Abstract
CD8+ T cells recognizing minor histocompatibility antigens (MiHA) on solid tumor cells may mediate effective graft-versus-tumor (GVT) reactivity after allogeneic stem cell transplantation (SCT). Previously, we identified LRH-1 as a hematopoietic-restricted MiHA encoded by the P2X5 gene. Here, we report that LRH-1 is aberrantly expressed on solid tumor cells. P2X5 mRNA expression is demonstrated in a significant portion of solid tumor cell lines, including renal cell carcinoma (RCC), melanoma, colorectal carcinoma, brain cancer and breast cancer. Importantly, P2X5 gene expression was also detected in a subset of primary solid tumor specimens derived from RCC, brain cancer and breast cancer patients. Furthermore, P2X5 expressing solid tumor cells can be effectively targeted by LRH-1-specific cytotoxic T lymphocytes under inflammatory conditions. The expression of HLA-B7 and CD54 on tumor cells increases upon cytokine stimulation resulting in improved T cell activation as observed by higher levels of degranulation and enhanced tumor cell lysis. Overall, hematopoietic-restricted MiHA LRH-1 is aberrantly expressed on solid tumor cells and may be used as target in GVT-specific immunotherapy after SCT.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Allogeneic hematopoietic stem cell transplantation (SCT) is an established curative treatment for patients with hematological malignancies and an experimental approach for solid tumors [3, 13]. Initial evidence for a beneficial graft-versus-tumor (GVT) effect in solid tumors was demonstrated in patients with metastatic breast cancer [10, 27]. Subsequent studies investigated the feasibility and safety of nonmyeloablative stem cell transplantation (NST) in order to reduce treatment-related toxicity [5, 6, 21, 22]. In these nonmyeloablative regimens, immunosuppression allows donor engraftment but frequently results in mixed chimerism. Conversion to complete donor chimerism can be achieved by withdrawal of immunosuppression and adoptive immunotherapy by donor lymphocyte infusion (DLI) [4]. Several studies have demonstrated that treatment with NST and DLI can induce tumor regressions in some types of metastatic solid tumors [5, 6, 21, 22]. However, this GVT effect is often accompanied by severe graft-versus-host disease (GVHD), which results in considerable morbidity and mortality. Furthermore, most responses in patients with metastatic solid tumors are partial and the rate of complete remissions is limited. Therefore, further development of SCT-based allogeneic immunotherapy for solid tumors requires novel strategies to selectively boost GVT immunity in the absence of GVHD.
The GVT effect is predominantly mediated by donor-derived T cells recognizing minor histocompatibility antigens (MiHA) on malignant cells of the recipient. However, ubiquitously expressed MiHA often induce GVHD, resulting in T cell-mediated destruction of normal tissues such as skin, liver and gut. Therefore, it would be highly beneficial to direct donor T cell immunity towards MiHA that are selectively co-expressed on hematopoietic cells and solid tumor cells by vaccination or T cell therapy. Only a few hematopoietic-restricted MiHA have been described that are aberrantly expressed on solid tumors, including HA-1 [12, 16], ECGF-1 [23] and BCL2A1 [2, 24]. Therefore, it is important to enlarge the panel of MiHA that can be used for immunotherapy of solid tumors.
Previously, we have identified a hematopoietic-restricted MiHA, designated LRH-1, which elicits an HLA-B7-restricted cytotoxic T cell (CTL) response in leukemia patients treated with DLI [7]. LRH-1 is encoded by the P2X5 gene and results from a single nucleotide frameshift polymorphism between recipient and donor cells. P2X5 expression has been demonstrated in myeloid progenitor cells and peripheral blood T cells, B cells and NK cells as well as lymphoid organs [7]. Furthermore, P2X5 is expressed in a broad range of lymphoid malignancies resulting in effective LRH-1-specific CTL-mediated lysis [20]. In contrast, P2X5 mRNA was not detected in primary cell cultures of non-hematopoietic origin such as fibroblasts, keratinocytes, melanocytes and proximal tubule renal epithelial cells. Moreover, P2X5 expression is not detected in prominent GVHD target tissues such as skin, liver, colon and small intestine [7].
Here, we investigated whether the P2X5-encoded LRH-1 epitope is expressed on solid tumors in addition to hematopoietic cells. We revealed that P2X5 mRNA is aberrantly expressed in several types of solid tumors, including renal cell carcinoma (RCC), melanoma, colorectal carcinoma, brain cancer and breast cancer. Furthermore, we demonstrated that P2X5-expressing solid tumor cell lines are efficiently recognized and lysed by LRH-1-specific CTL. These findings illustrate that LRH-1 can be used as target in the development of MiHA-specific immunotherapy after allogeneic SCT for metastatic solid tumors.
Materials and methods
Cell lines
The following solid tumor cell lines were used: RCC cell lines SKRC 1, 7, 10, 12, 17, 18, 24, 33, 35, 52, and 59 were kindly provided by Dr. E. Oosterwijk (Department of Urology, Radboud University Nijmegen Medical Centre (RUNMC), Nijmegen, The Netherlands); melanoma cell lines BLM, FM3, MEL397, and 518A2 were kindly provided by Dr. J. de Vries (Department of Tumor Immunology, RUNMC, Nijmegen, The Netherlands); colorectal carcinoma cell lines HT29, KATO3, HCT15, HCT116, RKO, SW480, and SW620, and brain cancer cell lines BE(-2)C, CHP212, DAOY, D283med, PFSK-1, SK-N-DZ, SK-N-SH, SW1088, TE671, and U-87MG, and breast cancer cell lines BT549, CAMA-1, MCF-7, MDA-MB-134VI, MDA-MB-157, MDA-MB-175VII, MDA-MB-361, MDA-MB-436, SK-BR-3, and T47D were kindly provided by Dr. M. Schutte (Department of Medical Oncology, Josephine Nefkens Institute, Erasmus University Medical Center, Rotterdam, The Netherlands). Skin-derived fibroblasts were kindly provided by S. van der Velde-Visser (Department of Human Genetics, RUNMC, Nijmegen, The Netherlands). All cell lines were cultured in Iscove’s modified Dulbecco’s medium (IMDM) (Invitrogen, Carlsbad, California) supplemented with 10% fetal calf serum (FCS). Prior to analysis solid tumor cell lines and fibroblasts and keratinocytes were cultured for 4 days in the absence or presence of inflammatory cytokines IFNγ (100 U/ml) and TNFα (10 ng/ml).
Primary tumor specimens
mRNA from 42 primary solid tumor specimens was used for gene expression analysis. mRNA from 15 RCC samples, containing ≥80% tumor cells, was kindly provided by Dr. E. Oosterwijk, (Department of Urology, RUNMC, Nijmegen, The Netherlands), mRNA from 13 brain tumors (4 medulla blastomas and 9 gliomas), containing ≥80% tumor cells [14], was kindly provided by H. Jacobs (Department of Pediatric Oncology, RUNMC, Nijmegen, The Netherlands), and mRNA from 14 breast cancer samples, containing 70–90% tumor cells [11], was kindly provided by Dr. P. Span (Department of Chemical Endocrinology, RUNMC, Nijmegen, The Netherlands).
CTL culture
CTL were cultured as previously described [7, 20]. CTL RP1 recognizes the 9-mer epitope TPNQRQNVC in the context of HLA-B*0702. The HLA-B7-alloreactive CTL clone KOR18 was kindly provided by Prof. E. Goulmy (Department of Immunohematology, Leiden University Medical Center, The Netherlands).
Immunofluorescence analysis
Tumor cell lines were studied for surface expression of HLA-B7 and adhesion molecules by flow cytometry. Cells were labeled with anti-HLA-B7 (clone BB7.1-PE; Chemicon Millipore, Billerica, MA), anti-CD54 (clone 84H10; Immunotech Beckman Coulter, Fullerton, CA), or anti-CD58 (clone AICD58; Immunotech) and analyzed using the Coulter FC500 flow cytometer (Beckman Coulter, Fullerton, CA). The fold induction in surface expression was calculated by dividing the mean fluorescence intensity (MFI) of cytokine-stimulated cells by the MFI of unstimulated cells.
Real-time quantitative RT-PCR of the P2X5 gene
P2X5 mRNA and CD45 mRNA expression were determined as previously described [7, 20]. The hydroxymethylbilane synthase (HMBS) housekeeping gene was used to normalize expression. The following gene-specific primers and Taqman probes were used: P2X5; P2X5-F 5′-TCCTGGCGTACCTGGTCGT-3′, P2X5-R 5′-CTTCATTCTCAGCACAGACGTTC-3′ and P2X5-probe 5′-(TET)-TGGGTGTTCCTGATAAAGAAGGGTTACCA-(TAMRA)-3′, CD45; CD45-F 5′-TTAGAAATGAGTCGCATAAGAATTGC-3′, CD45-R 5′-CTCCAGGATAGTCTCCATTGTGAAA-3′ and CD45-probe 5′-(TET)-ATTTCCGTGTAAAAGATC-(TAMRA)-3′, and HMBS; HMBS-F 5′-GGCAATGCGGCTGCAA-3′, HMBS-R 5′-GGGTACCCACGCGAATCAC-3′ and HMBS-probe 5′-(VIC)-CTCATCTTTGGGCTGTTTTCTTCCGCC-(TAMRA)-3′. P2X5 mRNA expression was quantified by determining calibration functions using JVM-2 as reference cell line. The level of P2X5 expression was calculated relative to the P2X5 expression in the JVM-2 cell line, which is susceptible to lysis by LRH-1-specific CTL.
P2X5 genotyping
P2X5 genotyping was performed as previously described [20]. The following gene-specific primers and Taqman probes were used: P2X5; P2X5-EXON3-F 5′-CCAAATCAAACCTCAGCACAGAC-3′, P2X5-EXON3-R 5′-CTCAGTGCCTCTCTGGTTCCTTA-3′, P2X5 5C allele-specific probe 5′-(FAM)-ATTGTGACCCCCAACCA-(MGB)-3′ and P2X5 4C allele-specific probe 5′-(VIC)-TGTGACCCCAACCAG-(MGB)-3′.
Flow cytometry-based cytotoxicity studies
Flow cytometry-based cytotoxicity assays were performed as previously described with minor adaptations [15, 20]. Characteristics of the solid tumor cell lines used as targets are described in Table 1. Endogenous HLA-B7+ cell lines were labeled with 2.5 μM carboxyfluorescein diacetate succimidyl ester (CFSE; Molecular Probes Europe, Leiden, The Netherlands). Alternatively, endogenously HLA-B7− cell lines were retrovirally transduced with LZRS-HLA-B*0702-IRES-EGFP as previously described [7] and used as targets without CFSE-labeling. Target cells (1 × 104) were co-cultured with unlabelled effector cells (3 × 104) at an E:T ratio of 3:1 in a total volume of 200 μl IMDM/10% FCS containing 25 U/ml IL-2 in 96-wells flat-bottom plates. For measurement of T cell degranulation at day 1, anti-CD107a (BD Biosciences, Franklin Lakes, NJ) was added to the T cells prior to co-culture. After 1–3 days of co-culture, cells were harvested and 7-amino-actinomycin D (7AAD) was added. Numbers of viable target cells were quantified by flow cytometry.
Results
Expression of the P2X5 gene in solid tumor cells
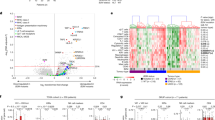
To define whether non-hematopoietic malignancies could be targets for LRH-1-specific CTL, we have analyzed P2X5 mRNA expression in solid tumor cell lines and primary tumor specimens by real-time quantitative RT-PCR. We tested a panel of 42 cell lines of various histological origin including RCC, melanoma, colorectal carcinoma, brain cancer and breast cancer. In addition, we analyzed primary tumor specimens derived from 15 patients with RCC, 13 patients with brain cancer, and 14 patients with breast cancer. Previously, we found that EBV-transformed lymphoblastoid cell lines (EBV-LCL) that have a mean expression level of 2.7 compared with the reference B cell line JVM-2 are significantly recognized by LRH-1-specific CTL [7]. In contrast, monocytes and fibroblasts with a mean P2X5 mRNA expression level of 0.28 and 0.10, respectively, are not susceptible to LRH-1 CTL-mediated lysis. Based on these observations, we used a cut-off P2X5 mRNA level of 0.4 to distinguish P2X5-positive from P2X5-negative cell types [7]. Interestingly, significant P2X5 mRNA expression was observed in 13 out of 42 (31%) solid tumor cell lines, covering all cancer types studied (Fig. 1). We detected P2X5 transcripts in 3 out of 11 RCC cell lines, in 2 out of 4 melanoma cell lines, in 4 out of 7 colorectal carcinoma cell lines, in 3 out of 10 brain cancer cell lines, and in 1 out of 10 breast cancer cell lines. In the primary tumor samples studied, we detected P2X5 mRNA levels of ≥0.4 in 8 out of 15 RCC samples, 4 out of 13 brain cancer samples, and 4 out of 14 breast cancer samples (Table 2). To determine whether P2X5 expression could be due to the presence of hematopoietic cells, we also analyzed expression of CD45. We calculated the ratio of P2X5/CD45 and considered that P2X5 expression in samples with a ratio <1.0 probably resulted from infiltrated hematopoietic cells. We observed that in at least 3 out of 15 RCC samples, 2 out of 13 brain cancer samples, and 2 out of 14 breast cancer samples P2X5 mRNA levels of ≥0.4 with a P2X5/CD45 ratio of >1.0 could be detected (Table 2). These findings corroborate with our results observed in solid tumor cell lines. Together, these data show that the LRH-1-encoding P2X5 gene is significantly expressed in a subset of solid tumors.
P2X5 gene expression in solid tumor cells. P2X5 mRNA expression was determined by real-time quantitative RT-PCR in 42 solid tumor cell lines. Cell lines from the following tumor types were analyzed: renal cell carcinoma (RCC; n = 11), melanoma (n = 4), colorectal carcinoma (n = 7), brain cancer (n = 10), and breast cancer (n = 10). Expression is shown relative to the P2X5 expression measured in reference B-cell line JVM-2, which is susceptible to lysis by LRH-1-specific CTL. The housekeeping gene HMBS was used for normalization. Solid tumor cell lines with P2X5 expression higher than 0.4 were considered positive and potential targets for LRH-1-specific CTL, based on previous expression and recognition studies. This arbitrary threshold is indicated with a dashed line
P2X5-expressing solid tumor cells are efficiently lysed by LRH-1-specific CTL
Target cell recognition by LRH-1-specific CTL is controlled by a single cytosine deletion polymorphism (rs5818907) in exon 3 of the P2X5 gene [7]. Therefore, P2X5-expressing tumor cell lines were genotyped at rs5818907 by allele-specific PCR. Based on this P2X5 genotyping analysis, we have selected the homozygous (C/C genotype) or heterozygous (C/– genotype) LRH-1+ tumor cell lines DAOY (brain cancer), BLM (melanoma), SKRC33 and SKRC18 (both RCC) as targets for LRH-1-specific CTL in cytotoxicity assays (Table 1). For these experiments, we used a flow cytometry-based cytotoxicity assay, which facilitates determination of target cell proliferation and death by both rapid and more slowly T cell effector mechanisms [15]. Moreover, the addition of low-dose IL-2 to the co-cultures prolongs CTL survival, which allows continuous exposure and serial killing of tumor cells by the CTL. Using this in vitro assay, we have demonstrated that LRH-1-specific CTL efficiently lysed LRH-1+ EBV-LCL, whereas no cytotoxicity was observed against LRH-1− EBV-LCL (Fig. 2a). Interestingly, regarding the solid tumor cell lines, we observed that the LRH-1+ brain cancer cell line DAOY could also be efficiently targeted by LRH-1-specific CTL (Fig. 2b). Microscopic analysis confirmed high susceptibility of DAOY for cytotoxicity by LRH-1-specific CTL (Fig. 2c). LRH-1+ melanoma cell line BLM, which has a relative high P2X5 mRNA expression level of 1.5, was less efficiently lysed by LRH-1-specific CTL (Fig. 3a). However, stimulation of BLM with inflammatory cytokines IFNγ and TNFα strongly enhanced susceptibility to lysis by LRH-1-specific CTL. Similar results were obtained with LRH-1+ RCC cell lines. Lysis of RCC cell line SKRC33 was significantly enhanced upon pre-incubation with IFNγ and TNFα (Fig. 4a). In contrast, LRH-1 CTL-mediated lysis of SKRC18 could not be enhanced by inflammatory cytokines, which might be associated with the relative low P2X5 mRNA expression of 0.42 (Fig. 4b). In addition, SKRC 18 did not endogenously express HLA-B7 and HLA-B7 expression could not be upregulated by cytokine pre-incubation (Fig. 5a). The observation that exogenous loading of SKRC18 with LRH-1 peptide resulted in complete lysis indicated that the low responsiveness is most likely caused by low epitope density at the cell surface. No cytotoxicity was observed against HLA-B7-transduced LRH-1− FM3 melanoma cells either unstimulated or pre-stimulated with inflammatory cytokines (Fig. 3b). Allo-HLA-B7-specific CTL lysed both LRH-1+ and LRH-1− solid tumor cell lines (Figs. 2, 3, 4). Taken together, these data demonstrate that LRH-1+ solid tumor cell lines which have sufficient P2X5 expression levels can be effectively targeted by LRH-1 CTL, especially under inflammatory conditions.
Cytotoxicity against solid tumor cell lines was determined after incubation with LRH-1-specific CTL (filled up triangle), HLA-B7-specific CTL (filled inverted triangle; positive control) or medium only (filled diamond). Survival of unstimulated and cytokine-stimulated target cells in the absence or presence of CTL at an E:T ratio of 3:1 is shown of an LRH-1 + and LRH-1 − EBV-LCL (a) and of LRH-1+ brain tumor cell line DAOY (b). P2X5 mRNA expression is shown between parentheses. Data are depicted as mean ± SD of triplicate wells. c Microscopic analysis of cytokine-stimulated DAOY cells at 40 h of co-culture with CTL or medium
Cytotoxicity against LRH-1 + melanoma cell line BLM (a) and LRH-1 − melanoma cell line FM3 (b) was determined after incubation with LRH-1-specific CTL (filled up triangle), HLA-B7-specific CTL (filled inverted triangle; positive control) or medium only (filled diamond). Survival of unstimulated and cytokine-stimulated target cells in the absence or presence of CTL at an E:T ratio of 3:1 is shown. P2X5 mRNA expression is shown between parentheses. Data are depicted as mean ± SD of triplicate wells
Cytotoxicity against LRH-1 + RCC cell lines SKRC33 (a) and SKRC18 (b) was determined after incubation with LRH-1-specific CTL (filled up triangle), HLA-B7-specific CTL (filled inverted triangle; positive control) or medium only (filled diamond). Survival of unstimulated and cytokine-stimulated target cells in the absence or presence of CTL at an E:T ratio of 3:1 is shown. P2X5 mRNA expression is shown between parentheses. Data are depicted as mean ± SD of triplicate wells. c LRH-1-peptide loading resulted in improved lysis of SKRC18
a The expression of adhesion and MHC molecules in epithelial cancer cell lines. Surface expression of HLA-B7, CD54 (ICAM-1) and CD58 (LFA-3) was determined by flow cytometry. Prior to analysis cells were cultured for 4 days in the absence or presence of inflammatory cytokines IFNγ and TNFα. Data are depicted as fold induction of the mean fluorescence intensity (MFI) upon cytokine stimulation. b Cytokine stimulation of cancer cells results in improved degranulation of CTL. Anti-CD107-antibody was added to the co-cultures of flow cytometry-based cytotoxicity assays. Degranulation of LRH-1-specific CTL was measured after 1 day of co-culture with solid tumor cell lines and control EBV-LCL. Data are depicted as mean ± SD of triplicate measurements
Enhanced tumor cell lysis under inflammatory conditions is associated with higher levels of degranulation by LRH-1-specific CTL
To determine whether up-regulation of P2X5 mRNA could explain the increased tumor cell lysis upon stimulation with inflammatory cytokines, we measured P2X5 mRNA levels of untreated and cytokine-treated solid tumor cell lines by real-time quantitative RT-PCR. However, no significant change in P2X5 mRNA expression was observed in the presence of cytokines (Table 1). In addition, we observed that P2X5 expression remains undetectable in keratinocytes and very low in skin-derived fibroblasts in the presence of inflammatory cytokines (Table 3). On the other hand, flowcytometric analysis showed that expression of adhesion molecule CD54 (ICAM-1) increased 3.0-fold to 7.4-fold in solid tumor cell lines upon cytokine stimulation, whereas no significant changes were observed in expression of CD58 (LFA-3) (Fig. 5a). Furthermore, cytokine stimulation resulted in more than threefold increase of HLA-B7 expression in cell lines which are endogenously HLA-B7+ (Fig. 5a).
Next, we determined whether up-regulation of HLA-B7 and CD54 expression on solid tumor cells resulted in increased activation of LRH-1-specific CTL. Therefore, we measured CTL degranulation by expression of CD107a on the cell surface after 1 day of co-incubation with solid tumor cell lines and control EBV-LCL. The proportion of LRH-1-specific CTL expressing CD107a following co-culture with LRH-1-positive and -negative EBV-LCL was 66 and 14%, respectively (Fig. 5b). Consistent with the cytotoxicity data observed in the flow cytometry-based cytotoxicity assays, the percentage CD107a+ CTL was high after co-culture with LRH-1+ solid tumor cell lines DAOY (32%) and SKRC33 (39%), but relatively low for BLM (8%) and SKRC18 (2%). In addition, when co-cultured with tumor cells pre-treated with inflammatory cytokines the proportion of CD107a+ LRH-1-specific CTL significantly increased to 60% for DAOY and 32% for BLM. SKRC18 tumor cells stimulated 25% of LRH-1-specific CTL to degranulate only after pre-treatment with cytokines and loading with LRH-1 peptide. Elevated levels of CTL degranulation correlated with better lysis of tumor cells in cytotoxicity assays.
Together, these results suggest that inflammatory cytokines improve the formation of an immunological synapse by up-regulation of MHC class I and CD54, resulting in higher levels of granule exocytosis and enhanced CTL reactivity. The increased activation of CTL upon cytokine stimulation results in improved tumor cell lysis as observed in cytotoxicity assays.
Discussion
MiHA are considered to play an important role in GVT reactivity after allogeneic SCT for solid tumors [2, 24, 26]. Especially MiHA with tumor-restricted tissue distribution are promising targets to boost GVT reactivity selectively without enhancing GVHD. For further development of tumor-specific immunotherapy it is important to enlarge the spectrum of molecularly identified MiHA which are selectively expressed in solid tumors and possibly hematopoietic cells. Here, we analyzed the expression of the LRH-1-encoding P2X5 gene in a large panel of solid tumor cell lines and primary tumor specimens using real-time quantitative RT-PCR. Significant P2X5 mRNA levels were detected in 31% of the solid tumor cell lines tested, including RCC, melanoma, colorectal carcinoma, brain cancer and breast cancer. Primary samples could be tested for RCC, brain cancer and breast cancer and showed similar numbers of P2X5 positive samples per tumor type. Therefore, LRH-1 belongs to the few hematopoietic-restricted MiHA that additionally show an aberrant expression on solid tumor cells. Expression on solid tumor cells was previously demonstrated for MiHA HA-1 [12, 16], ECGF-1 [23], and BCL2A1 [2, 24]. Enlargement of the panel of MiHA is essential for development of effective post-transplantation immunotherapy. We and others observed that only a part of the tested cell lines and patient samples are positive for MiHA expressed on solid tumors. In addition, the applicability of MiHA is dependent on the frequencies of the MiHA and presenting-HLA phenotypes. Therefore, identification of additional MiHA is essential for further improvement of tumor-specific immunotherapy.
All currently molecularly described MiHA are identified from patients with hematological malignancies or aplastic anemia. Molecular identification of MiHA from patients transplanted for a metastatic solid tumor has not yet been reported. However, Tykodi et al. [26] have described CD8+ CTL clones recognizing MiHA on RCC tumor cells that have been isolated from patients transplanted for metastatic RCC. They demonstrated that CTL clones from two different patients recognize the same MiHA. Though the gene encoding this MiHA was linked to chromosome 19q, they did not describe the molecular characterization of this MiHA yet. Dorrschuck et al. [8] obtained RCC-reactive CTL from co-cultures of mixed lymphocytes with tumor cells. Using microcapillary liquid chromatography and MALDI-TOF spectrometry they identified an HLA-A*0301-associated non-polymorphic peptide, encoded by the ubiquitously expressed Eps-15 homology domain-containing 2 (EHD2) gene. Since EHD2 is broadly expressed in malignant and normal tissues, antigens encoded by this gene are no candidates for tumor-specific immunotherapy.
Recently, the first RCC-associated antigen, CT-RCC1, was identified using allogeneic T cells isolated from a patient treated with allogeneic SCT for metastatic RCC [25]. The antigenic CT-RCC region is a part of the human endogenous retrovirus (HERV) type E locus. Interestingly, HERV-E is selectively expressed on RCC cells, but not on normal tissues including renal cells. However, CT-RCC is presented by HLA-A11 which is only present in a minority of the population. Thus, until more solid tumor-associated antigens or solid tumor-specific MiHA are identified, hematopoietic restricted MiHA co-expressed on solid tumors represent very useful targets for immunotherapy.
Here, two different flow cytometry-based assays were used to study tumor cell recognition. Target cell death was determined with a cytotoxicity assay to study survival kinetics of tumor cells up to 3 days. Cytolytic activation of LRH-1-specific CTL was measured by levels of degranulation. We demonstrated that LRH-1+ cell lines were susceptible to LRH-1-specific CTL-mediated lysis. Especially, brain tumor cell line DAOY was very efficiently lysed by LRH-1-specific CTL. Lysis of melanoma cell line BLM and RCC cell line SKRC33 was significantly enhanced by pre-incubation of target cells with inflammatory cytokines IFNγ and TNFα. P2X5 mRNA expression was not up-regulated in response to inflammatory cytokines, as was previously demonstrated for MiHA BCL2A1 in non-hematopoietic cells [17]. However, cytokine stimulation generally resulted in an increase of HLA-B7 and CD54 expression on tumor cells and higher levels of CD107a expression on LRH-1-specific CTL. Therefore, these results suggest that presence of inflammatory cytokines results in an improved immunological synapse and better T cell activation, which leads to increased target cell lysis. Inflammatory cytokines are known to play a role in anti-tumor immunity [9]. The importance of an inflammatory environment was previously demonstrated in a GVT study in mice, in which secretion of IFNγ was required for eradication of melanomas in vivo by MiHA-specific CTL. Release of IFNγ resulted in inhibition of tumor angiogenesis and up-regulation of MHC class I expression on tumor cells [18]. Importantly, we have demonstrated that P2X5 is selectively expressed in hematopoietic cells [7] and a subset of solid tumor cells, but not in GVHD target tissues such as skin, liver and gut [7]. In addition, here we show that cytokine treatment has an effect on improved lysis of solid tumor cells, whereas P2X5 expression remains undetectable in keratinocytes and very low in skin-derived fibroblasts in the presence of inflammatory cytokines.
Despite the lysis stimulating effect of inflammatory cytokines, partial lysis of RCC cell line SKRC18 could not be enhanced by cytokine pre-incubation. SKRC18 cells could only be efficiently lysed by LRH-1-specific CTL after exogenous loading with LRH-1 peptide. This may indicate that the P2X5 mRNA level, which was just above the threshold of 0.4 (Table 1), was not sufficient for complete lysis. In addition, SKRC 18 did not endogenously express HLA-B7 and HLA-B7 expression could not be up-regulated by cytokine pre-incubation. Another explanation may be a defect in intracellular antigen processing. Resistance to lysis due to defects in antigen-processing mechanisms was previously demonstrated for HA-1+ solid tumor cell lines [19]. Miyazaki et al. describe two HA-1H CTL-resistant cell lines, of which one cell line became susceptible to CTL-mediated lysis after cytokine treatment resulting in restoration of TAP-gene expression. Defects in antigen processing are important mechanisms for metastatic solid tumors to escape from immune-control and may limit the effectiveness of immunotherapy [1].
In conclusion, our data show that the P2X5 gene, which encodes MiHA LRH-1, is aberrantly expressed in a subset of solid tumor cell lines and primary solid tumors. P2X5 expressing tumor types include RCC, melanoma, colorectal carcinoma, brain tumor and breast tumor. Notably, LRH-1+ tumor cells are susceptible to LRH-1 CTL-mediated lysis as shown by flow cytometry-based cytotoxicity assays. This susceptibility may be strongly enhanced by the presence of inflammatory cytokines. Therefore, LRH-1 is an important addition to the panel of hematopoietic-restricted MiHA that are additionally expressed on solid tumor cells and can be exploited for post-transplantation GVT-specific immunotherapy.
References
Abele R, Tampe R (2004) The ABCs of immunology: structure and function of TAP, the transporter associated with antigen processing. Physiology 19(4):216–224
Akatsuka Y, Morishima Y, Kuzushima K, Kodera Y, Takahashi T (2007) Minor histocompatibility antigens as targets for immunotherapy using allogeneic immune reactions. Cancer Sci 98(8):1139–1146
Appelbaum FR (2001) Haematopoietic cell transplantation as immunotherapy. Nature 411(6835):385–389
Arya M, Chao D, Patel HR (2004) Allogeneic hematopoietic stem-cell transplantation: the next generation of therapy for metastatic renal cell cancer. Nat Clin Pract Oncol 1(1):32–38
Bregni M, Dodero A, Peccatori J, Pescarollo A, Bernardi M, Sassi I, Voena C, Zaniboni A, Bordignon C, Corradini P (2002) Nonmyeloablative conditioning followed by hematopoietic cell allografting and donor lymphocyte infusions for patients with metastatic renal and breast cancer. Blood 99(11):4234–4236
Childs R, Chernoff A, Contentin N, Bahceci E, Schrump D, Leitman S, Read EJ, Tisdale J, Dunbar C, Linehan WM, Young NS, Barrett AJ (2000) Regression of metastatic renal-cell carcinoma after nonmyeloablative allogeneic peripheral-blood stem-cell transplantation. N Engl J Med 343(11):750–758
de Rijke B, Horssen-Zoetbrood A, Beekman JM, Otterud B, Maas F, Woestenenk R, Kester M, Leppert M, Schattenberg AV, de Witte T, van de Wiel-van Kemenade E, Dolstra H (2005) A frameshift polymorphism in P2X5 elicits an allogeneic cytotoxic T lymphocyte response associated with remission of chronic myeloid leukemia. J Clin Invest 115(12):3506–3516
Dorrschuck A, Schmidt A, Schnurer E, Gluckmann M, Albrecht C, Wolfel C, Lennerz V, Lifke A, Di Natale C, Ranieri E, Gesualdo L, Huber C, Karas M, Wolfel T, Herr W (2004) CD8+ cytotoxic T lymphocytes isolated from allogeneic healthy donors recognize HLA class Ia/Ib-associated renal carcinoma antigens with ubiquitous or restricted tissue expression. Blood 104(8):2591–2599
Dunn GP, Koebel CM, Schreiber RD (2006) Interferons, immunity and cancer immunoediting. Nat Rev Immunol 6(11):836–848
Eibl B, Schwaighofer H, Nachbaur D, Marth C, Gachter A, Knapp R, Bock G, Gassner C, Schiller L, Petersen F, Niederwieser D (1996) Evidence for a graft-versus-tumor effect in a patient treated with marrow ablative chemotherapy and allogeneic bone marrow transplantation for breast cancer. Blood 88(4):1501–1508
Foekens JA, Atkins D, Zhang Y, Sweep FC, Harbeck N, Paradiso A, Cufer T, Sieuwerts AM, Talantov D, Span PN, Tjan-Heijnen VC, Zito AF, Specht K, Hoefler H, Golouh R, Schittulli F, Schmitt M, Beex LV, Klijn JG, Wang Y (2006) Multicenter validation of a gene expression-based prognostic signature in lymph node-negative primary breast cancer. J Clin Oncol 24(11):1665–1671
Fujii N, Hiraki A, Ikeda K, Ohmura Y, Nozaki I, Shinagawa K, Ishimaru F, Kiura K, Shimizu N, Tanimoto M, Harada M (2002) Expression of minor histocompatibility antigen, HA-1, in solid tumor cells. Transplantation 73(7):1137–1141
Horowitz MM, Gale RP, Sondel PM, Goldman JM, Kersey J, Kolb HJ, Rimm AA, Ringden O, Rozman C, Speck B (1990) Graft-versus-leukemia reactions after bone marrow transplantation. Blood 75(3):555–562
Jacobs JF, Grauer OM, Brasseur F, Hoogerbrugge PM, Wesseling P, Gidding CE, van de Rakt MW, Figdor CG, Coulie PG, de Vries IJ, Adema GJ (2008) Selective cancer-germline gene expression in pediatric brain tumors. J Neurooncol 88(3):273–280
Jedema I, van der Werff NM, Barge RM, Willemze R, Falkenburg JH (2004) New CFSE-based assay to determine susceptibility to lysis by cytotoxic T cells of leukemic precursor cells within a heterogeneous target cell population. Blood 103(7):2677–2682
Klein CA, Wilke M, Pool J, Vermeulen C, Blokland E, Burghart E, Krostina S, Wendler N, Passlick B, Riethmueller G, Goulmy E (2002) The hematopoietic system-specific minor histocompatibility antigen HA-1 shows aberrant expression in epithelial cancer cells. J Exp Med 196(3):359–368
Kloosterboer FM, Luxemburg-Heijs SA, van Soest RA, van Egmond HM, Willemze R, Falkenburg JH (2005) Up-regulated expression in nonhematopoietic tissues of the BCL2A1-derived minor histocompatibility antigens in response to inflammatory cytokines: relevance for allogeneic immunotherapy of leukemia. Blood 106(12):3955–3957
Meunier MC, Delisle JS, Bergeron J, Rineau V, Baron C, Perreault C (2005) T cells targeted against a single minor histocompatibility antigen can cure solid tumors. Nat Med 11(11):1222–1229
Miyazaki M, Akatsuka Y, Nishida T, Fujii N, Hiraki A, Ikeda K, Tsujimura K, Kuzushima K, Morishima Y, Sato S, Ueda R, Takahashi T (2003) Potential limitations in using minor histocompatibility antigen-specific cytotoxic T cells for targeting solid tumor cells. Clin Immunol 107(3):198–201
Overes IM, de Rijke B, Horssen-Zoetbrood A, Fredrix H, de Graaf AO, Jansen JH, van Krieken JH, Raymakers RA, van der Voort R, de Witte TM, Dolstra H (2008) Expression of P2X5 in lymphoid malignancies results in LRH-1-specific cytotoxic T-cell-mediated lysis. Br J Haematol 141(6):799–807
Pedrazzoli P, Da Prada GA, Giorgiani G, Schiavo R, Zambelli A, Giraldi E, Landonio G, Locatelli F, Siena S, Della Cuna GR (2002) Allogeneic blood stem cell transplantation after a reduced-intensity, preparative regimen: a pilot study in patients with refractory malignancies. Cancer 94(9):2409–2415
Rini BI, Zimmerman T, Stadler WM, Gajewski TF, Vogelzang NJ (2002) Allogeneic stem-cell transplantation of renal cell cancer after nonmyeloablative chemotherapy: feasibility, engraftment, and clinical results. J Clin Oncol 20(8):2017–2024
Slager EH, Honders MW, van der Meijden ED, Luxemburg-Heijs SA, Kloosterboer FM, Kester MG, Jedema I, Marijt WA, Schaafsma MR, Willemze R, Falkenburg JH (2006) Identification of the angiogenic endothelial-cell growth factor-1/thymidine phosphorylase as a potential target for immunotherapy of cancer. Blood 107(12):4954–4960
Spierings E, Wieles B, Goulmy E (2004) Minor histocompatibility antigens—big in tumour therapy. Trends Immunol 25(2):56–60
Takahashi Y, Harashima N, Kajigaya S, Yokoyama H, Cherkasova E, McCoy JP, Hanada KI, Mena O, Kurlander R, Abdul T, Srinivasan R, Lundqvist A, Malinzak E, Geller N, Lerman MI, Childs RW (2008) Regression of human kidney cancer following allogeneic stem cell transplantation is associated with recognition of an HERV-E antigen by T cells. J Clin Invest 118(3):1099–1109
Tykodi SS, Warren EH, Thompson JA, Riddell SR, Childs RW, Otterud BE, Leppert MF, Storb R, Sandmaier BM (2004) Allogeneic hematopoietic cell transplantation for metastatic renal cell carcinoma after nonmyeloablative conditioning: toxicity, clinical response, and immunological response to minor histocompatibility antigens. Clin Cancer Res 10(23):7799–7811
Ueno NT, Rondon G, Mirza NQ, Geisler DK, Anderlini P, Giralt SA, Andersson BS, Claxton DF, Gajewski JL, Khouri IF, Korbling M, Mehra RC, Przepiorka D, Rahman Z, Samuels BI, van Besien K, Hortobagyi GN, Champlin RE (1998) Allogeneic peripheral-blood progenitor-cell transplantation for poor-risk patients with metastatic breast cancer. J Clin Oncol 16(3):986–993
Acknowledgments
We thank Frans Maas for technical assistance with PCR. We thank Sandra Tjabringa (Department of Dermatology) for providing control and cytokine-stimulated keratinocytes. This work was supported by grants from the Dutch Cancer Society (KUN 2001-2358), Radboud University Nijmegen (UMCN 2002-29), the Bekales Foundation and the Ger Janssen Foundation.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
I. M. Overes and T. Henriëtte Levenga contributed equally to this study. P. H. De Mulder deceased in April 2007.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Overes, I.M., Henriëtte Levenga, T., Vos, J.C.M. et al. Aberrant expression of the hematopoietic-restricted minor histocompatibility antigen LRH-1 on solid tumors results in efficient cytotoxic T cell-mediated lysis. Cancer Immunol Immunother 58, 429–439 (2009). https://doi.org/10.1007/s00262-008-0569-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-008-0569-3