Abstract
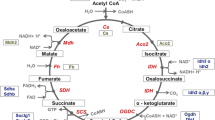
To date, the only reported metabolic and physiological roles for carnitine in Saccharomyces cerevisiae are related to the activity of the carnitine shuttle. In yeast, the shuttle transfers peroxisomal activated acetyl-residues to the mitochondria. However, acetyl-CoA can also be metabolised by the glyoxylate cycle to form succinate. The two pathways, therefore, provide a metabolic bypass for each other, and carnitine-dependent phenotypes have only been described in strains with non-functional peroxisomal citrate synthase, Cit2p. Here, we present evidence for a role of carnitine in stress protection that is independent of CIT2 and of the carnitine shuttle. Data show that carnitine improves growth during oxidative stress and in the presence of weak organic acids in wt and in CAT deletion strains. Our data also show that strains with single, double and triple deletions of the three CAT genes generally present identical phenotypes, but that the deletion of CAT2 decreases survival during oxidative stress in a carnitine-independent manner. Overexpression of single CAT genes does not lead to cross-complementation, suggesting a highly specific metabolic role for each enzyme. The data suggest that carnitine protects cells from oxidative and organic acid stress, while CAT2 contributes to the response to oxidative stress.









Similar content being viewed by others
References
Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG (1994) Current protocols in molecular biology. Wiley, New York
Barros MH, Bandy B, Tahara EB, Kowaltowski AJ (2004) Higher respiratory activity decreases mitochondrial reactive oxygen release and increases lifespan in Saccharomyces cerevisiae. J Biol Chem 279:49883–49888
Berben G, Dumont J, Gilliquet V, Bolle PA, Hilger F (1991) The YDp plasmids: a uniform set of vectors bearing versatile gene disruption cassettes for Saccharomyces cerevisiae. Yeast 7:475–477
Bieber LL (1988) Carnitine. Annual review of biochemistry 57:261–283
Calabrese V, Giuffrida Stella AM, Calvani M, Butterfield DA (2006) Acetylcarnitine and cellular stress response: roles in nutritional redox homeostasis and regulation of longevity genes. J Nutr Biochem 17:73–88
Gietz RD, Sugino A (1988) New yeast–Escherichia coli shuttle vectors constructed with in vitro mutagenized yeast genes lacking six-base pair restriction sites. Gene 74:527–534
Grey M, Brendel M (1994) Overexpression of the SNQ3/YAP1 gene confers hyperresistance to nitrosoguanidine in Saccharomyces cerevisiae via aglutathione-independent mechanism. Curr Genet 25:469–471
Hussain M, Lenard J (1991) Characterisation of PDR4, a Saccharomyces cerevisiae gene that confers pleiotropic drug resistance in high copy number. Gene 101:149–152
Jamieson DJ (1992) Saccharomyces cerevisiae has distinct responses to both hydrogen peroxide and menadione. J Bacteriol 174:6678–6681
Kawahata M, Masaki K, Fujii T, lefuji H (2006) Yeast genes involved in response to lactic and acetic: acidic conditions caused by the organic acids in Saccharomyces cerevisiae cultures induce expression of intracellular metal metabolism genes regulated by Aft1p. FEMS Yeast Res 6:924–936
Kispal G, Sumegi B, Dietmeier K, Bock I, Gajdos G, Tomcsanyi T, Sandor A (1993) Cloning and sequencing of a cDNA encoding Saccharomyces cerevisiae carnitine acetyltransferase. Use of the cDNA in gene disruption studies. J Biol Chem 268:1824–1829
Kunau WH, Dommes V, Schulz H (1995) Beta-oxidation of fatty acids in mitochondria, peroxisomes, and bacteria: a century of continued progress. Prog Lipid Res 34:267–342
Lee J, Lee B, Shin D, Kwak S, Bahk JD, Lim CO, Yun D (2002) Carnitine uptake by AGP2 in yeast Saccharomyces cerevisiae is dependent on Hog1 MAP kinase pathway. Mol Cells 13:407–412
Leenders F, Tesdorpf JG, Markus M, Engel T, Seedorf U, Adamski J (1996) Porcine 80-kDa protein reveals intrinsic 17 beta-hydroxysteroid dehydrogenase, fatty acyl-CoA-hydratase/dehydrogenase, and sterol transfer activities. J Biol Chem 271:5438–5442
Maiorella B, Blanch HW, Wilke CR (1983) By-product inhibition effects on ethanolic fermentation Saccharomyces cerevisiae. Biotech Bioeng 25:103–121
Narendranath NV, Thomas KC, Ingledew WM (2001) Effects of acetic acid and lactic acid on the growth of Saccharomyces cerevisiae in minimal medium. J Ind Microbiol Biotechnol 26:171–177
Palmieri L, Lasorsa FM, De-Palma A, Palmieri F, Runswick MJ, Walker JE (1997) Identification of the yeast ACR1 gene product as a succinate–fumarate transporter essential for growth on ethanol or acetate. FEBS Lett 417:114–118
Piper P, Calderon CO, Hatzixanthis K, Mollapour M (2001) Weak acid adaptation: the stress response that confers yeasts with resistance to organic acid food preservatives. Microbiology 147:2635–2642
Ramsey RR, Zammit VA (2004) Carnitine acetyltransferases and their influence on CoA pools and disease. Mol Aspects Med 25:475–493
Reddy JK, Mannaerts GP (1994) Peroxisomal lipid metabolism. Annu Rev Nutr 14:343–370
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Schmalix W, Bandlow W (1993) The ethanol-inducible YAT1 gene from yeast encodes a presumptive mitochondrial outer carnitine acetyltransferase. J Biol Chem 268:27428–27439
Schnell N, Krems B, Entian KD (1992) The PAR1(YAP1/SNQ3) gene of Saccharomyces cerevisiae, a c-Jun homologue, is involved in oxygen metabolism. Curr Genet 21:269–273
Schulz H (1991) Beta oxidation of fatty acids. Biochim Biophys Acta 1081:109–120
Steiber A, Kerner J, Hoppel CL (2004) Carnitine: a nutritional, biosynthetic and functional perspective. Mol Aspects Med 25:455–473
Stemple CJ, Davis MA, Hynes MJ (1998) The facC gene of Aspergillus nidulans encodes an acetate-inducible carnitine acetyltransferase. J Bacteriol 180:6242–6251
Swiegers JH, Dippenaar N, Pretorius IS, Bauer FF (2001) Carnitine-dependent metabolic activities in Saccharomyces cerevisiae: three carnitine acetyltransferases are essential in a carnitine-dependent strain. Yeast 18:585–595
Van Roermund CW, Elgersma Y, Singh N, Wanders RJ, Tabak HF (1995) The membrane of peroxisomes in Saccharomyces cerevisiae is impermeable to NAD(H) and acetyl-CoA under in vivo conditions. EMBO J 14:3480–3486
Volschenk H, Viljoen M, Grobler J, Petzold B, Bauer F, Subden RE, Young RA, Lonvaud A, Denayrolles M, van-Vuuren HJ (1997) Engineering pathways for malate degradation in Saccharomyces cerevisiae. Nat Biotechnol 15:253–257
Wanders RJ, Schutgens RB, Barth PG (1995) Peroxisomal disorders: a review. J Neuropathol Exp Neurol 54:726–739
Westermann B, Neupert W (2000) Mitochondria-targeted green fluorescent proteins: convenient tools for the study of organelle biogenesis in Saccharomyces cerevisiae. Yeast 16:1421–1427
Winston F, Dollard C, Ricupero-Hovasse SL (1995) Construction of a set of convenient Saccharomyces cerevisiae strains that are isogenic to S288C. Yeast 11:53–55
Acknowledgments
This work was supported by the South African Medical Research Council, the National Research Foundation of South Africa, and Winetech.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Hohmann.
Jaco Franken and Sven Kroppenstedt (in memoriam) contributed equally to this study.
Rights and permissions
About this article
Cite this article
Franken, J., Kroppenstedt, S., Swiegers, J.H. et al. Carnitine and carnitine acetyltransferases in the yeast Saccharomyces cerevisiae: a role for carnitine in stress protection. Curr Genet 53, 347–360 (2008). https://doi.org/10.1007/s00294-008-0191-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-008-0191-0