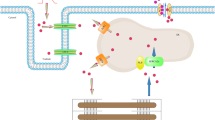
Abstract
In different pathological situations, cardiac cells undergo hyperosmotic stress (HS) and cell shrinkage. This change in cellular volume has been associated with contractile dysfunction and cell death. Given that nitric oxide (NO) is a well-recognized modulator of cardiac contractility and cell survival, we evaluated whether HS increases NO production and its impact on the negative inotropic effect observed during this type of stress. Superfusing cardiac myocytes with a hypertonic solution (HS: 440 mOsm) decreased cell volume and increased NO-sensitive DAF-FM fluorescence compared with myocytes superfused with an isotonic solution (IS: 309 mOsm). When cells were exposed to HS in addition to different inhibitors: L-NAME (NO synthase inhibitor), nitroguanidine (nNOS inhibitor), and Wortmannin (eNOS inhibitor) cell shrinkage occurred in the absence of NO release, suggesting that HS activates nNOS and eNOS. Consistently, western blot analysis demonstrated that maintaining cardiac myocytes in HS promotes phosphorylation and thus, activation of nNOS and eNOS compared to myocytes maintained in IS. HS-induced nNOS and eNOS activation and NO production were also prevented by AMPK inhibition with Dorsomorphin (DORSO). In addition, the HS-induced negative inotropic effect was exacerbated in the presence of either L-NAME, DORSO, ODQ (guanylate cyclase inhibitor), or KT5823 (PKG inhibitor), suggesting that NO provides contractile support via a cGMP/PKG-dependent mechanism. Our findings suggest a novel mechanism of AMPK-dependent NO release in cardiac myocytes with putative pathophysiological relevance determined, at least in part, by its capability to reduce the extent of contractile dysfunction associated with hyperosmotic stress.






Similar content being viewed by others
References
Allen DG, Smith GL (1987) The effects of hypertonicity on tension and intracellular calcium concentration in ferret ventricular muscle. J Physiol 383:425–439. https://doi.org/10.1113/jphysiol.1987.sp016418
Balligand JL, Ungureanu-Longrois D, Simmons WW, Pimental D, Malinski TA, Kapturczak M, Taha Z, Lowenstein CJ, Davidoff AJ, Kelly RA et al (1994) Cytokine-inducible nitric oxide synthase (iNOS) expression in cardiac myocytes. Characterization and regulation of iNOS expression and detection of iNOS activity in single cardiac myocytes in vitro. J Biol Chem 269:27580–27588 (PMID: 7525557)
Barbour HG, Loomis NM, Frankman RW, Warner JH (1924) Heat regulation and water exchange: V. The phase of blood dilution in man. J Physiol 59:300–305. https://doi.org/10.1113/jphysiol.1924.sp002185
Barouch LA, Harrison RW, Skaf MW, Rosas GO, Cappola TP, Kobeissi ZA, Hobai IA, Lemmon CA, Burnett AL, O’Rourke B, Rodriguez ER, Huang PL, Lima JA, Berkowitz DE, Hare JM (2002) Nitric oxide regulates the heart by spatial confinement of nitric oxide synthase isoforms. Nature 416:337–339. https://doi.org/10.1038/416005a
Bibli SI, Andreadou I, Chatzianastasiou A, Tzimas C, Sanoudou D, Kranias E, Brouckaert P, Coletta C, Szabo C, Kremastinos DT, Iliodromitis EK, Papapetropoulos A (2015) Cardioprotection by H2S engages a cGMP-dependent protein kinase G/phospholamban pathway. Cardiovasc Res 106:432–442. https://doi.org/10.1093/cvr/cvv129
Bice JS, Jones BR, Chamberlain GR, Baxter GF (2016) Nitric oxide treatments as adjuncts to reperfusion in acute myocardial infarction: a systematic review of experimental and clinical studies. Basic Res Cardiol 111:23. https://doi.org/10.1007/s00395-016-0540-y
Boo YC, Sorescu GP, Bauer PM, Fulton D, Kemp BE, Harrison DG, Sessa WC, Jo H (2003) Endothelial NO synthase phosphorylated at SER635 produces NO without requiring intracellular calcium increase. Free Radic Biol Med 35:729–741. https://doi.org/10.1016/s0891-5849(03)00397-6
Brette F, Calaghan SC, Lappin S, White E, Colyer J, Le Guennec JY (2000) Biphasic effects of hyposmotic challenge on excitation-contraction coupling in rat ventricular myocytes. Am J Physiol Heart Circ Physiol 279:H1963–H1971. https://doi.org/10.1152/ajpheart.2000.279.4.h1963
Chen ZP, Mitchelhill KI, Michell BJ, Stapleton D, Rodriguez-Crespo I, Witters LA, Power DA, Ortiz de Montellano PR, Kemp BE (1999) AMP-activated protein kinase phosphorylation of endothelial NO synthase. FEBS Lett 443:285–289. https://doi.org/10.1016/s0014-5793(98)01705-0
Fassett JT, Hu X, Xu X, Lu Z, Zhang P, Chen Y, Bache RJ (2013) AMPK attenuates microtubule proliferation in cardiac hypertrophy. Am J Physiol Heart Circ Physiol 304:H749–H758. https://doi.org/10.1152/ajpheart.00935.2011
Feron O, Belhassen L, Kobzik L, Smith TW, Kelly RA, Michel T (1996) Endothelial nitric oxide synthase targeting to caveolae. Specific interactions with caveolin isoforms in cardiac myocytes and endothelial cells. J Biol Chem 271:22810–22814. https://doi.org/10.1074/jbc.271.37.22810
Fulop M, Tannenbaum H, Dreyer N (1973) Ketotic hyperosmolar coma. Lancet 2:635–639. https://doi.org/10.1016/s0140-6736(73)92478-1
Galvez A, Morales MP, Eltit JM, Ocaranza P, Carrasco L, Campos X, Sapag-Hagar M, Diaz-Araya G, Lavandero S (2001) A rapid and strong apoptotic process is triggered by hyperosmotic stress in cultured rat cardiac myocytes. Cell Tissue Res 304:279–285. https://doi.org/10.1007/s004410100358
Garbincius JF, Michele DE (2015) Dystrophin-glycoprotein complex regulates muscle nitric oxide production through mechanoregulation of AMPK signaling. Proc Natl Acad Sci USA 112:13663–13668. https://doi.org/10.1073/pnas.1512991112
Gonano LA, Morell M, Burgos JI, Dulce RA, De Giusti VC, Aiello EA, Hare JM, Vila Petroff M (2014) Hypotonic swelling promotes nitric oxide release in cardiac ventricular myocytes: impact on swelling-induced negative inotropic effect. Cardiovasc Res 104:456–466. https://doi.org/10.1093/cvr/cvu230
Hao J, Kim HS, Choi W, Ha TS, Ahn HY, Kim CH (2010) Mechanical stretch-induced protection against myocardial ischemia-reperfusion injury involves AMP-activated protein kinase. Korean J Physiol Pharmacol 14:1–9. https://doi.org/10.4196/kjpp.2010.14.1.1
Hayashi Y, Nishio M, Naito Y, Yokokura H, Nimura Y, Hidaka H, Watanabe Y (1999) Regulation of neuronal nitric-oxide synthase by calmodulin kinases. J Biol Chem 274:20597–20602. https://doi.org/10.1074/jbc.274.29.20597
Heinzel FR, Gres P, Boengler K, Duschin A, Konietzka I, Rassaf T, Snedovskaya J, Meyer S, Skyschally A, Kelm M, Heusch G, Schulz R (2008) Inducible nitric oxide synthase expression and cardiomyocyte dysfunction during sustained moderate ischemia in pigs. Circ Res 103:1120–1127. https://doi.org/10.1161/circresaha.108.186015
Heusch G, Post H, Michel MC, Kelm M, Schulz R (2000) Endogenous nitric oxide and myocardial adaptation to ischemia. Circ Res 87:146–152. https://doi.org/10.1161/01.res.87.2.146
Hoffmann EK, Simonsen LO (1989) Membrane mechanisms in volume and pH regulation in vertebrate cells. Physiol Rev 69:315–382. https://doi.org/10.1152/physrev.1989.69.2.315
Howarth FC, Qureshi MA, White E (2002) Effects of hyperosmotic shrinking on ventricular myocyte shortening and intracellular Ca(2+) in streptozotocin-induced diabetic rats. Pflugers Arch 444:446–451. https://doi.org/10.1007/s00424-002-0830-0
Jourd’heuil D (2002) Increased nitric oxide-dependent nitrosylation of 4,5-diaminofluorescein by oxidants: implications for the measurement of intracellular nitric oxide. Free Radic Biol Med 33:676–684. https://doi.org/10.1016/s0891-5849(02)00955-3
Kaakinen M, Reichelt ME, Ma Z, Ferguson C, Martel N, Porrello ER, Hudson JE, Thomas WG, Parton RG, Headrick JP (2017) Cavin-1 deficiency modifies myocardial and coronary function, stretch responses and ischaemic tolerance: roles of NOS over-activity. Basic Res Cardiol 112:24. https://doi.org/10.1007/s00395-017-0613-6
Kakinuma Y, Zhang Y, Ando M, Sugiura T, Sato T (2004) Effect of electrical modification of cardiomyocytes on transcriptional activity through 5′-AMP-activated protein kinase. J Cardiovasc Pharmacol 44(Suppl 1):S435–S438. https://doi.org/10.1097/01.fjc.0000166318.91623.f9
Kimura C, Koyama T, Oike M, Ito Y (2000) Hypotonic stress-induced NO production in endothelium depends on endogenous ATP. Biochem Biophys Res Commun 274:736–740. https://doi.org/10.1006/bbrc.2000.3205
Kleindienst A, Battault S, Belaidi E, Tanguy S, Rosselin M, Boulghobra D, Meyer G, Gayrard S, Walther G, Geny B, Durand G, Cazorla O, Reboul C (2016) Exercise does not activate the beta3 adrenergic receptor-eNOS pathway, but reduces inducible NOS expression to protect the heart of obese diabetic mice. Basic Res Cardiol 111:40. https://doi.org/10.1007/s00395-016-0559-0
Lado MG, Sheu SS, Fozzard HA (1984) Effects of tonicity on tension and intracellular sodium and calcium activities in sheep heart. Circ Res 54:576–585. https://doi.org/10.1161/01.res.54.5.576
Li H, Wan A (2015) Fluorescent probes for real-time measurement of nitric oxide in living cells. Analyst 140:7129–7141. https://doi.org/10.1039/c5an01628b
Loiselle DS, Stienen GJ, van Hardeveld C, van der Meulen ET, Zahalak GI, Daut J, Elzinga G (1996) The effect of hyperosmolality on the rate of heat production of quiescent trabeculae isolated from the rat heart. J Gen Physiol 108:497–514. https://doi.org/10.1085/jgp.108.6.497
Maldonado C, Cea P, Adasme T, Collao A, Diaz-Araya G, Chiong M, Lavandero S (2005) IGF-1 protects cardiac myocytes from hyperosmotic stress-induced apoptosis via CREB. Biochem Biophys Res Commun 336:1112–1118. https://doi.org/10.1016/j.bbrc.2005.08.245
Martin C, Schulz R, Post H, Boengler K, Kelm M, Kleinbongard P, Gres P, Skyschally A, Konietzka I, Heusch G (2007) Microdialysis-based analysis of interstitial NO in situ: NO synthase-independent NO formation during myocardial ischemia. Cardiovasc Res 74:46–55. https://doi.org/10.1016/j.cardiores.2006.12.020
Matsubara M, Hayashi N, Jing T, Titani K (2003) Regulation of endothelial nitric oxide synthase by protein kinase C. J Biochem 133:773–781. https://doi.org/10.1093/jb/mvg099
Murphy BA, Fakira KA, Song Z, Beuve A, Routh VH (2009) AMP-activated protein kinase and nitric oxide regulate the glucose sensitivity of ventromedial hypothalamic glucose-inhibited neurons. Am J Physiol Cell Physiol 297:C750–C758. https://doi.org/10.1152/ajpcell.00127.2009
Netti VA, Iovane AN, Vatrella MC, Magnani ND, Evelson PA, Zotta E, Fellet AL, Balaszczuk AM (2016) Dehydration affects cardiovascular nitric oxide synthases and caveolins in growing rats. Eur J Nutr 55:33–43. https://doi.org/10.1007/s00394-014-0820-y
Netti VA, Iovane AN, Vatrella MC, Zotta E, Fellet AL, Balaszczuk AM (2016) Effects of nitric oxide system and osmotic stress on Aquaporin-1 in the postnatal heart. Biomed Pharmacother 81:225–234. https://doi.org/10.1016/j.biopha.2016.03.050
Niu X, Zhao L, Li X, Xue Y, Wang B, Lv Z, Chen J, Sun D, Zheng Q (2014) beta3-Adrenoreceptor stimulation protects against myocardial infarction injury via eNOS and nNOS activation. PLoS ONE 9:e98713. https://doi.org/10.1371/journal.pone.0098713
Pasquel FJ, Umpierrez GE (2014) Hyperosmolar hyperglycemic state: a historic review of the clinical presentation, diagnosis, and treatment. Diabetes Care 37:3124–3131. https://doi.org/10.2337/dc14-0984
Petroff MG, Kim SH, Pepe S, Dessy C, Marban E, Balligand JL, Sollott SJ (2001) Endogenous nitric oxide mechanisms mediate the stretch dependence of Ca2+ release in cardiomyocytes. Nat Cell Biol 3:867–873. https://doi.org/10.1038/ncb1001-867
Rassaf T, Poll LW, Brouzos P, Lauer T, Totzeck M, Kleinbongard P, Gharini P, Andersen K, Schulz R, Heusch G, Modder U, Kelm M (2006) Positive effects of nitric oxide on left ventricular function in humans. Eur Heart J 27:1699–1705. https://doi.org/10.1093/eurheartj/ehl096
Schulz R, Kelm M, Heusch G (2004) Nitric oxide in myocardial ischemia/reperfusion injury. Cardiovasc Res 61:402–413. https://doi.org/10.1016/j.cardiores.2003.09.019
Takeda-Nakazawa H, Harada N, Shen J, Kubo N, Zenner HP, Yamashita T (2007) Hyposmotic stimulation-induced nitric oxide production in outer hair cells of the guinea pig cochlea. Hear Res 227:59–70. https://doi.org/10.1016/j.heares.2006.09.007
Tsai EJ, Kass DA (2009) Cyclic GMP signaling in cardiovascular pathophysiology and therapeutics. Pharmacol Ther 122:216–238. https://doi.org/10.1016/j.pharmthera.2009.02.009
Vila-Petroff MG, Younes A, Egan J, Lakatta EG, Sollott SJ (1999) Activation of distinct cAMP-dependent and cGMP-dependent pathways by nitric oxide in cardiac myocytes. Circ Res 84:1020–1031. https://doi.org/10.1161/01.res.84.9.1020
Vitecek J, Reinohl V, Jones RL (2008) Measuring NO production by plant tissues and suspension cultured cells. Mol Plant 1:270–284. https://doi.org/10.1093/mp/ssm020
Xie H, Zhu PH (2006) Effect of osmotic stress on spontaneous calcium sparks in rat ventricular myocytes. Acta Pharmacol Sin 27:877–887. https://doi.org/10.1111/j.1745-7254.2006.00371.x
Xu X, Zhang P, Kwak D, Fassett J, Yue W, Atzler D, Hu X, Liu X, Wang H, Lu Z, Guo H, Schwedhelm E, Boger RH, Chen P, Chen Y (2017) Cardiomyocyte dimethylarginine dimethylaminohydrolase-1 (DDAH1) plays an important role in attenuating ventricular hypertrophy and dysfunction. Basic Res Cardiol 112:55. https://doi.org/10.1007/s00395-017-0644-z
Ziolo MT, Kohr MJ, Wang H (2008) Nitric oxide signaling and the regulation of myocardial function. J Mol Cell Cardiol 45:625–632. https://doi.org/10.1016/j.yjmcc.2008.07.015
Acknowledgements
The technical support of Monica Rando and Omar Castillo and confocal imaging assistance of Lucia Pagola are gratefully acknowledged.
Funding
This study was supported by Grants PICT 1678 from FONCyT and PIP 0270 from CONICET to M.V.P.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interest.
Rights and permissions
About this article
Cite this article
Morell, M., Burgos, J.I., Gonano, L.A. et al. AMPK-dependent nitric oxide release provides contractile support during hyperosmotic stress. Basic Res Cardiol 113, 7 (2018). https://doi.org/10.1007/s00395-017-0665-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-017-0665-7