Abstract
Purpose
Over recent years, 5-aminoluvolinic acid (5ALA) has been increasingly used for resection guidance in adult high-grade gliomas. However, amongst pediatric patients, publication of intraoperative fluorescence has been limited, with inconsistent outcomes. We describe our experience and intraoperative finding amongst children with various brain tumors that were given 5ALA prior to tumor resection.
Methods
Since October 2014, data regarding intraoperative findings amongst children that received 5ALA prior to tumor resection were prospectively collected. Inclusion criteria included any intracranial tumor amongst children 3–18 years of age. Data included intraoperative findings (regarding fluorescence of the tumor), as well as postoperative follow-up and documentation of complications.
Results
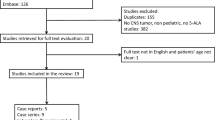
Fourteen children were included, covering a wide pathological spectrum: pilocytic astrocytoma (PA) (6), medulloblastoma (2), and one each of DNET, hemangiopericytoma, hemangioblastoma, ganglioneuroblastoma, oligodendroglioma grade II (OD), and ganglioglioma grade I. Fluorescence was clearly visible in one case (PA), and in a heterogeneous and slighter degree in two (PA, OD). One patient had a rash, fever, and leukocytosis 6 days after surgery and died 1 month later from extensive tumor progression (large cell medulloblastoma with leptomeningeal spread).
Conclusion
5ALA showed a low rate of fluorescence amongst this pediatric brain tumor cohort. These findings are consistent with the literature, where the role of 5ALA in guidance of pediatric brain tumor resection is limited mainly to glioblastoma multiforme. This stems not only from the low rate of significant fluorescence, but also from inherent structural properties of these lesions such as color, consistency, and invasion.



Similar content being viewed by others
References
Stummer W, Stocker S, Novotny A, Heimann A, Sauer O, Kempski O, Plesnila N, Wietzorrek J, Reulen HJ (1998) In vitro and in vivo porphyrin accumulation by C6 glioma cells after exposure to 5-aminolevulinic acid. J Photochem Photobiol B 45:160–169
Stummer W, Novotny A, Stepp H, Goetz C, Bise K, Reulen HJ (2000) Fluorescence-guided resection of glioblastoma multiforme by using 5-aminolevulinic acid-induced porphyrins: a prospective study in 52 consecutive patients. J Neurosurg 93:1003–1013
Stummer W, Pichlmeier U, Meinel T, Wiestler OD, Zanella F, Reulen HJ (2006) Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: a randomised controlled multicentre phase III trial. Lancet Oncol 7:392–401
Barbagallo GM, Certo F, Heiss K, Albanese V (2014) 5-ALA fluorescence-assisted surgery in pediatric brain tumors: report of three cases and review of the literature. Br J Neurosurg 28(6):750–754
Beez T, Sarikaya-Seiwert S, Steiger HJ, Hanggi D (2014) Fluorescence-guided surgery with 5-aminolevulinic acid for resection of brain tumors in children—a technical report. Acta Neurochir 156:597–604
Bernal Garcia LM, Cabezudo Artero JM, Royano Sanchez M, Marcelo Zamorano MB, Lopez Macias M (2015) Fluorescence-guided resection with 5-aminolevulinic acid of meningeal sarcoma in a child. Child’s nervous system : ChNS : official journal of the International Society for Pediatric Neurosurgery 31:1177–1180
Eicker S, Sarikaya-Seiwert S, Borkhardt A, Gierga K, Turowski B, Heiroth HJ, Steiger HJ, Stummer W (2011) ALA-induced porphyrin accumulation in medulloblastoma and its use for fluorescence-guided surgery. Cent Eur Neurosurg 72:101–103
Moriuchi S, Yamada K, Dehara M, Teramoto Y, Soda T, Imakita M, Taneda M (2011) Use of 5-aminolevulinic acid for the confirmation of deep-seated brain tumors during stereotactic biopsy. Report of 2 cases. J Neurosurg 115:278–280
Preuss M, Renner C, Krupp W, Christiansen H, Fischer L, Merkenschlager A, Kiess W, Muller W, Manzo N, Meixensberger J, Nestler U (2013) The use of 5-aminolevulinic acid fluorescence guidance in resection of pediatric brain tumors. Child’s nervous system : ChNS : official journal of the International Society for Pediatric Neurosurgery 29:1263–1267
Ruge JR, Liu J (2009) Use of 5-aminolevulinic acid for visualization and resection of a benign pediatric brain tumor. J Neurosurg Pediatr 4:484–486
Skjoth-Rasmussen J, Bogeskov L, Sehested A, Klausen C, Broholm H, Nysom K (2015) The use of 5-ALA to assist complete removal of residual non-enhancing part of childhood medulloblastoma: a case report. Child’s nervous system : ChNS : official journal of the International Society for Pediatric Neurosurgery 31:2173–2177
Stummer W, Rodrigues F, Schucht P, Preuss M, Wiewrodt D, Nestler U, Stein M, Artero JM, Platania N, Skjoth-Rasmussen J, Della Puppa A, Caird J, Cortnum S, Eljamel S, Ewald C, Gonzalez-Garcia L, Martin AJ, Melada A, Peraud A, Brentrup A, Santarius T, Steiner HH, European ALAPBTSG (2014) Predicting the “usefulness” of 5-ALA-derived tumor fluorescence for fluorescence-guided resections in pediatric brain tumors: a European survey. Acta Neurochir 156:2315–2324
Ostrom QT, de Blank PM, Kruchko C, Petersen CM, Liao P, Finlay JL, Stearns DS, Wolff JE, Wolinsky Y, Letterio JJ, Barnholtz-Sloan JS (2015) Alex’s lemonade stand foundation infant and childhood primary brain and central nervous system tumors diagnosed in the United States in 2007–2011. Neuro-Oncology 16(Suppl 10):x1–x36
Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD, Kleihues P, Ellison DW (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131:803–820
Kamp MA, Grosser P, Felsberg J, Slotty PJ, Steiger HJ, Reifenberger G, Sabel M (2012) 5-aminolevulinic acid (5-ALA)-induced fluorescence in intracerebral metastases: a retrospective study. Acta Neurochir 154:223–228 discussion 228
Novotny A, Xiang J, Stummer W, Teuscher NS, Smith DE, Keep RF (2000) Mechanisms of 5-aminolevulinic acid uptake at the choroid plexus. J Neurochem 75:321–328
Schucht P, Beck J, Vajtai I, Raabe A (2011) Paradoxical fluorescence after administration of 5-aminolevulinic acid for resection of a cerebral melanoma metastasis. Acta Neurochir 153:1497–1499
Utsuki S, Miyoshi N, Oka H, Miyajima Y, Shimizu S, Suzuki S, Fujii K (2007a) Fluorescence-guided resection of metastatic brain tumors using a 5-aminolevulinic acid-induced protoporphyrin IX: pathological study. Brain Tumor Pathol 24:53–55
Utsuki S, Oka H, Sato S, Shimizu S, Suzuki S, Tanizaki Y, Kondo K, Miyajima Y, Fujii K (2007b) Histological examination of false positive tissue resection using 5-aminolevulinic acid-induced fluorescence guidance. Neurol Med Chir 47:210–213 discussion 213-214
Hefti M, Albert I, Luginbuehl V (2012) Phenytoin reduces 5-aminolevulinic acid-induced protoporphyrin IX accumulation in malignant glioma cells. J Neuro-Oncol 108:443–450
Acknowledgments
We thank Mrs. Adina Sherer for editing assistance, and Mrs. Sigal Freedman for graphical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Roth, J., Constantini, S. 5ALA in pediatric brain tumors is not routinely beneficial. Childs Nerv Syst 33, 787–792 (2017). https://doi.org/10.1007/s00381-017-3371-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-017-3371-8