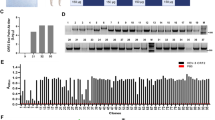
Abstract
Hepatitis E, a significant global public health issue in China, is caused by sporadic infections with regional hepatitis E virus (HEV) genotypes 1, 3, and 4. To date, most immunoassays currently used to test human sera for the presence of anti-HEV antibodies cannot identify HEV at the genotype level. However, such information would be useful for identifying the source of infecting virus. Therefore, here we describe the development of a competitive enzyme-linked immunosorbent assay (ELISA) for detecting anti-genotype 1 HEV antibodies in human sera. Using recombinant genotype 1 HEV ORF3 protein as immunogen, traditional hybridoma technology was employed to generate seven monoclonal antibodies (mAbs), of which two mAbs specifically reacted with the immunogen. One of these two mAbs, 1D2, was labeled with horseradish peroxidase (HRP) for use in competitive ELISA (cELISA). After cELISA optimization using a checkerboard assay design, the amount of ORF3SAR−55 as coating antigen (100 ng/well), HRP-1D2 mAb concentration (1 μg/mL), and test serum dilution (1:10) were selected and a result ≥ 19.5 was used as the cutoff for a positive result. Importantly, cross-genotype cELISA results indicated that the cELISA could not detect anti-genotype 3 rabbit and 4 swine HEV antibodies. Moreover, human sera confirmed as negative for anti-HEV antibodies using the commercial ELISA kit were all negative via cELISA. However, because the commercial ELISA kit detects anti-all genotypes HEV antibodies and the cELISA only detects anti-genotype 1 HEV antibodies, the consistence rate of two assays detecting positive sera is low. In summary, here a cELISA for detecting anti-genotype 1 HEV antibodies was developed for use in epidemiological investigations of genotype 1 HEV infections in humans.
Key points
• Seven mAbs were produced using genotype 1 HEV ORF3 protein as immunogen.
• One mAb that specifically bound to genotype 1 HEV ORF3 protein was selected and labeled for use in a cELISA to detect anti-genotype 1 HEV antibodies.
• The competitive ELISA developed here will aid clinical diagnosis of HEV infections and will be useful for large-scale serological testing of genotype 1 HEV infections in humans.





Similar content being viewed by others
Data availability
All data are provided in the manuscript, and the author promises to support the availability of the data.
References
Ahmad T, Nasir S, Musa TH, AlRyalat SAS, Khan M, Hui J (2021) Epidemiology, diagnosis, vaccines, and bibliometric analysis of the 100 top-cited studies on hepatitis E virus. Hum Vaccin Immunother 17(3):857–871. https://doi.org/10.1080/21645515.2020.1795458
Aslan AT, Balaban HY (2020) Hepatitis E virus: epidemiology, diagnosis, clinical manifestations, and treatment. World J Gastroenterol 26(37):5543–5560. https://doi.org/10.3748/wjg.v26.i37.5543
Cattoir L, Vercauteren KOA, Padalko E, De Beenhouwer H, Van Vaerenbergh K, Boel A (2021) Are VIDAS(R) anti-HEV IgM and IgG assays fit for reliable diagnosis of hepatitis E virus infections? comparison & case story telling. Acta Clin Belg 76(1):25–31. https://doi.org/10.1080/17843286.2019.1655245
Chen Y, Gong QL, Wang Q, Wang W, Wei XY, Jiang J, Ni HB (2021) Prevalence of hepatitis E virus among swine in China from 2010 to 2019: a systematic review and meta-analysis. Microb Pathog 150:104687–104698. https://doi.org/10.1016/j.micpath.2020.104687
Chen Y, Zhao Q, Liu B, Wang L, Sun Y, Li H, Wang X, Syed SF, Zhang G, Zhou EM (2016) A novel blocking ELISA for detection of antibodies against hepatitis E virus in domestic pigs. PLoS ONE 11(3):e0152639. https://doi.org/10.1371/journal.pone.0152639
Ding Q, Heller B, Capuccino JM, Song B, Nimgaonkar I, Hrebikova G, Contreras JE, Ploss A (2017) Hepatitis E virus ORF3 is a functional ion channel required for release of infectious particles. Proc Natl Acad Sci U S A 114(5):1147–1152. https://doi.org/10.1073/pnas.1614955114
Emerson SU, Zhang M, Meng XJ, Nguyen H, St Claire M, Govindarajan S, Huang YK, Purcell RH (2001) Recombinant hepatitis E virus genomes infectious for primates: importance of capping and discovery of a cis-reactive element. Proc Natl Acad Sci U S A 98(26):15270–15275. https://doi.org/10.1073/pnas.251555098
Feng Z (2016) Causation by HEV of extrahepatic manifestations remains unproven. Liver Int 36(4):477–479. https://doi.org/10.1111/liv.13085
Geng Y, Wang Y (2016) Epidemiology of hepatitis E. Adv Exp Med Biol 948:39–59. https://doi.org/10.1007/978-94-024-0942-0_3
Geng Y, Zhao C, Geng K, Wang C, Wang X, Liu H, Wang Y (2019) High seroprevalence of hepatitis E virus in rabbit slaughterhouse workers. Transbound Emerg Dis 66(2):1085–1089. https://doi.org/10.1111/tbed.13130
Gouttenoire J, Pollan A, Abrami L, Oechslin N, Mauron J, Matter M, Oppliger J, Szkolnicka D, Dao Thi VL, van der Goot FG, Moradpour D (2018) Palmitoylation mediates membrane association of hepatitis E virus ORF3 protein and is required for infectious particle secretion. PLoS Pathog 14(12):e1007471. https://doi.org/10.1371/journal.ppat.1007471
Gu Y, Tang X, Zhang X, Song C, Zheng M, Wang K, Zhang J, Ng MH, Hew CL, Li S, Xia N, Sivaraman J (2015) Structural basis for the neutralization of hepatitis E virus by a cross-genotype antibody. Cell Res 25(5):604–620. https://doi.org/10.1038/cr.2015.34
Heelan B (2000) Purification of monoclonal antibodies using protein a/g. Methods Mol Med 40:281–288. https://doi.org/10.1385/1-59259-076-4:281
Kamar N, Bendall R, Legrand-Abravanel F, Xia NS, Ijaz S, Izopet J, Dalton HR (2012) Hepatitis E. Lancet 379(9835):2477–2488. https://doi.org/10.1016/S0140-6736(11)61849-7
Knowles DP Jr, Gorham JR (1993) Advances in the diagnosis of some parasitic diseases by monoclonal antibody-based enzyme-linked immunosorbent assays. Rev Sci Tech 12(2):425–433. https://doi.org/10.20506/rst.12.2.692
Kodani M, Kamili NA, Tejada-Strop A, Poe A, Denniston MM, Drobeniuc J, Kamili S (2017) Variability in the performance characteristics of IgG anti-HEV assays and its impact on reliability of seroprevalence rates of hepatitis E. J Med Virol 89(6):1055–1061. https://doi.org/10.1002/jmv.24741
Lhomme S, Marion O, Abravanel F, Chapuy-Regaud S, Kamar N, Izopet J (2016) Hepatitis E Pathogenesis Viruses 8(8):212–229. https://doi.org/10.3390/v8080212
Liu B, Zhao Q, Sun Y, Wang X, Zhao J, Du T, Wang C, Xiao S, Mu Y, Zhang G, Luo J, Hsu WH, Zhou EM (2014) Development of a blocking ELISA for detection of antibodies against avian hepatitis E virus. J Virol Methods 204:1–5. https://doi.org/10.1016/j.jviromet.2014.03.023
Liu P, Li L, Wang L, Bu Q, Fu H, Han J, Zhu Y, Lu F, Zhuang H (2012) Phylogenetic analysis of 626 hepatitis E virus (HEV) isolates from humans and animals in China (1986–2011) showing genotype diversity and zoonotic transmission. Infect Genet Evol 12(2):428–434. https://doi.org/10.1016/j.meegid.2012.01.017
Liu B, Sun Y, Du T, Chen Y, Wang X, Huang B, Li H, Nan Y, Xiao S, Zhang G, Hiscox JA, Zhou EM, Zhao Q (2017) Rabbit hepatitis E virus is an opportunistic pathogen in specific- pathogenfree rabbits with the capability of cross-species transmission. Vet Microbiol 201:72–77
McHugh ML (2012) Interrater reliability: the kappa statistic. Biochem Med (Zagreb) 22(3):276–282. https://www.ncbi.nlm.nih.gov/pubmed/23092060
Nagashima S, Takahashi M, Kobayashi T, Tanggis NT, Nishiyama T, Primadharsini PP, Okamoto H (2017) Characterization of the quasi-enveloped hepatitis E virus particles released by the cellular exosomal pathway. J Virol 91(22):e00822-e817. https://doi.org/10.1128/JVI.00822-17
Nan Y, Wu C, Zhao Q, Zhou EM (2017) Zoonotic hepatitis E virus: an ignored risk for public health. Front Microbiol 8:2396–2416. https://doi.org/10.3389/fmicb.2017.02396
Nan Y, Zhang YJ (2016) Molecular biology and infection of hepatitis E virus. Front Microbiol 7:1419–1439. https://doi.org/10.3389/fmicb.2016.01419
Nelson KE, Labrique AB, Kmush BL (2019) Epidemiology of genotype 1 and 2 hepatitis E virus infections. Cold Spring Harb Perspect Med 9(6):a031732. https://doi.org/10.1101/cshperspect.a031732
Osterman A, Vizoso Pinto MG, Haase R, Nitschko H, Jager S, Sander M, Motz M, Mohn U, Baiker A (2012) Systematic screening for novel, serologically reactive Hepatitis E Virus epitopes. Virol J 9:28–36. https://doi.org/10.1186/1743-422X-9-28
Syed SF, Sun Y, Du T, Chen Y, Liu B, Wang X, Li H, Nan Y, Zhou EM, Zhao Q (2017) Evaluation of recombinant Chinese avian hepatitis E virus (CaHEV) ORF2 and ORF3 proteins for protection of chickens against CaHEV infection. Vaccine 35(27):3482–3489. https://doi.org/10.1016/j.vaccine.2017.05.030
Tang WF, Kong DG, Wang YH, Liu MQ, Hu Q (2019) Hepatitis E virus infection in Wuhan, central China. Arch Virol 164(1):27–32. https://doi.org/10.1007/s00705-018-4036-y
Tsarev SA, Emerson SU, Reyes GR, Tsareva TS, Legters LJ, Malik IA, Iqbal M, Purcell RH (1992) Characterization of a prototype strain of hepatitis E virus. Proc Natl Acad Sci U S A 89(2):559–563. https://doi.org/10.1073/pnas.89.2.559
Wang B, Meng XJ (2021) Structural and molecular biology of hepatitis E virus. Comput Struct Biotechnol J 19:1907–1916. https://doi.org/10.1016/j.csbj.2021.03.038
Wang S, Cheng X, Dai X, Dong C, Xu M, Liang J, Dong M, Purdy MA, Meng J (2013) Rabbit and human hepatitis E virus strains belong to a single serotype. Virus Res 176(1–2):101–106. https://doi.org/10.1016/j.virusres.2013.05.013
Wang X, Zhao Q, Dang L, Sun Y, Gao J, Liu B, Syed SF, Tao H, Zhang G, Luo J, Zhou EM (2015) Characterization of two novel linear B-cell epitopes in the capsid protein of avian hepatitis E virus (HEV) that are common to avian, swine, and human HEVs. J Virol 89(10):5491–5501. https://doi.org/10.1128/JVI.00107-15
Wang XJ, Zhao Q, Jiang FL, Liu BY, Zhao JN, Dang L, Sun YN, Mu Y, Xiao SQ, Wang CB, Hsu WH, Liu L, Widen F, Zhou EM (2014) Genetic characterization and serological prevalence of swine hepatitis E virus in Shandong province. China Vet Microbiol 172(3–4):415–424. https://doi.org/10.1016/j.vetmic.2014.06.006
Wang Y, Zhang H, Li Z, Gu W, Lan H, Hao W, Ling R, Li H, Harrison TJ (2001) Detection of sporadic cases of hepatitis E virus (HEV) infection in China using immunoassays based on recombinant open reading frame 2 and 3 polypeptides from HEV genotype 4. J Clin Microbiol 39(12):4370–4379. https://doi.org/10.1128/JCM.39.12.4370-4379.2001
Wu C, Wu X, Xia J (2020) Hepatitis E virus infection during pregnancy. Virol J 17(1):73–83. https://doi.org/10.1186/s12985-020-01343-9
Yang Y, Lin S, Nan Y, Ma Z, Yang L, Zhang Y (2016) A linear surface epitope in a proline-rich region of ORF3 product of genotype 1 hepatitis E virus. Viruses 8(8):227–238. https://doi.org/10.3390/v8080227
Yarbough PO, Tam AW, Fry KE, Krawczynski K, McCaustland KA, Bradley DW, Reyes GR (1991) Hepatitis E virus: identification of type-common epitopes. J Virol 65(11):5790–5797. https://doi.org/10.1128/JVI.65.11.5790-5797.1991
Yokoyama WM, Christensen M, Santos GD, Miller D, Ho J, Wu T, Dziegelewski M, Neethling FA (2013) Production of monoclonal antibodies. Curr Protoc Immunol 102:2.5.1-2.5.29. https://doi.org/10.1002/0471142735.im0205s102
Zhang W, He Y, Wang H, Shen Q, Cui L, Wang X, Shao S, Hua X (2010) Hepatitis E virus genotype diversity in eastern China. Emerg Infect Dis 16(10):1630–1632. https://doi.org/10.3201/eid1610.100873
Zhao C, Geng Y, Huang W, Ma H, Wang Y (2018) Valuable antibody detection method for classifying hepatitis E virus genotypes. J Med Virol 90(1):142–147. https://doi.org/10.1002/jmv.24915
Zhao C, Wang Y (2016) Laboratory diagnosis of HEV infection. Adv Exp Med Biol 948:191–209. https://doi.org/10.1007/978-94-024-0942-0_11
Zhao Q, Zhou EM, Dong SW, Qiu HK, Zhang L, Hu SB, Zhao FF, Jiang SJ, Sun YN (2010) Analysis of avian hepatitis E virus from chickens. China Emerg Infect Dis 16(9):1469–1472. https://doi.org/10.3201/eid1609.100626
Zhu FC, Huang SJ, Wu T, Zhang XF, Wang ZZ, Ai X, Yan Q, Yang CL, Cai JP, Jiang HM, Wang YJ, Ng MH, Zhang J, Xia NS (2014) Epidemiology of zoonotic hepatitis E: a community-based surveillance study in a rural population in China. PLoS ONE 9(1):e87154. https://doi.org/10.1371/journal.pone.0087154
Zhuang H, Cao XY, Liu CB, Wang GM (1991) Epidemiology of hepatitis E in China. Gastroenterol Jpn 26(Suppl 3):135–138. https://doi.org/10.1007/BF02779283
Funding
This research is founded by grants from Key International Cooperative Research Project of National Natural Science Foundation of China (No. 31720103919) to E-M.Z and National Natural Science Foundation of China to Q.Z (No. 31972676).
Author information
Authors and Affiliations
Contributions
YS and E-MZ conceived and designed the study. BZ, JF, YL, HL, MF, and BL performed the experiments. BZ, QZ, YN, YS, and E-MZ analyze the experimental data. BZ, QZ, YS, and E-MZ wrote and revised the manuscript. All the authors reviewed the manuscript and approved the final version.
Corresponding authors
Ethics declarations
Ethical approval
This article does not contain any studies with human participants performed by any of the authors. Animal studies with chicken were approved by Animal Care and Use Committee of Northwest Agriculture & Forestry University (NWSUAF, Permit Number: AE189693).
Conflict and interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, B., Fan, J., Luo, Y. et al. Development of a competitive ELISA for detecting antibodies against genotype 1 hepatitis E virus. Appl Microbiol Biotechnol 105, 8505–8516 (2021). https://doi.org/10.1007/s00253-021-11621-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-021-11621-3