holst AG, Svendsen JH, Olesen MS (2013) Mutations in genes encoding cardiac ion channels previously associated with sudden infant death syndrome (SIDS) are present with high frequency in new exome data. Can J Cardiol 29(9):1104–1109. doi:10.1016/j.cjca.2012.12.002
Ordovas JM, Smith CE (2010) Epigenetics and cardiovascular disease. Nat Rev Cardiol 7(9):510–519. doi:10.1038/nrcardio.2010.104
Acknowledgments
This work was supported by Ellen and Aage Andersen’s foundation. The authors thank Francisc-Raul Kantor for the bioinformatics support.
Conflict of interest
The authors declare that they have no conflict of interest. AGH is an employee of Novo Nordisk A/S, Denmark.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table S1
(DOCX 19 kb)
Rights and permissions
About this article
Cite this article
Hertz, C.L., Christiansen, S.L., Ferrero-Miliani, L. et al. Next-generation sequencing of 34 genes in sudden unexplained death victims in forensics and in patients with channelopathic cardiac diseases. Int J Legal Med 129, 793–800 (2015). https://doi.org/10.1007/s00414-014-1105-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00414-014-1105-y