Abstract
Purpose
Kisspeptins regulate the trophoblast invasion. The disturbance of this process might lead to the development of preeclampsia (PE). Diabetes mellitus (DM) is associated with the high rate of this complication. The main hypothesis was to investigate the placental protein expression of kisspeptin-1 (KISS1) and its receptor (KISS1R) in diabetic, preeclamptic, and healthy pregnancies.
Methods
Placentae (n = 65) were divided into the following groups: the control group (n = 20), either PE or non-PE type-1 diabetes mellitus (T1DM) (n = 10), either PE or non-PE type-2 diabetes mellitus (T2DM) (n = 10), either PE or non-PE gestational diabetes mellitus (GDM) (n = 10) and preeclampsia without diabetes (PE) (n = 15). Immunohistochemistry analysis was used for demonstrating the presence and location of KISS1/KISS1R in placental tissue and to measure the area of immunopositive expression. Correlation analyses were performed to detect the links between protein expression of these biomarkers and the main obstetric outcomes.
Results
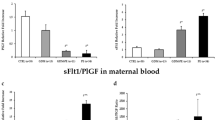
The highest placental protein expressions of KISS1 were detected in the PE (35.4%) and GDM (33.2%) groups. In case of DM, levels of KISS1 expression depended on the presence of PE and were higher compared with DM no PE and control groups: (30.6%) in T1DM + PE and (30.1%) in T2DM + PE group. The lowest expression was detected in the control group (14.1%). The expression of KISS1R was higher in DM and PE compared to the control group. We detected the strong direct link between PE and placental expression of KISS1 (r = 0.81) and KISS1R (r = 0.56), and inverse correlation link between KISS1 and preterm birth weight (r = − 0.73). The low correlation links were found between KISS1 and IUGR (r = 0.29), and preterm birth (r = 0.24). The same trend was detected for KISS1R. We did not find any significant correlations between placental expressions of KISS/KISS1R and placental weight or HbA1c levels.
Conclusion
Increased expression levels of KISS1 and KISS1R in case of diabetes mellitus may play a role in the altered placentation process and lead to the development of preeclampsia.


Similar content being viewed by others
References
Hiden U, Bilban M, Knöfler M, Desoye G (2007) Kisspeptins and the placenta: regulation of trophoblast invasion. Rev Endocr Metab Disord 8:31–39. https://doi.org/10.1007/s11154-007-9030-8
Gottsch ML, Clifton DK, Steiner RA (2009) From KISS1 to kisspeptins: an historical perspective and suggested nomenclature. Peptides 30:4–9. https://doi.org/10.1016/j.peptides.2008.06.016
Janneau J-L, Maldonado-Estrada J, Tachdjian G et al (2002) Transcriptional expression of genes involved in cell invasion and migration by normal and tumoral trophoblast cells. J Clin Endocrinol Metab 87:5336–5339. https://doi.org/10.1210/jc.2002-021093
Mead EJ, Maguire JJ, Kuc RE, Davenport AP (2009) Kisspeptins: a multifunctional peptide system with a role in reproduction, cancer and the cardiovascular system. Br J Pharmacol 151:1143–1153. https://doi.org/10.1038/sj.bjp.0707295
Ohtaki T, Shintani Y, Honda S et al (2001) Metastasis suppressor gene KiSS-1 encodes peptide ligand of a G-protein-coupled receptor. Nature 411:613–617. https://doi.org/10.1038/35079135
Yan C, Wang H, Boyd DD (2001) KiSS-1 Represses 92-kDa type IV collagenase expression by down-regulating NF-κB binding to the promoter as a consequence of IκBα-induced block of p65/p50 nuclear translocation. J Biol Chem 276:1164–1172. https://doi.org/10.1074/jbc.M008681200
Cartwright JE, Williams PJ (2012) Altered placental expression of kisspeptin and its receptor in pre-eclampsia. J Endocrinol 214:79–85. https://doi.org/10.1530/JOE-12-0091
Bilban M, Ghaffari-Tabrizi N, Hintermann E et al (2004) Kisspeptin-10, a KiSS-1/metastin-derived decapeptide, is a physiological invasion inhibitor of primary human trophoblasts. J Cell Sci 117:1319–1328. https://doi.org/10.1242/jcs.00971
Horikoshi Y, Matsumoto H, Takatsu Y et al (2003) Dramatic elevation of plasma metastin concentrations in human pregnancy: metastin as a novel placenta-derived hormone in humans. J Clin Endocrinol Metab 88:914–919. https://doi.org/10.1210/jc.2002-021235
Steegers EA, von Dadelszen P, Duvekot JJ, Pijnenborg R (2010) Pre-eclampsia. Lancet 376:631–644. https://doi.org/10.1016/S0140-6736(10)60279-6
Kawamura T, Kakogawa J, Takeuchi Y et al (2007) Measurement of placental oxygenation by transabdominal near-infrared spectroscopy. Am J Perinatol 24:161–166. https://doi.org/10.1055/s-2006-958155
Nelson SM, Coan PM, Burton GJ, Lindsay RS (2009) Placental structure in type 1 diabetes: relation to fetal insulin, leptin, and IGF-I. Diabetes 58:2634–2641. https://doi.org/10.2337/db09-0739
Desoye G, Hauguel-de Mouzon S (2007) The Human placenta in gestational diabetes mellitus: the insulin and cytokine network. Diabetes Care 30:S120–S126. https://doi.org/10.2337/dc07-s203
Metzger BE (2010) International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care 33:676–682. https://doi.org/10.2337/dc09-1848
Brown MA, Magee LA, Kenny LC et al (2018) The hypertensive disorders of pregnancy: ISSHP classification, diagnosis & management recommendations for international practice. Pregnancy Hypertens 13:291–310. https://doi.org/10.1016/j.preghy.2018.05.004
Roseweir AK, Katz AA, Millar RP (2012) Kisspeptin-10 inhibits cell migration in vitro via a receptor-GSK3 beta-FAK feedback loop in HTR8SVneo cells. Placenta 33:408–415. https://doi.org/10.1016/j.placenta.2012.02.001
Huppertz B (2008) Placental origins of preeclampsia. Hypertension 51:970–975. https://doi.org/10.1161/HYPERTENSIONAHA.107.107607
Raymond D, Peterson E (2011) A critical review of early-onset and late-onset preeclampsia. Obstet Gynecol Surv 66:497–506. https://doi.org/10.1097/OGX.0b013e3182331028
Colomiere M, Permezel M, Riley C et al (2009) Defective insulin signaling in placenta from pregnancies complicated by gestational diabetes mellitus. Eur J Endocrinol 160:567–578. https://doi.org/10.1530/EJE-09-0031
Ćetković A, Miljic D, Ljubić A et al (2012) Plasma kisspeptin levels in pregnancies with diabetes and hypertensive disease as a potential marker of placental dysfunction and adverse perinatal outcome. Endocr Res 37:78–88. https://doi.org/10.3109/07435800.2011.639319
Vazquez-Alaniz F, Galaviz-Hernandez C, Marchat LA et al (2011) Comparative expression profiles for KiSS-1 and REN genes in preeclamptic and healthy placental tissues. Eur J Obstet Gynecol Reprod Biol 159:67–71. https://doi.org/10.1016/j.ejogrb.2011.07.019
Zhang H, Long Q, Ling L et al (2011) Elevated expression of KiSS-1 in placenta of preeclampsia and its effect on trophoblast. Reprod Biol 11:99–115. https://doi.org/10.1016/S1642-431X(12)60048-5
Matjila M, Millar R, van der Spuy Z, Katz A (2016) Elevated placental expression at the maternal–fetal interface but diminished maternal circulatory kisspeptin in preeclamptic pregnancies. Pregnancy Hypertens An Int J Women’s Cardiovasc Heal 6:79–87. https://doi.org/10.1016/j.preghy.2015.11.001
Qiao C, Wang C, Zhao J et al (2012) Elevated expression of KiSS-1 in placenta of chinese women with early-onset preeclampsia. PLoS ONE 7:e48937. https://doi.org/10.1371/journal.pone.0048937
Qiao C, Cheng D-L, Zhang S-L et al (2005) The role of KiSS-1 and matrix metalloproteinase-9 in regulation of invasion of trophoblasts. Zhonghua Yi Xue Za Zhi 85:839–842
Loegl J, Nussbaumer E, Cvitic S et al (2017) GDM alters paracrine regulation of feto-placental angiogenesis via the trophoblast. Lab Investig 97:409–418. https://doi.org/10.1038/labinvest.2016.149
Armstrong RA, Reynolds RM, Leask R et al (2009) Decreased serum levels of kisspeptin in early pregnancy are associated with intra-uterine growth restriction and pre-eclampsia. Prenat Diagn 29:982–985. https://doi.org/10.1002/pd.2328
Smets EML, Deurloo KL, Go ATJI et al (2008) Decreased plasma levels of metastin in early pregnancy are associated with small for gestational age neonates. Prenat Diagn 28:299–303. https://doi.org/10.1002/pd.1969
Acknowledgements
We thank all the patients who participated in this study and staff who assisted with sample collection.
Author information
Authors and Affiliations
Contributions
KRV: Project development, Manuscript writing, Data analysis. DAO: Project development, Data Collection. AEN: Data analysis, Data collection. OAR: Data collection. AON: Manuscript editing. PVO: Manuscript editing. KIM: Manuscript editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
The local ethics committee approved the presented study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kapustin, R.V., Drobintseva, A.O., Alekseenkova, E.N. et al. Placental protein expression of kisspeptin-1 (KISS1) and the kisspeptin-1 receptor (KISS1R) in pregnancy complicated by diabetes mellitus or preeclampsia. Arch Gynecol Obstet 301, 437–445 (2020). https://doi.org/10.1007/s00404-019-05408-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-019-05408-1