Abstract
Introduction
Despite recent developments in the diagnosis and treatment of prostate cancer, the advanced stages still have poor survival rates. This warrants further exploration of related molecular targets for patient screening, detection of metastatic disease, and treatment/treatment monitoring. Recent studies have indicated that neurotensin receptors (NTSRs) and their ligand neurotensin (NTS) critically affect the progression of prostate cancers. In this study, we evaluated the expression of neurotensin receptor1 (NTSR1) in patient tissues and performed NTSR1 PET imaging in a prostate cancer animal model.
Methods
The NTSR1 expression was evaluated in 97 cases of prostate cancer and 100 cases of benign prostatic hyperplasia (BPH) of clinical patients by immunohistochemistry staining. The expression profile of PSMA and GRPR was also performed for comparison. The mRNA expression of NTSR1 in LnCap and PC-3 cells was measured by PCR. NTSR1 PET, and biodistribution studies were performed in PC-3 xenografts using 18F-DEG-VS-NT.
Results
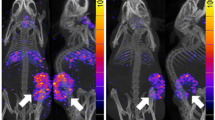
NTSR1 showed high or moderate expression in 91.8% of prostate cancer tissue, compared with PSMA (86.7%) and GRPR (65.3%). All examined PSMA-negative tissues showed positive NTSR1 expression, suggesting the potential complementary role of NTSR1 targeted imaging or therapy. Only 8% of BPH shows strong or moderate expression of NTSR1, which is significantly lower than that in prostate cancer (91.8%). PCR results indicated LNCap (an androgen-dependent prostate cancer cell) showed negative NTSR1 expression while PC-3 demonstrated positive expression (an androgen-independent prostate cancer cell), which correlated well with previously reported western blot results. In a preclinical animal model, NTSR1 targeted PET probe 18F-DEG-VS-NT demonstrated prominent tumor accumulation and low background.
Conclusion
We have demonstrated that NTSR1 is a promising molecular marker for prostate cancer based on patient tissue staining. The NTSR targeted probe 18F-DEG-VS-NT demonstrated high tumor to background contrast in animal models, which could be valuable in selecting patients for therapies targeting NTSR1 as well as monitoring therapeutic efficacy during treatment accordingly.





Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2017. CA Cancer J Clin. 2017;67(1):7–30. https://doi.org/10.3322/caac.21387.
Arrighi N, Bodei S, Zani D, Simeone C, Cunico SC, Missale C, et al. Nerve growth factor signaling in prostate health and disease. Growth Factors. 2010;28(3):191–201. https://doi.org/10.3109/08977190903578678.
Swift SL, Burns JE, Maitland NJ. Altered expression of neurotensin receptors is associated with the differentiation state of prostate cancer. Cancer Res. 2010;70(1):347. https://doi.org/10.1158/0008-5472.CAN-09-1252.
Sehgal I, Powers S, Huntley B, Powis G, Pittelkow M, Maihle NJ. Neurotensin is an autocrine trophic factor stimulated by androgen withdrawal in human prostate cancer. Proc Natl Acad Sci. 1994;91(11):4673. https://doi.org/10.1073/pnas.91.11.4673.
Seethalakshmi L, Mitra SP, Dobner PR, Menon M, Carraway RE. Neurotensin receptor expression in prostate cancer cell line and growth effect of NT at physiological concentrations. Prostate. 1997;31(3):183–92. https://doi.org/10.1002/(sici)1097-0045(19970515)31:3<183::aid-pros7>3.0.co;2-m.
Morgat C, Mishra AK, Varshney R, Allard M, Fernandez P, Hindie E. Targeting neuropeptide receptors for cancer imaging and therapy: perspectives with bombesin, neurotensin, and neuropeptide-Y receptors. J Nucl Med. 2014;55(10):1650–7. https://doi.org/10.2967/jnumed.114.142000.
Yin X, Wang M, Wang H, Deng H, He T, Tan Y, et al. Evaluation of neurotensin receptor 1 as a potential imaging target in pancreatic ductal adenocarcinoma. Amino Acids. 2017;49(8):1325–35. https://doi.org/10.1007/s00726-017-2430-5.
Deng H, Wang H, Wang M, Li Z, Wu Z. Synthesis and evaluation of 64Cu-DOTA-NT-Cy5.5 as a dual-modality PET/fluorescence probe to image neurotensin receptor-positive tumor. Mol Pharm. 2015;12(8):3054–61. https://doi.org/10.1021/acs.molpharmaceut.5b00325.
Valerie NC, Casarez EV, Dasilva JO, Dunlap-Brown ME, Parsons SJ, Amorino GP, et al. Inhibition of neurotensin receptor 1 selectively sensitizes prostate cancer to ionizing radiation. Cancer Res. 2011;71(21):6817–26. https://doi.org/10.1158/0008-5472.CAN-11-1646.
Elshafae SM, Hassan BB, Supsavhad W, Dirksen WP, Camiener RY, Ding H, et al. Gastrin-releasing peptide receptor (GRPr) promotes EMT, growth, and invasion in canine prostate cancer. Prostate. 2016;76(9):796–809. https://doi.org/10.1002/pros.23154.
Kabasakal L, Demirci E, Ocak M, Akyel R, Nematyazar J, Aygun A, et al. Evaluation of PSMA PET/CT imaging using a 68Ga-HBED-CC ligand in patients with prostate cancer and the value of early pelvic imaging. Nucl Med Commun. 2015;36(6):582–7. https://doi.org/10.1097/MNM.0000000000000290.
Lapidus RG, Tiffany CW, Isaacs JT, Slusher BS. Prostate-specific membrane antigen (PSMA) enzyme activity is elevated in prostate cancer cells. Prostate. 2000;45(4):350–4. https://doi.org/10.1002/1097-0045(20001201)45:4<350::aid-pros10>3.0.co;2-u.
Sun Y, Ma X, Zhang Z, Sun Z, Loft M, Ding B, et al. Preclinical study on GRPR-targeted 68Ga-probes for PET imaging of prostate cancer. Bioconjug Chem. 2016;27(8):1857–64. https://doi.org/10.1021/acs.bioconjchem.6b00279.
Gleason D. Histologic grading of prostate carcinoma. Pathology of the prostate. New York: Churchill Livingstone; 1990.
Ng CF, Chiu PK, Lam NY, Lam HC, Lee KW, Hou SS. The prostate health index in predicting initial prostate biopsy outcomes in Asian men with prostate-specific antigen levels of 4-10 ng/mL. Int Urol Nephrol. 2014;46(4):711–7. https://doi.org/10.1007/s11255-013-0582-0.
Maoret JJ, Pospai D, Rouyerfessard C, Couvineau A, Laboisse C, Voisin T, et al. Neurotensin receptor and its mRNA are expressed in many human colon cancer cell lines but not in normal colonic epithelium: binding studies and RT-PCR experiments. Biochem Biophys Res Commun. 1994;203(1):465–71. https://doi.org/10.1006/bbrc.1994.2205.
Deng H, Wang H, Zhang H, Wang M, Giglio B, Ma X, et al. Imaging neurotensin receptor in prostate cancer with 64Cu-labeled neurotensin analogs. Mol Imaging. 2017;16:1536012117711369. https://doi.org/10.1177/1536012117711369.
Wu Z, Li L, Liu S, Yakushijin F, Yakushijin K, Horne D, et al. Facile preparation of a thiol-reactive 18F-labeling agent and synthesis of 18F-DEG-VS-NT for PET imaging of a neurotensin receptor–positive tumor. J Nucl Med. 2014;55(7):1178–84. https://doi.org/10.2967/jnumed.114.137489.
Nadler RB, Humphrey PA, Smith DS, Catalona WJ, Ratliff TL. Effect of inflammation and benign prostatic hyperplasia on elevated serum prostate specific antigen levels. J Urol. 1995;154(2 Pt 1):407–13. https://doi.org/10.1016/S0022-5347(01)67064-2.
Malati T, Kumari GR, Murthy PV, Reddy Ch R, Prakash BS. Prostate specific antigen in patients of benign prostate hypertrophy and carcinoma prostate. Indian J Clin Biochem. 2006;21(1):34–40. https://doi.org/10.1007/BF02913064.
Pandit-Taskar N, O’Donoghue JA, Ruan S, Lyashchenko SK, Carrasquillo JA, Heller G, et al. First-in-human imaging with 89Zr-Df-IAB2M anti-PSMA minibody in patients with metastatic prostate cancer: pharmacokinetics, biodistribution, dosimetry, and lesion uptake. J Nucl Med. 2016;57(12):1858–64. https://doi.org/10.2967/jnumed.116.176206.
Freimanis A, Lietuvietis V, Skrivelis V. 68Ga-PSMA ligand PET/CT in patients with prostate cancer: first experience. Eur Urol Suppl. 2017;16(5):e2220. https://doi.org/10.1016/s1569-9056(17)31363-5.
Maina T, Bergsma H, Kulkarni HR, Mueller D, Charalambidis D, Krenning EP, et al. Preclinical and first clinical experience with the gastrin-releasing peptide receptor-antagonist [68Ga]SB3 and PET/CT. Eur J Nucl Med Mol Imaging. 2016;43(5):964–73. https://doi.org/10.1007/s00259-015-3232-1.
Zhang J, Niu G, Lang L, Li F, Fan X, Yan X, et al. Clinical translation of a dual integrin αvβ3- and gastrin-releasing peptide receptor-targeting PET radiotracer, 68Ga-BBN-RGD. J Nucl Med. 2017;58(2):228–34. https://doi.org/10.2967/jnumed.116.177048.
Uprimny C, Kroiss AS, Decristoforo C, Fritz J, von Guggenberg E, Kendler D, et al. 68Ga-PSMA-11 PET/CT in primary staging of prostate cancer: PSA and Gleason score predict the intensity of tracer accumulation in the primary tumour. Eur J Nucl Med Mol Imaging. 2017;44(6):941–9. https://doi.org/10.1007/s00259-017-3631-6.
Calais J, Czernin J, Cao M, Kishan AU, Hegde JV, Shaverdian N, et al. 68Ga-PSMA-11 PET/CT mapping of prostate cancer biochemical recurrence after radical prostatectomy in 270 patients with a PSA level of less than 1.0 ng/mL: impact on salvage radiotherapy planning. J Nucl Med. 2018;59(2):230–7. https://doi.org/10.2967/jnumed.117.201749.
Bouchelouche K, Turkbey B, Choyke PL. PSMA PET and radionuclide therapy in prostate cancer. Semin Nucl Med. 2016;46(6):522–35. https://doi.org/10.1053/j.semnuclmed.2016.07.006.
Zacho HD, Nielsen JB, Haberkorn U, Stenholt L, Petersen LJ. 68Ga-PSMA PET/CT for the detection of bone metastases in prostate cancer: a systematic review of the published literature. Clin Physiol Funct Imaging. 2017;0(0). doi:https://doi.org/10.1111/cpf.12480.
Laidler P, Dulinska J, Lekka M, Lekki J. Expression of prostate specific membrane antigen in androgen-independent prostate cancer cell line PC-3. Arch Biochem Biophys. 2005;435(1):1–14. https://doi.org/10.1016/j.abb.2004.12.003.
Lee F, Yu A, Anderson A, Marrinucci D, Magargal WW, DiPippo VA, et al. Expression of prostate-specific membrane antigen (PSMA) on circulating tumor cells (CTCs) in castration-resistant prostate cancer. J Clin Oncol. 2014;32(4_suppl):266. https://doi.org/10.1200/jco.2014.32.4_suppl.266.
Dai J, Hensel J, Wang N, Kruithof-de Julio M, Shiozawa Y. Mouse models for studying prostate cancer bone metastasis. Bonekey Rep. 2016;5:777. https://doi.org/10.1038/bonekey.2016.4.
Rybalov M, Ananias HJ, Hoving HD, van der Poel HG, Rosati S, de Jong IJ. PSMA, EpCAM, VEGF and GRPR as imaging targets in locally recurrent prostate cancer after radiotherapy. Int J Mol Sci. 2014;15(4):6046–61. https://doi.org/10.3390/ijms15046046.
Beer M, Montani M, Gerhardt J, Wild P, Hany T, Hermanns T, et al. Profiling gastrin-releasing peptide receptor in prostate tissues: clinical implications and molecular correlates. Prostate. 2012;72:318–25. https://doi.org/10.1002/pros.21434.
Markwalder R, Reubi JC. Gastrin-releasing peptide receptors in the human prostate: relation to neoplastic transformation. Cancer Res. 1999;59(5):1152–9.
Epstein JI, Egevad L, Amin MB, Delahunt B, Srigley JR, Humphrey PA, et al. The 2014 International Society of Urological Pathology (ISUP) consensus conference on Gleason grading of prostatic carcinoma: definition of grading patterns and proposal for a new grading system. Am J Surg Pathol. 2016;40(2):244–52. https://doi.org/10.1097/PAS.0000000000000530.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (NSFC) (Grant No. 81471689, 91859207, and 81771873) and UNC-Chapel Hill LCCC pilot grant (Wu).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants and tissue samples were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The protocol was approved by the Human Research Ethics Committee of Xiangya Hospital, Central South University. Informed consent was obtained from all individual participants included in the study. All procedures performed in studies involving animals were in accordance with the ethical standards of UNC-CH, and the protocol was approved by the UNC Institutional Animal Care and Use Committee.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Translational Research.
Electronic supplementary material
ESM 1
(DOCX 389 kb)
Rights and permissions
About this article
Cite this article
He, T., Wang, M., Wang, H. et al. Evaluation of neurotensin receptor 1 as potential biomarker for prostate cancer theranostic use. Eur J Nucl Med Mol Imaging 46, 2199–2207 (2019). https://doi.org/10.1007/s00259-019-04355-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-019-04355-y