Abstract
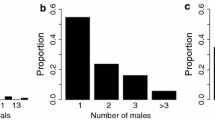
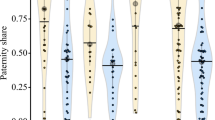
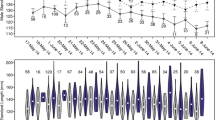
Determining the patterns and causes of variation in reproductive success is key to understanding mating systems and sexual selection, but they are challenging to study in cryptobenthic coral reef fishes. Here, we investigate characteristics of breeding habitat, the genetic mating system, and correlates of male mating success in the neon goby Elacatinus lori. This fish is primarily found living and breeding within the yellow tube sponge Aplysina fistularis in Belize — it has become a focus of marine population ecology research, but knowledge of its behavioral ecology is lacking. First, we show that sponges occupied by breeding males tend to be larger than those occupied by residents. Second, we show that E. lori males exhibit a polygynous mating system: some males breed with multiple females simultaneously and/or sequentially. Third, we show that male size is positively related to multiple metrics of male mating success: number of days paired with females, number of clutches laid, and total reproductive output. Male size, however, explains only a small proportion of the variation in male mating success, suggesting that other aspects of the males’ phenotype and ecological context need to be quantified to better explain variation in mating success. More generally, our results suggest that when female movements are restricted, it is necessary to measure habitat characteristics and male traits at smaller spatial scales to gain a deeper understanding of their effects on mating systems and sexual selection.
Significance statement
Measuring male reproductive success in a wild population is notoriously difficult, particularly in cryptobenthic coral reef fishes. Often, mating success is used as a proxy for reproductive success because it is easier to measure. Variation in mating success is often predicted by characteristics of the individual and its breeding habitat. Here, we simultaneously describe the (i) breeding habitat, (ii) genetic mating system, and (iii) relationship between male traits, habitat characteristics, and mating success in the neon goby Elacatinus lori. By investigating these three elements together, we provide solid foundations for understanding this species’ mating system. More generally, we suggest that investigations of variation in mating success may be improved by approaching analyses at smaller, more biologically relevant, spatial scales. Our findings highlight the challenges and opportunities of investigating mating systems of cryptobenthic coral reef fishes in the wild.





Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available in a GitHub repository, https://github.com/robinkellyfrancis/GobyMatingSuccess.
References
Alonzo SH (2004) Uncertainty in territory quality affects the benefits of usurpation in a Mediterranean wrasse. Behav Ecol 15:278–285. https://doi.org/10.1093/beheco/arh007
Alonzo SH (2010) Social and coevolutionary feedbacks between mating and parental investment. Trends Ecol Evol 25:99–108. https://doi.org/10.1016/j.tree.2009.07.012
Avise JC, Jones AG, Walker DE, DeWoody JA (2002) Genetic mating systems and reproductive natural histories of fishes: lessons for ecology and evolution. Annu Rev Genet 36:19–45. https://doi.org/10.1146/annurev.genet.36.030602.090831
Barneche DR, Robertson DR, White CR, Marshall DJ (2018) Fish reproductive-energy output increases disproportionately with body size. Science 360:642–645. https://doi.org/10.1126/science.aao6868
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Bose APH, Cogliati KM, Luymes N, Bass AH, Marchaterre MA, Sisneros JA, Bolker BM, Balshine S (2018) Phenotypic traits and resource quality as factors affecting male reproductive success in a toadfish. Behav Ecol 29:496–507. https://doi.org/10.1093/beheco/ary002
Branconi R, Barbasch TA, Francis RK, Srinivasan M, Jones GP, Buston PM (2020) Ecological and social constraints combine to promote evolution of non-breeding strategies in clownfish. Commun Biol 3:649. https://doi.org/10.1038/s42003-020-01380-8
Brandl SJ, Goatley CHR, Bellwood DR, Tornabene L (2018) The hidden half: ecology and evolution of cryptobenthic fishes on coral reefs. Biol Rev 93:1846–1873. https://doi.org/10.1111/brv.12423
Brooks R, Kemp DJ (2001) Can older males deliver the good genes? Trends Ecol Evol 16:308–313. https://doi.org/10.1016/S0169-5347(01)02147-4
Brouwer L, Griffith SC (2019) Extra-pair paternity in birds. Mol Ecol 28:4864–4882. https://doi.org/10.1111/mec.15259
Burke T (1989) DNA fingerprinting and other methods for the study of mating success. Trends Ecol Evol 4:139–144. https://doi.org/10.1016/0169-5347(89)90213-9
Buston PM, Elith J (2011) Determinants of reproductive success in dominant pairs of clownfish: a boosted regression tree analysis. J Anim Ecol 80:528–538. https://doi.org/10.1111/j.1365-2656.2011.01803.x
Candolin U, Voigt HR (2001) Correlation between male size and territory quality: consequence of male competition or predation susceptibility? Oikos 95:225–230. https://doi.org/10.1034/j.1600-0706.2001.950204.x
Chen Y, Jackson DA, Harvey HH (1992) A comparison of von Bertalanffy and polynomial functions in modelling fish growth data. Can J Fish Aquat Sci 49:1228–1235. https://doi.org/10.1139/f92-138
Chowdhury MZI, Turin TC (2020) Variable selection strategies and its importance in clinical prediction modelling. Fam Community Health 8:e000262–e000262. https://doi.org/10.1136/fmch-2019-000262
Clark RG, Shutler D (1999) Avian habitat selection: pattern from process in nest-site use by ducks. Ecology 80:272–287. https://doi.org/10.1890/0012-9658(1999)080[0272:AHSPFP]2.0.CO;2
Clutton-Brock TH (1988) Reproductive success: studies of individual variation in contrasting breeding systems. University of Chicago Press
Clutton-Brock TH, Parker GA (1992) Potential reproductive rates and the operation of sexual selection. Q Rev Biol 67:437–456. https://doi.org/10.1086/417793
Cody ML (1981) Habitat selection in birds: the roles of vegetation structure, competitors, and productivity. BioSci 31:107–113. https://doi.org/10.2307/1308252
Colin PL (2002) A new species of sponge-dwelling Elacatinus (Pisces: Gobiidae) from the Western Caribbean. Zootaxa 106:20. https://doi.org/10.11646/zootaxa.106.1.1
Colin PL (2010) Fishes as living tracers of connectivity in the tropical western North Atlantic: I. distribution of the neon gobies, genus Elacatinus (Pisces: Gobiidae). Zootaxa 2370:36–52. https://doi.org/10.11646/zootaxa.2370.1.2
Cooke SJ, Philipp DP, Wahl DH, Weatherhead PJ (2006) Energetics of parental care in six syntopic centrarchid fishes. Oecologia 148:235–249. https://doi.org/10.1007/s00442-006-0375-6
Côte IM, Hunte W (1989) Male and female mate choice in the redlip blenny: why bigger is better. Anim Behav 38:78–88. https://doi.org/10.1016/S0003-3472(89)80067-3
D’Aloia CC, Bogdanowicz SM, Andrés JA, Buston PM (2022) Population assignment tests uncover rare long-distance marine larval dispersal events. Ecology 103:e03559. https://doi.org/10.1002/ecy.3559
D’Aloia CC, Bogdanowicz SM, Francis RK, Majoris JE, Harrison RG, Buston PM (2015) Patterns, causes, and consequences of marine larval dispersal. P Natl Acad Sci USA 112:13940. https://doi.org/10.1073/pnas.1513754112
D’Aloia CC, Bogdanowicz SM, Harrison RG, Buston PM (2014) Seascape continuity plays an important role in determining patterns of spatial genetic structure in a coral reef fish. Mol Ecol 23:2902–2913. https://doi.org/10.1111/mec.12782
D’Aloia CC, Bogdanowicz SM, Harrison RG, Buston PM (2017) Cryptic genetic diversity and spatial patterns of admixture within Belizean marine reserves. Conserv Genet 18:211–223. https://doi.org/10.1007/s10592-016-0895-5
D’Aloia CC, Bogdanowicz SM, Majoris JE, Harrison RG, Buston PM (2013) Self-recruitment in a Caribbean reef fish: a method for approximating dispersal kernels accounting for seascape. Mol Ecol 22:2563–2572. https://doi.org/10.1111/mec.12274
D’Aloia CC, Majoris JE, Buston PM (2011) Predictors of the distribution and abundance of a tube sponge and its resident goby. Coral Reefs 30:777. https://doi.org/10.1007/s00338-011-0755-1
D’Aloia CC, Xuereb A, Fortin MJ, Bogdanowicz SM, Buston PM (2018) Limited dispersal explains the spatial distribution of siblings in a reef fish population. Mar Ecol Prog Ser 607:143–154. https://doi.org/10.3354/meps12792
Emlen ST, Oring LW (1977) Ecology, sexual selection, and the evolution of mating systems. Science 197:215–223. https://doi.org/10.1126/science.327542
García-Borboroglu P, Yorio P (2004) Effects of microhabitat preferences on kelp gull Larusdominicanus breeding performance. J Avian Biol 35:162–169. https://doi.org/10.1111/j.0908-8857.2004.03149.x
Griffith SC, Owens IPF, Thuman KA (2002) Extra pair paternity in birds: a review of interspecific variation and adaptive function. Mol Ecol 11:2195–2212. https://doi.org/10.1046/j.1365-294X.2002.01613.x
Hastings PA (1992) Nest-site size as a short-term constraint on the reproductive success of paternal fishes. Environ Biol Fish 34:213–218. https://doi.org/10.1007/BF00002397
Hernaman V, Munday PL (2007) Evolution of mating systems in coral reef gobies and constraints on mating system plasticity. Coral Reefs 26:585–595. https://doi.org/10.1007/s00338-007-0222-1
Hughes C (1998) Integrating molecular techniques with field methods in studies of social behavior: a revolution results. Ecology 79:383–399. https://doi.org/10.1890/0012-9658(1998)079[0383:IMTWFM]2.0.CO;2
Jakob EM, Marshall SD, Uetz GW (1996) Estimating fitness: a comparison of body condition indices. Oikos 77:61–67. https://doi.org/10.2307/3545585
Jombart T (2008) adegenet: A R package for the multivariate analysis of genetic markers. Bioinformatics 24:1403–1405. https://doi.org/10.1093/bioinformatics/btn129
Jones AG, Avise JC (2001) Mating systems and sexual selection in male-pregnant pipefishes and seahorses: insights from microsatellite-based studies of maternity. J Hered 92:150–158. https://doi.org/10.1093/jhered/92.2.150
Jones OR, Wang J (2010) COLONY: a program for parentage and sibship inference from multilocus genotype data. Mol Ecol Resour 10(3):551–555. https://doi.org/10.1111/j.1755-0998.2009.02787.x
Jones AG, Rosenqvist G, Berglund A, Avise JC (1999) The genetic mating system of a sex-role-reversed pipefish (Syngnathustyphle): a molecular inquiry. Behav Ecol Sociobiol 46:357–365. https://doi.org/10.1007/s002650050630
Kelly CD (2008) The interrelationships between resource-holding potential, resource-value and reproductive success in territorial males: how much variation can we explain? Behav Ecol Sociobiol 62:855–871. https://doi.org/10.1007/s00265-007-0518-8
Koukouras A, Russo A, Voultsiadou-Koukoura E, Dounas C, Chintiroglou C (1992) Relationship of sponge macrofauna with the morphology of their hosts in the North Aegean Sea. Int Rev Ges Hydrobio Hydrograph 77:609–619. https://doi.org/10.1002/iroh.19920770406
Lindström K (1992) The effect of resource holding potential, nest size and information about resource quality on the outcome of intruder-owner conflicts in the sand goby. Behav Ecol Sociobiol 30:53–58. https://doi.org/10.1007/BF00168594
Majoris JE, Foretich MA, Hu Y, Nickles KR, Di Persia CL, Chaput R, Schlatter E, Webb JF, Paris CB, Buston PM (2021) An integrative investigation of sensory organ development and orientation behavior throughout the larval phase of a coral reef fish. Sci Rep 11:12377. https://doi.org/10.1038/s41598-021-91640-2
Majoris JE, Francisco FA, Atema J, Buston PM (2018a) Reproduction, early development, and larval rearing strategies for two sponge-dwelling neon gobies Elacatinus lori and E. colini. Aquaculture 483:286–295. https://doi.org/10.1016/j.aquaculture.2017.10.024
Majoris JE, D’Aloia CC, Francis RK, Buston PM (2018b) Differential persistence favors habitat preferences that determine the distribution of a reef fish. Behav Ecol 29:429–439. https://doi.org/10.1093/beheco/arx189
Martin TE (1987) Food as a limit on breeding birds: a life-history perspective. Annu Rev Ecol S 18:453–487. https://doi.org/10.1146/annurev.es.18.110187.002321
Martin TE (1993) Nest predation and nest sites. Bioscience 43:523–532. https://doi.org/10.2307/1311947
McMurray SE, Blum JE, Pawlik JR (2008) Redwood of the reef: growth and age of the giant barrel sponge Xestospongia muta in the Florida Keys. Mar Biol 155:159–171. https://doi.org/10.1007/s00227-008-1014-z
McMurray SE, Pawlik JR, Finelli CM (2014) Trait-mediated ecosystem impacts: how morphology and size affect pumping rates of the Caribbean giant barrel sponge. Aquat Biol 23:1–13. https://doi.org/10.3354/ab00612
Møller AP, Birkhead TR (1994) The evolution of plumage brightness in birds is related to extrapair paternity. Evolution 48:1089–1100. https://doi.org/10.1111/j.1558-5646.1994.tb05296.x
Møller AP, Jennions MD (2002) How much variance can be explained by ecologists and evolutionary biologists? Oecologia 132:492–500. https://doi.org/10.1007/s00442-002-0952-2
Morganti TM, Ribes M, Yahel G, Coma R (2019) Size is the major determinant of pumping rates in marine sponges. Front Physiol 10:1474. https://www.frontiersin.org/article/https://doi.org/10.3389/fphys.2019.01474
Orians GH (1969) On the evolution of mating systems in birds and mammals. Am Nat 103:589–603. https://doi.org/10.1086/282628
Perrone M, Zaret TM (1979) Parental care patterns of fishes. Am Nat 113:351–361. https://doi.org/10.1086/283394
Pröhl H (2002) Population differences in female resource abundance, adult sex ratio, and male mating success in Dendrobatespumilio. Behav Ecol 13:175–181. https://doi.org/10.1093/beheco/13.2.175
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, https://www.R-project.org/
Rickborn AJ, Buston PM (2015) Life-history transitions of the coral-reef fish Elacatinuslori. J Fish Biol 86:637–650. https://doi.org/10.1111/jfb.12587
Rosenzweig ML (1981) A theory of habitat selection. Ecology 62:327–335. https://doi.org/10.2307/1936707
Rueger T, Harrison HB, Gardiner NM, Berumen ML, Jones GP (2019) Extra-pair mating in a socially monogamous and paternal mouth-brooding cardinalfish. Mol Ecol 28:2625–2635. https://doi.org/10.1111/mec.15103
Saenz-Agudelo P, Jones GP, Thorrold SR, Planes S (2015) Mothers matter: contribution to local replenishment is linked to female size, mate replacement and fecundity in a fish metapopulation. Mar Biol 162:3–14. https://doi.org/10.1007/s00227-014-2556-x
Sillero-Zubiri C, Gottelli D, Macdonald DW (1996) Male philopatry, extra-pack copulations and inbreeding avoidance in Ethiopian wolves (Canissimensis). Behav Ecol Sociobiol 38:331–340. https://doi.org/10.1007/s002650050249
Taborsky M (1994) Sneakers, satellites, and helpers: parasitic and cooperative behavior in fish reproduction. Adv Stud Behav 23:e100
Taborsky M (2008) Alternative reproductive tactics in fish. In: Oliveira RF, Taborsky M, Brockmann HJ (eds) Alternative reproductive tactics: an integrative approach. Cambridge University Press, Cambridge, pp 251–299
Truett GE, Heeger P, Mynatt RL, Truett AA, Walker JA, Warman ML (2000) Preparation of PCR-quality mouse genomic DNA with hot sodium hydroxide and tris (HotSHOT). Biotechniques 29:52–54. https://doi.org/10.2144/00291bm09
Venables WN, Ripley BD (2002) Modern applied statistics with S, 4th edn. Springer, New York, https://www.stats.ox.ac.uk/pub/MASS4/
Warner RR (1984) Mating behavior and hermaphroditism in coral reef fishes. Am Sci 72:128–136
Westneat DF (1987) Extra-pair fertilizations in a predominantly monogamous bird: genetic evidence. Anim Behav 35:877–886. https://doi.org/10.1016/S0003-3472(87)80123-9
Whiteman EA, Côté IM (2003) Social monogamy in the cleaning goby Elacatinus evelynae: ecological constraints or net benefit? Anim Behav 66:281–291. https://doi.org/10.1006/anbe.2003.2200
Wilkinson CR, Cheshire AC (1989) Patterns in the distribution of sponge populations across the central Great Barrier Reef. Coral Reefs 8:127–134. https://doi.org/10.1007/BF00338268
Wong MYL (2010) Ecological constraints and benefits of philopatry promote group-living in a social but non-cooperatively breeding fish. Proc R Soc Lond B 277:353–358. https://doi.org/10.1098/rspb.2009.1453
Wong MYL, Jordan LA, Marsh-Rollo S, St-Cyr S, Reynolds JO, Stiver KA, Desjardins JK, Fitzpatrick JL, Balshine S (2012) Mating systems in cooperative breeders: the roles of resource dispersion and conflict mitigation. Behav Ecol 23:521–530. https://doi.org/10.1093/beheco/arr218
Wong MYL, Uppaluri C, Medina A, Seymour J, Buston PM (2016) The four elements of within-group conflict in animal societies: an experimental test using the clown anemonefish, Amphiprion percula. Behav Ecol Sociobiol 70:1467–1475. https://doi.org/10.1007/s00265-016-2155-6
Zabala J, Zuberogoita I (2014) Individual quality explains variation in reproductive success better than territory quality in a long-lived territorial raptor. PLoS ONE 9:e90254. https://doi.org/10.1371/journal.pone.0090254
Acknowledgements
We thank Diana Acosta, Derek Scolaro, Alissa Rickborn, E Schlatter, and Romain Chaput for assistance with field work; Alben David, Kevin David, and Earl David for captaining our boats; and the staff at International Zoological Expeditions. This manuscript forms part of RKF’s doctoral dissertation requirements at Boston University. RKF would like to thank the members of her committee, Dr. Robert Warner and Dr. Sarah Davies, for comments on earlier versions of the manuscript. We also thank two anonymous reviewers, for editing and improving this manuscript.
Funding
Our research was funded by the National Science Foundation and the Trustees of Boston University (grant numbers: OCE-1260424; OCE-1459546). Additional funding was provided by a Graduate Research Abroad Fellowship and the Warren McLeod Annual Fellowship awarded to RKF by the Biology Department and the Marine Program of Boston University.
Author information
Authors and Affiliations
Contributions
RKF, KAC, JEM, CCD, and PMB collected field data. RKF, CCD, and SB conducted laboratory work. RKF, CCD, SB, TR, and PMB were involved with analyses. All authors were involved with writing and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All applicable international, national, and/or institutional guidelines to conduct our study were followed. Approval for the use of animals in our study was permitted by the Institutional Animal Care and Use Committee at Boston University. Approval for field work was permitted by the Belize Fisheries Department.
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by J. Mann
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is a contribution to the Topical Collection Measuring individual reproductive success in the wild—Guest Editors: Marco Festa-Bianchet, Janet Mann
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Francis, R.K., Catalano, K.A., Majoris, J.E. et al. Characteristics of breeding habitat, genetic mating system, and determinants of male mating success in the sponge-dwelling goby Elacatinus lori. Behav Ecol Sociobiol 76, 157 (2022). https://doi.org/10.1007/s00265-022-03263-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-022-03263-w