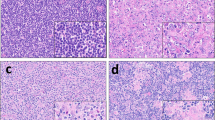
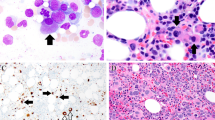
Abstract
Acute leukemias of ambiguous lineage represent a heterogeneous group of rare, poorly characterized leukemias with adverse outcome. No larger studies have yet performed a combined approach of molecular and clinical characterization of acute undifferentiated leukemia (AUL) and biphenotypic acute leukemia (BAL) in adults. Here we describe 16 adults with AUL and 26 with BAL and performed mutational as well as expression studies of genes with prognostic impact in acute leukemia (BAALC, ERG, MN1, WT1, and IGFBP7). AUL showed overexpression of these genes compared to T-lymphoblastic leukemia (T-ALL), B-precursor ALL, and to acute myeloid leukemia (AML). Genotype alterations were not detectable in AUL. BAL samples were characterized by frequent WT1 mutations (18 %) and BCR-ABL translocations (30 %). ALL-based treatment protocols induced complete remissions in 40 % and AML-like therapies in 22 % of AUL/BAL patients. The outcome in both groups was very poor; a long-term survival was only observed in patients undergoing allogeneic stem cell transplantation (SCT). Our findings indicate that AUL and BAL share important molecular and high-risk features of both myeloid and lymphoid leukemias. BAL patients exhibited genetic alterations, which can be targeted therapeutically. Importantly, ALL therapy might be more effective than AML protocols and AUL/BAL patients should be considered for allogeneic SCT.


Similar content being viewed by others
References
Bene MC, Castoldi G, Knapp W, Ludwig WD, Matutes E, Orfao A, Van't Veer MB (1995) Proposals for the immunological classification of acute leukemias. European Group for the Immunological Characterization of Leukemias (EGIL). Leukemia 9:1783–1786
Bene MC, Bernier M, Casasnovas RO, Castoldi G, Knapp W, Lanza F, Ludwig WD, Matutes E, Orfao A, Sperling C, Van't Veer MB (1998) The reliability and specificity of c-kit for the diagnosis of acute myeloid leukemias and undifferentiated leukemias. The European Group for the Immunological Classification of Leukemias (EGIL). Blood 92:596–599
Vardiman JW, Thiele J, Arber DA, Brunning RD, Borowitz MJ, Porwit A, Harris NL, Le Beau MM, Hellstrom-Lindberg E, Tefferi A, Bloomfield CD (2009) The 2008 revision of the World Health Organization (WHO) classification of myeloid neoplasms and acute leukemia: rationale and important changes. Blood 114:937–951. doi:10.1182/blood-2009-03-209262
Borowitz MJ, Béné M-C, Harris NL, Porwit A, Matutes E (2009) WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. WHO Classification of Tumours, Volume 2, Chapter 7: 150 pp
Bene MC, Porwit A (2012) Acute leukemias of ambiguous lineage. Semin Diagn Pathol 29:12–18
Zhang Y, Wu D, Sun A, Qiu H, He G, Jin Z, Tang X, Miao M, Fu Z, Han Y (2011) Clinical characteristics, biological profile, and outcome of biphenotypic acute leukemia: a case series. Acta Haematol 125:210–218. doi:10.1159/000322594
Matutes E, Pickl WF, Van't Veer M, Morilla R, Swansbury J, Strobl H, Attarbaschi A, Hopfinger G, Ashley S, Bene MC, Porwit A, Orfao A, Lemez P, Schabath R, Ludwig WD (2011) Mixed-phenotype acute leukemia: clinical and laboratory features and outcome in 100 patients defined according to the WHO 2008 classification. Blood 117. doi:10.1182/blood-2010-10-314682
Legrand O, Perrot JY, Simonin G, Baudard M, Cadiou M, Blanc C, Ramond S, Viguie F, Marie JP, Zittoun R (1998) Adult biphenotypic acute leukaemia: an entity with poor prognosis which is related to unfavourable cytogenetics and P-glycoprotein over-expression. Br J Haematol 100:147–155
Killick S, Matutes E, Powles RL, Hamblin M, Swansbury J, Treleaven JG, Zomas A, Atra A, Catovsky D (1999) Outcome of biphenotypic acute leukemia. Haematologica 84:699–706
Rubio MT, Dhedin N, Boucheix C, Bourhis JH, Reman O, Boiron JM, Gallo JH, Lheritier V, Thomas X, Fiere D, Vernant JP (2003) Adult T-biphenotypic acute leukaemia: clinical and biological features and outcome. Br J Haematol 123:842–849
Tiribelli M, Damiani D, Masolini P, Candoni A, Calistri E, Fanin R (2004) Biological and clinical features of T-biphenotypic acute leukaemia: report from a single centre. Br J Haematol 125:814–815
Aribi A, Bueso-Ramos C, Estey E, Estrov Z, O'Brien S, Giles F, Faderl S, Thomas D, Kebriaei P, Garcia-Manero G, Pierce S, Cortes J, Kantarjian H, Ravandi F (2007) Biphenotypic acute leukaemia: a case series. Br J Haematol 138:213–216
Xu XQ, Wang JM, Lu SQ, Chen L, Yang JM, Zhang WP, Song XM, Hou J, Ni X, Qiu HY (2009) Clinical and biological characteristics of adult biphenotypic acute leukemia in comparison with that of acute myeloid leukemia and acute lymphoblastic leukemia: a case series of a Chinese population. Haematologica 94:919–927. doi:10.3324/haematol.2008.003202
Zheng C, Wu J, Liu X, Ding K, Cai X, Zhu W (2009) What is the optimal treatment for biphenotypic acute leukemia? Haematologica 94:1778–1780. doi:10.3324/haematol.2009.014829
Owaidah TM, Al BA, Iqbal MA, Elkum N, Roberts GT (2006) Cytogenetics, molecular and ultrastructural characteristics of biphenotypic acute leukemia identified by the EGIL scoring system. Leukemia 20:620–626
Lee JH, Min YH, Chung CW, Kim BK, Yoon HJ, Jo DY, Shin HJ, Bang SM, Won JH, Zang DY, Kim HJ, Chi HS, Lee KH, Cheong JW, Kim JS, Kim SH, Park S, Park SY, Chung JS, Lee JH, Park CJ (2008) Prognostic implications of the immunophenotype in biphenotypic acute leukemia. Leuk Lymphoma 49:700–709. doi:10.1080/10428190701843247
Rubnitz JE, Onciu M, Pounds S, Shurtleff S, Cao X, Raimondi SC, Behm FG, Campana D, Razzouk BI, Ribeiro RC, Downing JR, Pui CH (2009) Acute mixed lineage leukemia in children: the experience of St Jude Children's Research Hospital. Blood 113:5083–5089. doi:10.1182/blood-2008-10-187351
Brito-Babapulle F, Pullon H, Layton DM, Etches A, Huxtable A, Mangi M, Bellingham AJ, Mufti GJ (1990) Clinicopathological features of acute undifferentiated leukaemia with a stem cell phenotype. Br J Haematol 76:210–214
Bassan R, Biondi A, Benvestito S, Tini ML, Abbate M, Viero P, Barbui T, Rambaldi A (1992) Acute undifferentiated leukemia with CD7+ and CD13+ immunophenotype. Lack of molecular lineage commitment and association with poor prognostic features. Cancer 69:396–404
Cuneo A, Ferrant A, Michaux JL, Bosly A, Chatelain B, Stul M, Dal CP, Dierlamm J, Cassiman JJ, Hossfeld DK, Castoldi G, Van den Berghe H (1996) Cytogenetic and clinicobiological features of acute leukemia with stem cell phenotype: study of nine cases. Cancer Genet Cytogenet 92:31–36
Heesch S, Goekbuget N, Stroux A, Tanchez JO, Schlee C, Burmeister T, Schwartz S, Blau O, Keilholz U, Busse A, Hoelzer D, Thiel E, Hofmann WK, Baldus CD (2010) Prognostic implications of mutations and expression of the Wilms tumor 1 (WT1) gene in adult acute T-lymphoblastic leukemia. Haematologica 95:942–949. doi:10.3324/haematol.2009.016386
Heesch S, Schlee C, Neumann M, Stroux A, Kuhnl A, Schwartz S, Haferlach T, Goekbuget N, Hoelzer D, Thiel E, Hofmann WK, Baldus CD (2010) BAALC-associated gene expression profiles define IGFBP7 as a novel molecular marker in acute leukemia. Leukemia 24:1429–1436. doi:10.1038/leu.2010.130
Kuhnl A, Gokbuget N, Stroux A, Burmeister T, Neumann M, Heesch S, Haferlach T, Hoelzer D, Hofmann WK, Thiel E, Baldus CD (2010) High BAALC expression predicts chemoresistance in adult B-precursor acute lymphoblastic leukemia. Blood 115:3737–3744. doi:10.1182/blood-2009-09-241943
Kuhnl A, Kaiser M, Neumann M, Fransecky L, Heesch S, Radmacher M, Marcucci G, Bloomfield CD, Hofmann WK, Thiel E, Baldus CD (2011) High expression of IGFBP2 is associated with chemoresistance in adult acute myeloid leukemia. Leuk Res 35:1585–1590. doi:10.1016/j.leukres.2011.08.006
Schwartz S, Rieder H, Schlager B, Burmeister T, Fischer L, Thiel E (2003) Expression of the human homologue of rat NG2 in adult acute lymphoblastic leukemia: close association with MLL rearrangement and a CD10(−)/CD24(−)/CD65s(+)/CD15(+) B-cell phenotype. Leukemia 17:1589–1595
Baldus CD, Martus P, Burmeister T, Schwartz S, Gokbuget N, Bloomfield CD, Hoelzer D, Thiel E, Hofmann WK (2007) Low ERG and BAALC expression identifies a new subgroup of adult acute T-lymphoblastic leukemia with a highly favorable outcome. J Clin Oncol 25:3739–3745. doi:10.1200/JCO.2007.11.5253
Heuser M, Beutel G, Krauter J, Dohner K, von Neuhoff N, Schlegelberger B, Ganser A (2006) High meningioma 1 (MN1) expression as a predictor for poor outcome in acute myeloid leukemia with normal cytogenetics. Blood 108:3898–3905. doi:10.1182/blood-2006-04-014845
Burmeister T, Meyer C, Schwartz S, Hofmann J, Molkentin M, Kowarz E, Schneider B, Raff T, Reinhardt R, Gokbuget N, Hoelzer D, Thiel E, Marschalek R (2009) The MLL recombinome of adult CD10-negative B-cell precursor acute lymphoblastic leukemia: results from the GMALL study group. Blood 113:4011–4015
Burmeister T, Reinhardt R (2008) A multiplex PCR for improved detection of typical and atypical BCR-ABL fusion transcripts. Leuk Res 32:579–585
Langer C, Radmacher MD, Ruppert AS, Whitman SP, Paschka P, Mrozek K, Baldus CD, Vukosavljevic T, Liu CG, Ross ME, Powell BL, de la Chapelle A, Kolitz JE, Larson RA, Marcucci G, Bloomfield CD (2008) High BAALC expression associates with other molecular prognostic markers, poor outcome, and a distinct gene-expression signature in cytogenetically normal patients younger than 60 years with acute myeloid leukemia: a Cancer and Leukemia Group B (CALGB) study. Blood 111:5371–5379
Marcucci G, Baldus CD, Ruppert AS, Radmacher MD, Mrozek K, Whitman SP, Kolitz JE, Edwards CG, Vardiman JW, Powell BL, Baer MR, Moore JO, Perrotti D, Caligiuri MA, Carroll AJ, Larson RA, de la Chapelle A, Bloomfield CD (2005) Overexpression of the ETS-related gene, ERG, predicts a worse outcome in acute myeloid leukemia with normal karyotype: a Cancer and Leukemia Group B study. J Clin Oncol 23:9234–9242. doi:10.1200/JCO.2005.03.6137
Baldus CD, Burmeister T, Martus P, Schwartz S, Gokbuget N, Bloomfield CD, Hoelzer D, Thiel E, Hofmann WK (2006) High expression of the ETS transcription factor ERG predicts adverse outcome in acute T-lymphoblastic leukemia in adults. J Clin Oncol 24:4714–4720. doi:10.1200/JCO.2006.06.1580
Grosveld GC (2007) MN1, a novel player in human AML. Blood Cells Mol Dis 39:336–339
Menssen HD, Renkl HJ, Rodeck U, Maurer J, Notter M, Schwartz S, Reinhardt R, Thiel E (1995) Presence of Wilms' tumor gene (wt1) transcripts and the WT1 nuclear protein in the majority of human acute leukemias. Leukemia 9:1060–1067
Haferlach C, Kern W, Schindela S, Kohlmann A, Alpermann T, Schnittger S, Haferlach T (2011) Gene expression of BAALC, CDKN1B, ERG, and MN1 adds independent prognostic information to cytogenetics and molecular mutations in adult acute myeloid leukemia. Genes Chromosomes Cancer. doi:10.1002/gcc.20950
LeMaistre A, Childs CC, Hirsch-Ginsberg C, Reuben J, Cork A, Trujillo JM, Andersson B, McCredie KB, Freireich E, Stass SA (1988) Heterogeneity in acute undifferentiated leukemia. Hematol Pathol 2:79–90
Von LM, Breems D, Van BS, Adriaansen H, Grosveld G (1992) Characterization of the translocation breakpoint sequences of two DEK-CAN fusion genes present in t(6;9) acute myeloid leukemia and a SET-CAN fusion gene found in a case of acute undifferentiated leukemia. Genes Chromosomes Cancer 5:227–234
Kim J, Lee SG, Song J, Kim SJ, Rha SY, Lee KA, Park TS, Choi JR (2010) Molecular characterization of alternative SET-NUP214 fusion transcripts in a case of acute undifferentiated leukemia. Cancer Genet Cytogenet 201:73–80. doi:10.1016/j.cancergencyto.2010.05.010
Carbonell F, Swansbury J, Min T, Matutes E, Farahat N, Buccheri V, Morilla R, Secker-Walker L, Catovsky D (1996) Cytogenetic findings in acute biphenotypic leukaemia. Leukemia 10:1283–1287
Daley GQ, Van Etten RA, Baltimore D (1990) Induction of chronic myelogenous leukemia in mice by the P210bcr/abl gene of the Philadelphia chromosome. Science 247:824–830
Burmeister T, Schwartz S, Bartram CR, Gokbuget N, Hoelzer D, Thiel E (2008) Patients' age and BCR-ABL frequency in adult B-precursor ALL: a retrospective analysis from the GMALL study group. Blood 112:918–919. doi:10.1182/blood-2008-04-149286
Matsumoto Y, Taki T, Fujimoto Y, Taniguchi K, Shimizu D, Shimura K, Uchiyama H, Kuroda J, Nomura K, Inaba T, Shimazaki C, Horiike S, Taniwaki M (2009) Monosomies 7p and 12p and FLT3 internal tandem duplication: possible markers for diagnosis of T/myeloid biphenotypic acute leukemia and its clonal evolution. Int J Hematol 89:352–358. doi:10.1007/s12185-009-0268-7
Thiede C, Steudel C, Mohr B, Schaich M, Schakel U, Platzbecker U, Wermke M, Bornhauser M, Ritter M, Neubauer A, Ehninger G, Illmer T (2002) Analysis of FLT3-activating mutations in 979 patients with acute myelogenous leukemia: association with FAB subtypes and identification of subgroups with poor prognosis. Blood 99:4326–4335
Neumann M, Heesch S, Gokbuget N, Schwartz S, Schlee C, Benlasfer O, Farhadi-Sartangi N, Thibaut J, Burmeister T, Hoelzer D, Hofmann WK, Thiel E, Baldus CD (2012) Clinical and molecular characterization of early T-cell precursor leukemia: a high-risk subgroup in adult T-ALL with a high frequency of FLT3 mutations. Blood Cancer J 2:e55
King-Underwood L, Pritchard-Jones K (1998) Wilms' tumor (WT1) gene mutations occur mainly in acute myeloid leukemia and may confer drug resistance. Blood 91:2961–2968
Summers K, Stevens J, Kakkas I, Smith M, Smith LL, Macdougall F, Cavenagh J, Bonnet D, Young BD, Lister TA, Fitzgibbon J (2007) Wilms' tumour 1 mutations are associated with FLT3-ITD and failure of standard induction chemotherapy in patients with normal karyotype AML. Leukemia 21:550–551. doi:10.1038/sj.leu.2404514
Paschka P, Marcucci G, Ruppert AS, Whitman SP, Mrozek K, Maharry K, Langer C, Baldus CD, Zhao W, Powell BL, Baer MR, Carroll AJ, Caligiuri MA, Kolitz JE, Larson RA, Bloomfield CD (2008) Wilms' tumor 1 gene mutations independently predict poor outcome in adults with cytogenetically normal acute myeloid leukemia: a cancer and leukemia group B study. J Clin Oncol 26:4595–4602. doi:10.1200/JCO.2007.15.2058
Virappane P, Gale R, Hills R, Kakkas I, Summers K, Stevens J, Allen C, Green C, Quentmeier H, Drexler H, Burnett A, Linch D, Bonnet D, Lister TA, Fitzgibbon J (2008) Mutation of the Wilms' tumor 1 gene is a poor prognostic factor associated with chemotherapy resistance in normal karyotype acute myeloid leukemia: the United Kingdom Medical Research Council Adult Leukaemia Working Party. J Clin Oncol 26:5429–5435. doi:10.1200/JCO.2008.16.0333
Tosello V, Mansour MR, Barnes K, Paganin M, Sulis ML, Jenkinson S, Allen CG, Gale RE, Linch DC, Palomero T, Real P, Murty V, Yao X, Richards SM, Goldstone A, Rowe J, Basso G, Wiernik PH, Paietta E, Pieters R, Horstmann M, Meijerink JP, Ferrando AA (2009) WT1 mutations in T-ALL. Blood 114:1038–1045. doi:10.1182/blood-2008-12-192039
Keilholz U, Letsch A, Busse A, Asemissen AM, Bauer S, Blau IW, Hofmann WK, Uharek L, Thiel E, Scheibenbogen C (2009) A clinical and immunologic phase 2 trial of Wilms tumor gene product 1 (WT1) peptide vaccination in patients with AML and MDS. Blood 113:6541–6548. doi:10.1182/blood-2009-02-202598
Acknowledgments
We want to thank Liliana Mochmann for critical reading of the manuscript and Ouidad Benlasfer for excellent technical assistance.
The authors thank the following institutions for kindly providing clinical data: Aachen: Medizinische Klinik IV—Hämatologie und Onkologie (Prof. Dr. med. Tim H. Brümmendorf); Bad Saarow: Klinik für Innere Medizin III Hämatologie, Onkologie und Palliativmedizin—Sarkomzentrum Berlin-Brandenburg (PD Dr. med. Peter Reichardt); Bonn: Medizinische Klinik III für Hämatologie und Onkologie Universitätsklinikum Bonn (Prof. Dr. med. Peter Brossart); Bremen: Klinikum Bremen-Mitte gGmbH Medizinische Klinik I (Prof. Dr. med. Bernd Hertenstein); Cottbus: Carl-Thiem-Klinikum Cottbus, II. Medizinische Klinik (Prof. Dr. med. Hjalmar B. Steinhauer); Duisburg: Med. Klinik II St. Johannes-Hospital (Prof. Dr. med. C. Aul); Düsseldorf: Universitätsklinikum Düsseldorf Klinik für Hämatologie und Onkologie (Prof. Dr. med. Rainer Haas); Essen: Evangelisches Krankenhaus Essen-Werden gGmbH Klinik für Hämatologie, Onkologie und Stammzelltransplantation (PD Dr. med. Peter Reimer); Essen: Zentrum f. Innere Medizin Med. Klinik u. Poliklinik Abt. f. Hämatologie Universität Essen (Prof. Dr. med. Ulrich Dührsen); Frankfurt (Oder): Medizinische Klinik I (Prof. Dr. med. Michael Kiehl); Freiburg: Medizinische Universitätsklinik Abt. Innere Medizin I (Prof. Dr. Dr. h.c. R. Mertelsmann); Göttingen: Medizinische Universitäts-Klinik Abteilung für Hämatologie/Onkologie (Prof. Dr. med. Lorenz Trümper); Hagen: Kath. Krankenhaus Hagen gem. GmbH St.-Marien-Hospital Klinik für Hämatologie und Onkologie (Dr. med. Hans-Walter Lindemann); Hamburg: Asklepios Klinik St. Georg Hämatologische Abteilung (Prof. Dr. med. N. Schmitz); Hamburg: Asklepios Klinik Altona II. Medizinische Abteilung (Dr. med. D. Braumann); Hamm: Med. Klinik Abteilung für Hämatologie-Onkologie Evangelisches Krankenhaus Hamm (Prof. Dr. med. Jörg Schubert); Homburg/Saar: Universitätsklinikum des Saarlandes Klinik für Innere Medizin I—Onkologie, Hämatologie (Prof. Dr. med. Michael Pfreundschuh); Jena: Universitätsklinikum Jena Klinik für Innere Medizin II Abteilung Hämatologie und Internistische Onkologie (Prof. Dr. med. Andreas Hochhaus); Kaiserslautern: Medizinische Klinik I, Westpfalz-Klinikum GmbH, Standort I Kaiserslautern (Prof. Dr. med. Hartmut Link); Karlsruhe: Städt. Klinikum Karlsruhe, Medizinische Klinik III, Schwerpunkt Onkologie, Hämatologie (Prof. Dr. med. Martin Bentz); Kiel: Universitätsklinikum Schleswig-Holstein Campus Kiel, II. Med. Klinik u. Poliklinik (Prof. Dr. Dr. M. Kneba/M. Brüggemann); Magdeburg: Universitätsklinikum Magdeburg A.ö.R Zentrum für Innere Medizin Klinik für Hämatologie/Onkologie (Prof. Dr. med. Th. Fischer); Mainz: III. Medizinische Klinik und Poliklinik Universitätsmedizin der Johannes Gutenberg-Universität (Prof. Dr. med. Matthias Theobald); Meschede: St. Walburga-Krankenhaus Meschede GmbH (PD Dr. med. M. Schwonzen); Minden: Klinikum Minden Abt. Hämatologie/Onkologie (Prof. Dr. med. M. Griesshammer); Nürnberg: Klinikum Nürnberg Nord Medizinische Klinik 5 (Prof. Dr. med. M. Wilhelm); Oldenburg: Klinikum Oldenburg Innere Medizin II (Prof. Dr. med. C.-H. Köhne); Potsdam: Klinikum Ernst von Bergmann Medizinische Klinik (Prof. Dr. med. G. Maschmeyer); Stuttgart: Robert Bosch-Krankenhaus Abt. Hämatologie/Onkologie (Prof. Dr. med. W. Aulitzky); Tübingen: Medizinische Klinik und Poliklinik Abteilung 2 Hämatologie, Onkologie, Immunologie und Rheumatologie Ambulanz (Prof. Dr. med. L. Kanz); Ulm: Medizinische Universitätsklinik Innere Medizin III (Prof. Dr. med. H. Döhner); Wiesbaden: Horst-Schmidt-Kliniken, Innere Medizin III, Hämatologie/Onkologie (Prof. Dr. med. Norbert Frickhofen); Wuppertal: HELIOS Klinikum Wuppertal Med. Klinik 1 (PD Dr. med. A. Raghavachar)
Funding
This study was supported by research funding from the Gutermuth Stiftung and the Deutsche Krebshilfe to C.D.B.
Conflict of interest disclosure
The authors declare no competing financial interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Sandra Heesch and Martin Neumann contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 200 kb)
Rights and permissions
About this article
Cite this article
Heesch, S., Neumann, M., Schwartz, S. et al. Acute leukemias of ambiguous lineage in adults: molecular and clinical characterization. Ann Hematol 92, 747–758 (2013). https://doi.org/10.1007/s00277-013-1694-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-013-1694-4