Abstract
Purpose
The poor outcomes in glioblastoma necessitate new therapeutic target. Isoprenylcysteine carboxyl methyltransferase (ICMT), a unique enzyme of the final step of prenylation that modifies activities of oncogenic proteins, represents a promising target for many cancers.
Methods
Expression pattern, function and downstream pathway of ICMT in glioblastoma were analyzed using immunohistochemistry, ELISA, cellular assays and immunoblotting method. Combinatory effect was analyzed using Chou-Talalay approach.
Results
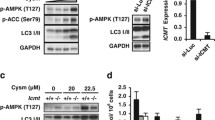
Upregulation of ICMT expression is a common phenomenon in glioblastoma patients regardless of clinicopathological characteristics. Gain-of-function and loss-of-function analysis support the role of ICMT in glioblastoma growth and survival but not migration. Importantly, pharmacological inhibitors of ICMT are effectively against glioblastoma cells while sparing normal neuron cells, and furthermore that they act synergistically with chemotherapeutic drugs. Consistently, ICMT inhibitor UCM-1336 significantly inhibits glioblastoma growth without causing toxicity in mice. Mechanistic studies demonstrate that Ras/Raf/Mek/Erk rather than Ras/PI3K/Akt/mTOR is the downstream pathway of ICMT-mediated glioblastoma growth.
Conclusions
Our findings provide the proof-of-concept of pharmacologically targeting ICMT in the treatment of glioblastoma via deactivation of Ras/Raf/Mek/Erk.








Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.
References
Batash R, Asna N, Schaffer P, Francis N, Schaffer M (2017) Glioblastoma multiforme, diagnosis and treatment recent literature review. Curr Med Chem 24(27):3002–3009. https://doi.org/10.2174/0929867324666170516123206
Le Rhun E, Preusser M, Roth P, Reardon DA, van den Bent M, Wen P, Reifenberger G, Weller M (2019) Molecular targeted therapy of glioblastoma. Cancer Treat Rev 80:101896. https://doi.org/10.1016/j.ctrv.2019.101896
Palsuledesai CC, Distefano MD (2015) Protein prenylation: enzymes, therapeutics, and biotechnology applications. ACS Chem Biol 10(1):51–62. https://doi.org/10.1021/cb500791f
Winter-Vann AM, Casey PJ (2005) Post-prenylation-processing enzymes as new targets in oncogenesis. Nat Rev Cancer 5(5):405–412. https://doi.org/10.1038/nrc1612
Lau HY, Tang J, Casey PJ, Wang M (2017) Isoprenylcysteine carboxyl methyltransferase is critical for malignant transformation and tumor maintenance by all RAS isoforms. Oncogene 36(27):3934–3942. https://doi.org/10.1038/onc.2016.508
Chai TF, Manu KA, Casey PJ, Wang M (2020) Isoprenylcysteine carboxyl methyltransferase is required for the impact of mutant KRAS on TAZ protein level and cancer cell self-renewal. Oncogene 39(31):5373–5389. https://doi.org/10.1038/s41388-020-1364-7
Zhu Y, Hu Q, Li H (2019) Isoprenylcysteine carboxyl methyltransferase is associated with nasopharyngeal carcinoma chemoresistance and Ras activation. Biochem Biophys Res Commun 516(3):784–789. https://doi.org/10.1016/j.bbrc.2019.06.074
Marin-Ramos NI, Balabasquer M, Ortega-Nogales FJ, Torrecillas IR, Gil-Ordonez A, Marcos-Ramiro B, Aguilar-Garrido P, Cushman I, Romero A, Medrano FJ, Gajate C, Mollinedo F, Philips MR, Campillo M, Gallardo M, Martin-Fontecha M, Lopez-Rodriguez ML, Ortega-Gutierrez S (2019) A Potent isoprenylcysteine carboxyl methyltransferase (ICMT) inhibitor improves survival in Ras-driven acute myeloid leukemia. J Med Chem 62(13):6035–6046. https://doi.org/10.1021/acs.jmedchem.9b00145
Pan Q, Liu R, Banu H, Ma L, Li H (2018) Inhibition of isoprenylcysteine carboxyl methyltransferase sensitizes common chemotherapies in cervical cancer via ras-dependent pathway. Biomed Pharmacother 99:169–175. https://doi.org/10.1016/j.biopha.2018.01.048
Liu Q, Chen J, Fu B, Dai J, Zhao Y, Lai L (2018) Isoprenylcysteine carboxyl methyltransferase regulates ovarian cancer cell response to chemotherapy and Ras activation. Biochem Biophys Res Commun 501(2):556–562. https://doi.org/10.1016/j.bbrc.2018.05.038
Teh JT, Zhu WL, Ilkayeva OR, Li Y, Gooding J, Casey PJ, Summers SA, Newgard CB, Wang M (2015) Isoprenylcysteine carboxyl methyltransferase regulates mitochondrial respiration and cancer cell metabolism. Oncogene 34(25):3296–3304. https://doi.org/10.1038/onc.2014.260
Xu J, Zhu Y, Wang F, Zhou Y, Xia G, Xu W (2019) ICMT contributes to hepatocellular carcinoma growth, survival, migration and chemoresistance via multiple oncogenic pathways. Biochem Biophys Res Commun 518(3):584–589. https://doi.org/10.1016/j.bbrc.2019.08.094
Magkouta S, Pappas A, Moschos C, Vazakidou ME, Psarra K, Kalomenidis I (2016) Icmt inhibition exerts anti-angiogenic and anti-hyperpermeability activities impeding malignant pleural effusion. Oncotarget 7(15):20249–20259. https://doi.org/10.18632/oncotarget.7912
Lau HY, Ramanujulu PM, Guo D, Yang T, Wirawan M, Casey PJ, Go ML, Wang M (2014) An improved isoprenylcysteine carboxyl methyltransferase inhibitor induces cancer cell death and attenuates tumor growth in vivo. Cancer Biol Ther 15(9):1280–1291. https://doi.org/10.4161/cbt.29692
Ramanujulu PM, Yang T, Yap SQ, Wong FC, Casey PJ, Wang M, Go ML (2013) Functionalized indoleamines as potent, drug-like inhibitors of isoprenylcysteine carboxyl methyltransferase (ICMT). Eur J Med Chem 63:378–386. https://doi.org/10.1016/j.ejmech.2013.02.007S0223-5234(13)00093-7[pii]
Bergman JA, Hahne K, Song J, Hrycyna CA, Gibbs RA (2012) S-farnesyl-thiopropionic acid (FTPA) triazoles as potent inhibitors of isoprenylcysteine carboxyl methyltransferase. ACS Med Chem Lett 3(1):15–19. https://doi.org/10.1021/ml200106d
Chou TC (2010) Drug combination studies and their synergy quantification using the Chou-Talalay method. Cancer Res 70(2):440–446. https://doi.org/10.1158/0008-5472.CAN-09-1947
Yadav B, Wennerberg K, Aittokallio T, Tang J (2015) Searching for drug synergy in complex dose-response landscapes using an interaction potency model. Comput Struct Biotechnol J 13:504–513. https://doi.org/10.1016/j.csbj.2015.09.001
Wan W, Zhang X, Huang C, Chen L, Yang X, Bao K, Peng T (2020) Monensin inhibits glioblastoma angiogenesis via targeting multiple growth factor receptor signaling. Biochem Biophys Res Commun 530(2):479–484. https://doi.org/10.1016/j.bbrc.2020.05.057
Winter-Vann AM, Baron RA, Wong W, Dela Cruz J, York JD, Gooden DM, Bergo MO, Young SG, Toone EJ, Casey PJ (2005) A small-molecule inhibitor of isoprenylcysteine carboxyl methyltransferase with antitumor activity in cancer cells. Proc Natl Acad Sci USA 102(12):4336–4341
Ou A, Yung WKA, Majd N (2020) Molecular Mechanisms of Treatment resistance in glioblastoma. Int J Mol Sci. https://doi.org/10.3390/ijms22010351
Kolch W (2000) Meaningful relationships: the regulation of the Ras/Raf/MEK/ERK pathway by protein interactions. Biochem J 351(Pt 2):289–305
Rodriguez-Viciana P, Warne PH, Dhand R, Vanhaesebroeck B, Gout I, Fry MJ, Waterfield MD, Downward J (1994) Phosphatidylinositol-3-OH kinase as a direct target of Ras. Nature 370(6490):527–532. https://doi.org/10.1038/370527a0
Tang Z, Li C, Kang B, Gao G, Li C, Zhang Z (2017) GEPIA: a web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res 45(W1):W98–W102. https://doi.org/10.1093/nar/gkx247
Borini Etichetti C, Di Benedetto C, Rossi C, Baglioni MV, Bicciato S, Del Sal G, Menacho-Marquez M, Girardini J (2019) Isoprenylcysteine carboxy methyltransferase (ICMT) is associated with tumor aggressiveness and its expression is controlled by the p53 tumor suppressor. J Biol Chem 294(13):5060–5073. https://doi.org/10.1074/jbc.RA118.006037
Cushman I, Casey PJ (2009) Role of isoprenylcysteine carboxyl methyltransferase-catalyzed methylation in Rho function and migration. J Biol Chem 284(41):27964–27973. https://doi.org/10.1074/jbc.M109.025296
Svilar D, Dyavaiah M, Brown AR, Tang JB, Li J, McDonald PR, Shun TY, Braganza A, Wang XH, Maniar S, St Croix CM, Lazo JS, Pollack IF, Begley TJ, Sobol RW (2012) Alkylation sensitivity screens reveal a conserved cross-species functionome. Mol Cancer Res 10(12):1580–1596. https://doi.org/10.1158/1541-7786.MCR-12-0168
Prior IA, Lewis PD, Mattos C (2012) A comprehensive survey of Ras mutations in cancer. Cancer Res 72(10):2457–2467. https://doi.org/10.1158/0008-5472.CAN-11-2612
Knobbe CB, Reifenberger J, Reifenberger G (2004) Mutation analysis of the Ras pathway genes NRAS, HRAS, KRAS and BRAF in glioblastomas. Acta Neuropathol 108(6):467–470. https://doi.org/10.1007/s00401-004-0929-9
Holmen SL, Williams BO (2005) Essential role for Ras signaling in glioblastoma maintenance. Cancer Res 65(18):8250–8255. https://doi.org/10.1158/0008-5472.CAN-05-1173
Stephen AG, Esposito D, Bagni RK, McCormick F (2014) Dragging ras back in the ring. Cancer Cell 25(3):272–281. https://doi.org/10.1016/j.ccr.2014.02.017
Funding
This work was supported by Science and Technology Innovation seedling project of Sichuan (2016065).
Author information
Authors and Affiliations
Contributions
Contribution to study design and its implementation: WFW, WFX, WP, JGX; Data analysis and interpretation: WFW, WFX, WP, LGC, ZYL, JGX; Manuscript writing and editing: WFW, JGX. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflict of interest.
Ethical approval
All experimental protocols were approved by the institutional review board of Southwest Medical University. This study was conducted in accordance with the Declaration of Helsinki. All methods were carried out in accordance with relevant guidelines and regulations.
Informed consent
Informed consent was obtained from all subjects.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wan, W., Xiao, W., Pan, W. et al. Isoprenylcysteine carboxyl methyltransferase is critical for glioblastoma growth and survival by activating Ras/Raf/Mek/Erk. Cancer Chemother Pharmacol 89, 401–411 (2022). https://doi.org/10.1007/s00280-022-04401-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-022-04401-x