Abstract
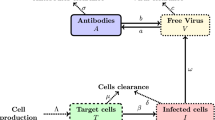
Antibodies that bind viral surface proteins can limit the spread of the infection through neutralizing and non-neutralizing functions. During both acute and chronic Human Immunodeficiency Virus infection, antibody–virion immune complexes are formed, but fail to ensure protection. In this study, we develop a mathematical model of multivalent antibody binding and use it to determine the dynamical interactions that lead to immune complexes formation and the role of complexes with increased numbers of bound antibodies in the pathogenesis of the disease. We compare our predictions with published temporal virus and immune complex data from acute infected patients. Finally, we derive quantitative and qualitative conditions needed for antibody-induced protection.







Similar content being viewed by others
References
IgG Human ELISA Kit ab100547. http://www.abcam.com/IgG-Human-ELISA-Kit-ab100547.html. Accessed 27 Aug 2013
Aasa-Chapman M, Hayman A, Newton P, Cornforth D, Williams I, Borrow P, Balfe P, McKnight A (2004) Development of the antibody response in acute HIV-1 infection. AIDS 18:371–381
Banks H, Bortz D (2005) A parameter sensitivity methodology in the context of HIV delay equation models. J Math Biol 50:607–625
Bonhoeffer S, May R, Shaw G, Nowak M (1997) Virus dynamics and drug therapy. Proc Nat Acad Sci USA 94:6971–6976
Bonsignori M, Alam S, Liao H, Verkoczy L, Tomaras G, Haynes B, Moody M (2012) HIV-1 antibodies from infection and vaccination: insights for guiding vaccine design. Trends Microbiol 20(11):532–539
Bortz D, Nelson P (2004) Sensitivity analysis of a nonlinear lumped parameter model of HIV infection dynamics. Bull Math Biol 66:1009–1026
Burrer R, Haessig-Einius S, Aubertin A, Moog C (2005) Neutralizing as well as non-neutralizing polyclonal immunoglobulin (Ig)G from infected patients capture HIV-1 via antibodies directed against the principal immunodominant domain of gp41. Virology 333:102–113
Burton D, Poignard P, Stanfield R, Wilson I (2012) Broadly neutralizing antibodies present new prospects to counter highly antigenically diverse viruses. Science 337(6091):183–186
Center R, Leapman R, Lebowitz J, Arthur L, Earl P, Moss B (2002) Oligomeric structure of the human immunodeficiency virus type 1 envelope protein on the virion surface. J Virol 76:78637867
Ciupe S, De Leenheer P, Kepler T (2011) Paradoxical suppression of poly-specific broadly neutralizing antibodies in the presence of strain-specific neutralizing antibodies following HIV infection. J Theor Biol 277(1):55–66
Dati F, Schumann G, Thomas L, Aguzzi F, Baudner S, Bienvenu J, Blaabjerg O, Blirup-Jensen S, Carlstrm A, Petersen P, Johnson A, Milford-Ward A, Ritchie R, Svendsen P, Whicher J (1996) Consensus of a group of professional societies and diagnostic companies on guidelines for interim reference ranges for 14 proteins in serum based on the standardization against the IFCC/BCR/CAP reference material (CRM 470). Eur J Clin Chem Clin Biochem 34:517–520
Dimmock N (1993) Neutralization of animal viruses. Curr Top Microbiol Immunol 183:1–149
Gonzalez-Quintela A, Alende R, Gude F, Campos J, Rey J, Meijide L, Fernandez-Merino C, Vidal C (2007) Serum levels of immunoglobulins (IgG, IgA, IgM) in a general adult population and their relationship with alcohol consumption, smoking and common metabolic abnormalities. Clin Exp Immunol 151:42–50
Gray E, Moore P, Choge I, Decker J, Bibollet-Ruche F, Li H, Leseka N, Treurnicht F, Mlisana K, Shaw G, Karim S, Williamson C, Morris L (2007) Neutralizing antibody responses in acute human immunodeficiency virus type 1 subtype C infection. J Virol 81:6187–6196
Igarashi T, Brown C, Azadegan A, Haigwood N, Dimitrov D, Martin M, Shibata R (1999) Human immunodeficiency virus type 1 neutralizing antibodies accelerate clearance of cell-free virions from blood plasma. Nat Med 5(2):211–216
Klasse P, Sattentau Q (2002) Occupancy and mechanism in antibody-mediated neutralization of animal viruses. J Gen Virol 83:2091–2108
Kuhmann S, Platt E, Kozak S, Kabat D (2000) Cooperation of multiple CCR5 coreceptors is required for infections by human immunodeficiency virus type 1. J Virol 74:7005–7015
Kwong P, Mascola J (2012) Human antibodies that neutralize HIV-1: identification, structures, and B cell ontogenies. Immunity 37(3):412–425
Lagarias J, Reeds J, Wright M, Wright P (1998) Convergence properties of the Nelder–Mead simplex method in low dimensions. SIAM J Optim 9(1):112–147
Little S, McLean A, Spina C, Richman D, Havlir D (1999) Viral dynamics of acute HIV-1 infection. J Exp Med 190:841–850
Liu J, Bartesaghi A, Borgnia M, Sapiro G, Subramaniam S (2008) Molecular architecture of native HIV-1 gp120 trimers. Nature 455:109–113
Liu P, Overman R, Yates N, Alam S, Vandergrift N, Chen Y, Graw F, Freel S, Kappes J, Ochsenbauer C, Montefiori D, Gao F, Perelson A, Cohen M, Haynes B, Tomaras G (2011) Dynamic antibody specificities and virion concentrations in circulating immune complexes in acute to chronic HIV-1 infection. J Virol 85:11196–11207
Magnus C (2013) Virus neutralisation: new insights from kinetic neutralisation curves. PLoS Comput Biol 9:e1002,900
Magnus C, Brandenberg O, Rusert P, Trkola A, Regoes R (2013) Mathematical models: a key to understanding HIV envelope interactions? J Immunol Methods (in press)
Magnus C, Regoes R (2010) Estimating the stoichiometry of HIV neutralization. PLoS Comput Biol 6(3):e1000713
Magnus C, Regoes R (2011) Restricted occupancy models for neutralization of hiv virions and populations. J Theor Biol 283:192–202
Magnus C, Regoes R (2012) Analysis of the subunit stoichiometries in viral entry. PLoS One 7(3):e33441
Magnus C, Rusert P, Bonhoeffer S, Trkola A, Regoes R (2009) Estimating the stoichiometry of human immunodeficiency virus entry. J Virol 83:1523–1531
Markowitz M, Louie M, Hurley A, Sun E, Di Mascio M, Perelson A, Ho D (2003) A novel antiviral intervention results in more accurate assessment of human immunodeficiency virus type 1 replication dynamics and T-cell decay in vivo. J Virol 77:5037–5038
McLain L, Dimmock N (1994) Single- and multi-hit kinetics of immunoglobulin g neutralization of human immunodeficiency virus type 1 by monoclonal antibodies. J Gen Virol 75:1457–1460
Moore P, Crooks E, Porter L, Zhu P, Cayanan C, Grise H, Corcoran P, Zwick M, Franti M, Morris L, Roux K, Burton D, Binley J (2006) Nature of nonfunctional envelope proteins on the surface of human immunodeficiency virus type 1. J Virol 80:2515–2528
Mouquet H, Scheid J, Zoller M, Krogsgaard M, Ott R, Shukair S, Artyomov M, Pietzsch J, Connors M, Pereyra F, Walker B, Ho D, Wilson P, Seaman M, Eisen H, Chakraborty A, Hope T, Ravetch J, Wardemann H, Nussenzweig M (2010) Polyreactivity increases the apparent affinity of anti-HIV antibodies by heteroligation. Nature 467:591–595
Mulampaka S, Dixit N (2011) Estimating the threshold surface density of Gp120–CCR5 complexes necessary for HIV-1 envelope-mediated cell-cell fusion. PLoS One 6(5):e19941
Perelson A, Nuemann A, Markowitz M, Leonard J, Ho D (1996) HIV-1 dynamics in vivo: virion clearance rate, infected cell lifespan, and viral generation time. Science 271:1582–1586
Pilgrim A, Pantaleo G, Cohen O, Fink L, Zhou J, Zhou J, Bolognesi D, Fauci A, Montefiori D (1997) Neutralizing antibody responses to human immunodeficiency virus type 1 in primary infection and long-term-nonprogressive infection. J Infect Dis 176:924–932
Platt E, Durnin J, Shinde U, Kabat D (2007) An allosteric rheostat in HIV-1 gp120 reduces CCR5 stoichiometry required for membrane fusion and overcomes diverse entry limitations. J Mol Biol 374:64–79
Plotkin S (2009) Sang Froid in a time of trouble: is a vaccine against HIV possible? J Int AIDS Soc 12:2
Ramratnam B, Bonhoeffer S, Binley J, Hurley A, Zhang L, Mittler J, Markowitz M, Moore J, Perelson A, Ho D (1999) Rapid production and clearance of HIV-1 and hepatitis C virus assessed by large volume plasma apheresis. Lancet 354:1782–1785
Ribeiro R, Qin L, Chavez L, Li D, Self S, Perelson A (2010) Estimation of the initial viral growth rate and basic reproductive number during acute HIV-1 infection. J Virol 84:6096–6102
Richman D, Wrin T, Little S, Petropoulos C (2003) Rapid evolution of the neutralizing antibody response to HIV type 1 infection. Proc Natl Acad Sci USA 100:4144–4149
Sachsenberg N, Perelson A, Yerly S, Schockmel G, Leduc D, Hirschel B, Perrin L (1998) Turnover of CD4 and CD8 T lymphocytes in HIV-1 infection as measured by Ki-67 antigen. J Exp Med 187:1295–1303
Sattentau Q (2013) Envelope glycoprotein trimers as HIV-1 vaccine immunogens. Vaccines 1:497–512
Schwesinger F, Ros R, Strunz T, Anselmetti D, Guntherodt HJ, Honegger A, Jermutus L, Tiefenauer L, Pluckthun A (2000) Unbinding forces of single antibody–antigen complexes correlate with their thermal dissociation rates. Proc Natl Acad Sci USA 97(18):9972–9977
Stafford M, Corey L, Cap Y, Daar E, Ho D, Perelson A (2000) Modeling plasma virus concentration during primary HIV infection. J Theor Biol 203:285–301
Tabei S, Li Y, Weigert M, Dinner A (2012) Model for competition from self during passive immunization, with application to broadly neutralizing antibodies for HIV. Vaccine 30:607–613
Tomaras G, Haynes B (2009) HIV-1-specific antibody responses during acute and chronic HIV-1 infection. Curr Opin HIV AIDS 4(5):373–379
Tomaras G, Haynes B (2010) Strategies for eliciting HIV-1 inhibitory antibodies. Curr Opin HIV AIDS 5(5):421–427
Tomaras G, Yates N, Liu P, Qin L, Fouda G, Chavez L, Decamp A, Parks R (2008) Initial B-cell responses to transmitted human immunodeficiency virus type 1: virion-binding immunoglobulin IgM and IgG antibodies followed by plasma anti-gp41 antibodies with ineffective control of initial viremia. J Virol 82(24):12449–12463
Tran E, Borgnia M, Kuybeda O, Schauder D, Bartesaghi A, Frank G, Sapiro G, Milne J, Subramaniam S (2012) Structural mechanism of trimeric HIV-1 envelope glyco protein activation. PLoS Pathog 8:e1002,797
Wei X, Decker J, Wang S, Hui H, Kappes J, Wu X, Salazar-Gonzalez J, Salazar M, Kilby J, Saag M, Komarova K, Nowak M, Hahn B, Kwong P, Shaw G (2003) Antibody neutralization and escape by HIV-1. Nature 422:307–312
Yang X, Kurteva S, Lee S, Sodroski J (2005) Stoichiometry of antibody neutralization of human immunodeficiency virus type 1. J Virol 79(6):3500–3508
Zalevsky J, Chamberlain A, Horton H, Karki S, Leung I, Sproule T, Lazar G, Roopenian D, Desjarlais J (2010) Enhanced antibody half-life improves in vivo activity. Nat Biotechnol 28:157–159
Zhou T, Xu L, Dey B, Hessell A, Van Ryk D, Xiang SH, Yang X, Zhang MY, Zwick M, Arthos J, Burton D, Dimitrov D, Sodroski J, Wyatt R, Nabel G, Kwong P (2007) Structural definition of a conserved neutralization epitope on HIV-1 gp120. Nature 445:732–737
Zhu P, Liu J, Bess J Jr et al (2006) Distribution and three-dimensional structure of AIDS virus envelope spikes. Nature 441:847–852
Zhu P, Winkler H, Chertova E, Taylor K, Roux K (2008) Cryoelectron tomography of HIV-1 envelope spikes: further evidence for tripod-like legs. PLoS Pathog 4:e1000,203
Acknowledgments
We gratefully acknowledge the help of Nicholas Sorrenson and support from NSF Grant DMS-1214582. We want to thank the reviewers for their comments and suggestions, which helped improve the paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ciupe, S.M. Mathematical model of multivalent virus–antibody complex formation in humans following acute and chronic HIV infections. J. Math. Biol. 71, 513–532 (2015). https://doi.org/10.1007/s00285-014-0826-3
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-014-0826-3