Abstract
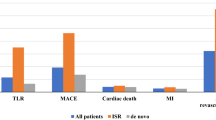
In 2013, a drug-coated balloon catheter (DCB) (SeQuent Please) for the treatment of coronary in-stent restenosis (ISR) was approved in Japan. The pre-marketing Japan domestic NP001 study demonstrated better outcomes of the DCB (n = 138) compared to plain balloon angioplasty (n = 72). After the introduction to marketing, a post-marketing surveillance (PMS) (n = 396) was conducted to evaluate the safety and efficacy of the DCB in Japanese routine clinical practice. The aim of this paper was to assess differences between the pre-marketing NP001 study and the PMS. Compared to the NP001 study, more complex lesions were treated in the PMS (type B2/C: 69.0% vs 20.4%, total occlusion: 11.2% vs 0%, p < 0.001, respectively) and target lesion was more frequently ISR related to drug-eluting stent (DES) (79.5% vs 39.4%, p < 0.001). Regarding clinical outcomes, the rate of target lesion revascularization (TLR) was higher in the PMS than in the NP001 study (TLR: 12.9% at 7 months and 17.6% at 12 months vs 2.8% at 6 months, p = 0.001, p < 0.001, respectively). Multivariable logistic regression analysis revealed that DES-ISR was a risk factor of TLR after DCB treatment for ISR (odds ratio: 5.77, 95% CI 1.75–18.95, p = 0.004). Among representative published trials using DCB for ISR, clinical outcomes are often worse in DES-ISR trials than those in bare metal stent-ISR trials. The rates of TLR in previous DES-ISR trials are similar to that in the current PMS (TLR at 12 months: 22.1% for ISAR-DESIRE 3, 15.3% for PEPCAD-DES, and 13.0% for RIBS IV). The effectiveness and safety of DCB for coronary ISR have been confirmed in the Japanese real-world survey. PMS would be useful to evaluate the safety and effectiveness of medical products throughout their total life cycles.
Similar content being viewed by others
Abbreviations
- BMS:
-
Bare metal stents
- BR:
-
Binary restenosis
- CABG:
-
Coronary artery bypass grafting
- CI:
-
Confidence intervals
- DAPT:
-
Dual antiplatelet therapy
- DCB:
-
Drug-coated balloon
- DES:
-
Drug-eluting stents
- DS:
-
Diameter stenosis
- GPSP:
-
Good post-marketing surveillance practice
- ISR:
-
In-stent restenosis
- LAD:
-
Left anterior descending artery
- LCX:
-
Left circumflex artery
- LMT:
-
Left main trunk
- LLL:
-
Late lumen loss
- MACE:
-
Major adverse cardiac events
- MI:
-
Myocardial infarction
- OR:
-
Odds ratio
- PCI:
-
Percutaneous coronary intervention
- PMDA:
-
Pharmaceuticals and Medical Devices Agency
- PMS:
-
Post-marketing surveillance
- POBA:
-
Plain old balloon angioplasty
- QCA:
-
Quantitative coronary angiography
- RCA:
-
Right coronary artery
- RVD:
-
Reference vessel diameter
- TLR:
-
Target lesion revascularization
- TVF:
-
Target vessel failure
- TVR:
-
Target vessel revascularization
References
Pharmaceuticals and Medical Devices Agency. Use-results survey system under the pharmaceuticals and medical devices act. https://www.pmda.go.jp/files/000160083.pdf. Accessed 17 Mar 2020. (in Japanese)
Pharmaceuticals and Medical Devices Agency. Fundamental point of view about use-results survey system. https://www.pmda.go.jp/files/000197726.pdf. Accessed 17 Mar 2020. (in Japanese)
Konishi A, Ho M, Shirai Y, Shirato H (2018) First approval of improved medical device conditional on use-result survey in Japan—regulatory review of polymer-free drug-coated biofreedom coronary stent. Circ J 82:1487–1490
Konishi A, Ho M, Ouchi T, Mitsutake Y, Shirato H (2019) Regulatory approval review of transcatheter mitral valve repair—difference in the indication between the USA and Japan. J Cardiol 74:13–18
Pharmaceuticals and Medical Devices Agency. Re-examinations. https://www.pmda.go.jp/review-services/reexamine-reevaluate/re-evaluations/0009.html. Accessed 17 Mar 2020. (in Japanese)
Review Report: SeQuent Please drug eluting balloon. http://www.pmda.go.jp/medical_devices/2013/M201300030/530100000_22500BZX00322000_A100_1.pdf. Accessed 17 Mar 2020. (in Japanese)
Re-evaluation Report: SeQuent Please drug eluting balloon. https://www.pmda.go.jp/medical_devices_reexam/2020/M20200319001/530100000_22500BZX00322000_100_1.pdf. Accessed 25 Mar 2020. (in Japanese)
Mehran R, Dangas G, Abizaid AS, Mintz GS, Lansky AJ, Satler LF, Pichard AD, Kent KM, Stone GW, Leon MB (1999) Angiographic patterns of in-stent restenosis: classification and implications for long-term outcome. Circulation 100:1872–1878
Lemos PA, van Mieghem CA, Arampatzis CA, Hoye A, Ong AT, McFadden E, Sianos G, van der Giessen WJ, de Feyter PJ, van Domburg RT, Serruys PW (2004) Post-sirolimus-eluting stent restenosis treated with repeat percutaneous intervention: late angiographic and clinical outcomes. Circulation 109:2500–2502
Mishkel GJ, Moore AL, Markwell S, Shelton MC, Shelton ME (2007) Long-term outcomes after management of restenosis or thrombosis of drug-eluting stents. J Am Coll Cardiol 49:181–184
Steinberg DH, Gaglia MA Jr, Pinto Slottow TL, Roy P, Bonello L, De Labriolle A, Lemesle G, Torguson R, Kineshige K, Xue Z, Suddath WO, Kent KM, Satler LF, Pichard AD, Lindsay J, Waksman R (2009) Outcome differences with the use of drug-eluting stents for the treatment of in-stent restenosis of bare-metal stents versus drug-eluting stents. Am J Cardiol 103:491–495
Stettler C, Wandel S, Allemann S, Kastrati A, Morice MC, Schömig A, Pfisterer ME, Stone GW, Leon MB, de Lezo JS, Goy JJ, Park SJ, Sabaté M, Suttorp MJ, Kelbaek H, Spaulding C, Menichelli M, Vermeersch P, Dirksen MT, Cervinka P, Petronio AS, Nordmann AJ, Diem P, Meier B, Zwahlen M, Reichenbach S, Trelle S, Windecker S, Jüni P (2007) Outcomes associated with drug-eluting and bare-metal stents: a collaborative network meta-analysis. Lancet 370:937–948
Byrne RA, Neumann FJ, Mehilli J, Pinieck S, Wolff B, Tiroch K, Schulz S, Fusaro M, Ott I, Ibrahim T, Hausleiter J, Valina C, Pache J, Laugwitz KL, Massberg S, Kastrati A, ISAR-DESIRE 3 investigators (2013) Paclitaxel-eluting balloons, paclitaxel-eluting stents, and balloon angioplasty in patients with restenosis after implantation of a drug-eluting stent (ISAR-DESIRE 3): a randomised, open-label trial. Lancet 381:461–467
Rittger H, Waliszewski M, Brachmann J, Hohenforst-Schmidt W, Ohlow M, Brugger A, Thiele H, Birkemeyer R, Kurowski V, Schlundt C, Zimmermann S, Lonke S, von Cranach M, Markovic S, Daniel WG, Achenbach S, Wöhrle J (2015) Long-term outcomes after treatment with a paclitaxel-coated balloon versus balloon angioplasty: insights from the PEPCAD-DES Study (Treatment of Drug-eluting Stent [DES] In-Stent Restenosis With SeQuent Please Paclitaxel-Coated Percutaneous Transluminal Coronary Angioplasty [PTCA] Catheter). JACC Cardiovasc Interv 8:1695–1700
Alfonso F, Pérez-Vizcayno MJ, Cárdenas A, García del Blanco B, García-Touchard A, López-Minguéz JR, Benedicto A, Masotti M, Zueco J, Iñiguez A, Velázquez M, Moreno R, Mainar V, Domínguez A, Pomar F, Melgares R, Rivero F, Jiménez-Quevedo P, Gonzalo N, Fernández C, Macaya C, RIBS IV Study Investigators (under auspices of Interventional Cardiology Working Group of Spanish Society of Cardiology) (2015) A prospective randomized trial of drug-eluting balloons versus everolimus-eluting stents in patients with in-stent restenosis of drug-eluting stents: the RIBS IV Randomized Clinical Trial. J Am Coll Cardiol 66:23–33
Scheller B, Clever YP, Kelsch B, Hehrlein C, Bocksch W, Rutsch W, Haghi D, Dietz U, Speck U, Böhm M, Cremers B (2012) Long-term follow-up after treatment of coronary in-stent restenosis with a paclitaxel-coated balloon catheter. JACC Cardiovasc Interv 5:323–330
Unverdorben M, Vallbracht C, Cremers B, Heuer H, Hengstenberg C, Maikowski C, Werner GS, Antoni D, Kleber FX, Bocksch W, Leschke M, Ackermann H, Boxberger M, Speck U, Degenhardt R, Scheller B (2015) Paclitaxel-coated balloon catheter versus paclitaxel-coated stent for the treatment of coronary in-stent restenosis: the three-year results of the PEPCAD II ISR study. EuroIntervention 11:926–934
Alfonso F, Pérez-Vizcayno MJ, García Del Blanco B, Otaegui I, Masotti M, Zueco J, Veláquez M, Sanchís J, García-Touchard A, Lázaro-García R, Moreu J, Bethencourt A, Cuesta J, Rivero F, Cárdenas A, Gonzalo N, Jiménez-Quevedo P, Fernández C, RIBS V Study Investigators (2016) Long-term results of everolimus-eluting stents versus drug-eluting balloons in patients with bare-metal in-stent restenosis: 3-year follow-up of the RIBS V Clinical Trial. JACC Cardiovasc Interv 9:1246–1255
JCS 2018 Guideline on Revascularization of Stable Coronary Artery Disease. http://j-circ.or.jp/guideline/. Accessed 17 Mar 2020. (in Japanese)
Neumann FJ, Sousa-Uva M, Ahlsson A, Alfonso F, Banning AP, Benedetto U, Byrne RA, Collet JP, Falk V, Head SJ, Jüni P, Kastrati A, Koller A, Kristensen SD, Niebauer J, Richter DJ, Seferovic PM, Sibbing D, Stefanini GG, Windecker S, Yadav R, Zembala MO, ESC Scientific Document Group (2019) 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J 40:87–165
Kang IS, Shehata I, Shin DH, Kim JS, Kim BK, Ko YG, Choi D, Jang Y, Hong MK (2016) Comparison between drug-coated balloon angioplasty and second-generation drug-eluting stent placement for the treatment of in-stent restenosis after drug-eluting stent implantation. Heart Vessels 31:1405–1411
Sakamoto Y, Yamawaki M, Araki M, Kobayashi N, Mori S, Tsutsumi M, Honda Y, Hirano K, Ito Y (2019) Comparison of 12-month angiographic outcomes between repeat drug-eluting stent implantation and drug-coated balloon treatment for restenotic lesion caused by stent fracture. Heart Vessels 34:1589–1594
Akutsu N, Ogaku A, Koyama Y, Fujito H, Ebuchi Y, Migita S, Morikawa T, Tamaki T, Mineki T, Kougo T, Kojima K, Iida K, Murata N, Nishida T, Oshima T, Sudo M, Kitano D, Haruta H, Fukamachi D, Takayama T, Hiro T, Hirayama A, Okumura Y (2019) Effect of drug-coated balloon angioplasty on in-stent restenotic coronary lesions analyzed with optical coherence tomography and serial coronary artery angioscopy. Heart Vessels 34:1925–1935
Her AY, Shin ES, Chung JH, Kim YH, Garg S, Lee JM, Doh JH, Nam CW, Koo BK (2019) Plaque modification and stabilization after paclitaxel-coated balloon treatment for de novo coronary lesions. Heart Vessels 34:1113–1121
Acknowledgments
We thank the members of the PMDA review team for their input and Editage (https://www.editage.com) for English editing. The views expressed in this article are those of the authors and do not necessarily reflect the official views of the PMDA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest. This paper was written with no external funding.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mitsutake, Y., Konishi, A., Shiba, T. et al. Differences in clinical outcomes between pre- and post-marketing clinical study following paclitaxel-coated balloon catheter treatment for coronary in-stent restenosis: from the Japanese regulatory viewpoint. Heart Vessels 36, 155–162 (2021). https://doi.org/10.1007/s00380-020-01676-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-020-01676-z