Abstract
Purpose
To correlate the presence of fungi with symptom flares, pain and urinary severity in a prospective, longitudinal study of women with IC/BPS enrolled in the MAPP Research Network.
Methods
Flare status, pelvic pain, urinary severity, and midstream urine were collected at baseline, 6 and 12 months from female IC/BPS participants with at least one flare and age-matched participants with no reported flares. Multilocus PCR coupled with electrospray ionization/mass spectrometry was used for identification of fungal species and genus. Associations between “mycobiome” (species/genus presence, relative abundance, Shannon’s/Chao1 diversity indices) and current flare status, pain, urinary severity were evaluated using generalized linear mixed models, permutational multivariate analysis of variance, Wilcoxon’s rank-sum test.
Results
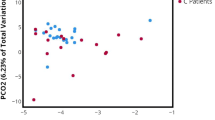
The most specific analysis detected 13 fungal species from 8 genera in 504 urine samples from 202 females. A more sensitive analysis detected 43 genera. No overall differences were observed in fungal species/genus composition or diversity by flare status or pain severity. Longitudinal analyses suggested greater fungal diversity (Chao1 Mean Ratio 3.8, 95% CI 1.3–11.2, p = 0.02) and a significantly greater likelihood of detecting any fungal species (OR = 5.26, 95% CI 1.1–25.8, p = 0.04) in high vs low urinary severity participants. Individual taxa analysis showed a trend toward increased presence and relative abundance of Candida (OR = 6.63, 95% CI 0.8–58.5, p = 0.088) and Malassezia (only identified in ‘high’ urinary severity phenotype) for high vs low urinary symptoms.
Conclusion
This analysis suggests the possibility that greater urinary symptom severity is associated with the urinary mycobiome urine in some females with IC/BPS.


Similar content being viewed by others
References
Sutcliffe S, Colditz GA, Goodman MS et al (2014) Urological chronic pelvic pain syndrome symptom flares: characterization of the full range of flares at two sites in the Multidisciplinary Approach to the Study of Chronic Pelvic Pain (MAPP) Research Network. BJU Int 114:916–925
Ackerman AL, Underhill DM (2017) The mycobiome of the human urinary tract: potential roles for fungi in urology. Ann Transl Med 5:31. https://doi.org/10.21037/atm.2016.12.69
Clemens JQ, Mullins C, Kusek JW et al (2014) The MAPP research network: a novel study of urologic chronic pelvic pain syndromes. BMC Urol 14:57. https://doi.org/10.1186/1471-2490-14-57
Nickel JC, Stephens A, Landis JR et al (2016) Assessment of the lower urinary tract microbiota during symptom flare in women with urologic chronic pelvic pain syndrome: a MAPP Network Study. J Urol 195:356–362
Landis JR, Williams DA, Lucia MS et al (2014) The MAPP research network: design, patient characterization and operations. BMC Urol 14:58. https://doi.org/10.1186/1471-2490-14-58
Griffith JW, Stephens-Shields AJ, Hou X et al (2016) Pain and urinary symptoms should not be combined into a single score: psychometric findings from the MAPP Research Network. J Urol 195:949–954
Ecker DJ, Sampath R, Massire C et al (2008) Ibis T5000: a universal biosensor approach for microbiology. Nat Rev Microbiol 6(7):553–558. https://doi.org/10.1038/nrmicro1918
Shin JH, Ranken R, Sefers SE et al (2013) Detection, identification, and distribution of fungi in bronchoalveolar lavage specimens by use of multilocus PCR coupled with electrospray ionization/mass spectrometry. J Clin Microbiol 51:136–141
Nickel JC, Stephens A, Landis R et al (2015) Search for microorganisms in men with urologic chronic pelvic pain syndrome: a culture-independent analysis in the MAPP research network. J Urol 194:127–135
Chao A (1984) Non-parametric estimation of the number of classes in a population. Scand J Stat 11:265–270
Shannon CE, Weaver W (1949) The mathematical theory of communication. The University of Illinois Press, Urbana, p 117
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc B 57:289–300
Thomas-White KJ, Kliethermes S, Rickey L et al (2017) Evaluation of the urinary microbiota of women with uncomplicated stress urinary incontinence. Am J Obstet Gynecol 216(1):55.e1–55.e16. https://doi.org/10.1016/j.ajog.2016.07.049
Pearce MM, Hilt EE, Rosenfeld AB et al (2014) The female urinary microbiome: a comparison of women with and without urgency urinary incontinence. MBio 5(4):e01283-14. https://doi.org/10.1128/mBio
Karstens L, Asquith M, Davin S et al (2016) Does the urinary microbiome play a role in urgency urinary incontinence and its severity? Front Cell Infect Microbiol. 6:78. https://doi.org/10.3389/fcimb.2016.00078
Pearce MM, Zilliox MJ, Rosenfeld AB et al (2015) The female urinary microbiome in urgency urinary incontinence. Am J Obstet Gynecol 213(3):347.e1-11. https://doi.org/10.1016/j.ajog.2015.07.009
Komesu YM, Richter HE, Carper B et al (2018) The urinary microbiome in women with mixed urinary incontinence compared to similarly aged controls. Int Urogynecol J 45:67. https://doi.org/10.1007/s00192-018-3683-6
Brubaker L, Nager CW, Richter HE et al (2014) Urinary bacteria in adult women with urgency urinary incontinence. Int Urogynecol 25(9):1179–1184. https://doi.org/10.1007/s00192-013-2325-2
Fok CS, Gao X, Lin H et al (2018) Urinary symptoms are associated with certain urinary microbes in urogynecologic surgical patients. Int Urogynecol J 29:1765–1771
Fouts DE, Pieper R, Szpakowski S et al (2012) Integrated next-generation sequencing of 16S rDNA and metaproteomics differentiate the healthy urine microbiome from asymptomatic bacteriuria in neuropathic bladder associated with spinal cord injury. J Transl Med 28(10):174. https://doi.org/10.1186/1479-5876-10-174
Siddiqui H, Lagesen K, Nederbragt AJ et al (2012) Alterations of microbiota in urine from women with interstitial cystitis. BMC Microbiol 12:205. https://doi.org/10.1186/1471-2180-12-205
Chen Y, Chen Z, Guo R et al (2011) Correlation between gastrointestinal fungi and varying degrees of chronic hepatitis B virus infection. Diagn Microbiol Infect Dis 70(4):492–498. https://doi.org/10.1016/j.diagmicrobio.2010.04.005
Zhang E, Tanaka T, Tajima M et al (2011) Characterization of the skin fungal microbiota in patients with atopic dermatitis and in healthy subjects. Microbiol Immunol 55(9):625–632. https://doi.org/10.1111/j.1348-0421.2011.00364.x
Park HK, Ha MH, Park SG et al (2012) Characterization of the fungal microbiota (mycobiome) in healthy and dandruff-afflicted human scalps. PLoS ONE 7(2):e32847. https://doi.org/10.1371/journal.pone.0032847
Ott SJ, Kühbacher T, Musfeldt M et al (2008) Fungi and inflammatory bowel diseases: alterations of composition and diversity. Scand J Gastroenterol 43:831–841
Delhaes L, Monchy S, Fréalle E et al (2012) The airway microbiota in cystic fibrosis: a complex fungal and bacterial community—implications for therapeutic management. PLoS ONE 7(4):e36313. https://doi.org/10.1371/journal.pone.0036313
Knutsen AP, Bush RK, Demain JG et al (2012) Fungi and allergic lower respiratory tract diseases. J Allergy Clin Immunol 129:280–291
Sokol H, Leducq V, Aschard H et al (2017) Fungal microbiota dysbiosis in IBD. Gut 66:1039–1048
Funding
The authors declare that this project was supported (including salary support) by peer reviewed research grants from the US National Institutes of Health: NIDDK: U01DK103271 (JCN, JEK, BS, JH, GDE), U01DK082316 (AS, JRL), U01DK082333 (AVB), U01DK103260 (JTA, ALA, JK), U01DK082315 (SS).
Author information
Authors and Affiliations
Consortia
Contributions
JCN, AS: protocol development, data management, data analysis, and manuscript writing/editing. JRL: protocol development, data management, data analysis, and manuscript editing. CM: protocol/project development, data collection and management, data analysis, and manuscript writing/editing. AvanB, GDE: protocol development, data collection and management, data analysis, and manuscript writing/editing. JTA, ALA, JK: protocol development, data analysis, and manuscript editing. SS: protocol/project development, data collection and management, data analysis, and manuscript editing. JEK, BS, JH: data collection and management, data analysis, and manuscript editing.
Corresponding author
Ethics declarations
Conflict of interest
CM is an employee of the NIH/NIDDK. The authors report no other potential conflict of interest.
Ethical responsibilities of the authors
The authors agree to the conditions outlined in the submission instructions.
Informed consent
Written informed general consent for specimen procurement and data analysis was obtained from all individual participants included in the study.
Research involving human participants
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Research involving animals
This article does not contain any studies with animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nickel, J.C., Stephens, A., Landis, J.R. et al. Urinary fungi associated with urinary symptom severity among women with interstitial cystitis/bladder pain syndrome (IC/BPS). World J Urol 38, 433–446 (2020). https://doi.org/10.1007/s00345-019-02764-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-019-02764-0