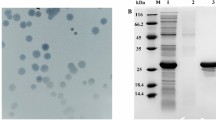
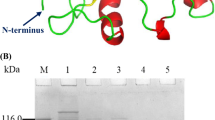
Abstract
Foodborne pathogens have caused many public health incidents and heavy economic burden. Endolysins have been proven to have efficient bactericidal activity against pathogens with low incidence of resistance. In this study, the recombinant endolysin LysSP1 encoded by Salmonella Typhimurium lytic bacteriophage SLMP1 was obtained by prokaryotic expression, and its characteristics were analyzed. Ethylenediaminetetraacetic acid (EDTA) can be used as the outer membrane permeabilizer to increase the bactericidal activity of LysSP1. Under the synergism of 5 mmol/L EDTA, LysSP1 exhibited a strong bactericidal activity against Salmonella Typhimurium ATCC14028. LysSP1 was stable at 4°C for 7 days and at −20°C for 180 days. LysSP1 remained the optimal activity at 40°C and was efficiently active at alkaline condition (pH 8.0–10.0). Divalent metal ions could not enhance the bactericidal activity of LysSP1 and even caused the significant reduction of bactericidal activity. LysSP1 not only could lyse Salmonella, but also could lyse other Gram-negative strains and Gram-positive strains. These results indicated that LysSP1 is a broad-spectrum endolysin and has potential as an antimicrobial agent against Salmonella and other foodborne pathogens.
Key Points
• Recombinant endolysin LysSP1 can be prepared by prokaryotic expression.
• LysSP1 has stable nature and strong bactericidal activity on Salmonella Typhimurium with EDTA.
• LysSP1 has a broad range of hosts including Gram-negative bacteria and Gram-positive bacteria.









Similar content being viewed by others
References
Ajuebor J, McAuliffe O, O’Mahony J, Ross RP, Hill C, Coffey A (2016) Bacteriophage endolysins and their applications. Sci Prog 99(2):183–199. https://doi.org/10.3184/003685016X14627913637705
Cheng M, Zhang Y, Li X, Liang J, Hu L, Gong P, Zhang L, Cai R, Zhang H, Ge J, Ji Y, Guo Z, Feng X, Sun C, Yang Y, Lei L, Han W, Gu J (2017) Endolysin LysEF-P10 shows potential as an alternative treatment strategy for multidrug-resistant Enterococcus faecalis infections. Sci Rep 7(1):10164. https://doi.org/10.1038/s41598-017-10755-7
Deveau H, Garneau JE, Moineau S (2010) CRISPR/Cas system and its role in phage-bacteria interactions. Annu Rev Microbiol 64(1):475–493. https://doi.org/10.1146/annurev.micro.112408.134123
Díez-Martínez R, De Paz HD, García-Fernández E, Bustamante N, Euler CW, Fischetti VA, Menendez M, García P (2015) A novel chimeric phage lysin with high in vitro and in vivo bactericidal activity against Streptococcus pneumoniae. J Antimicrob Chemother 70(6):1763–1773. https://doi.org/10.1093/jac/dkv038
Ding Y, Zhang Y, Huang C, Wang J, Wang X (2020) An endolysin LysSE24 by bacteriophage LPSE1 confers specific bactericidal activity against multidrug-resistant Salmonella strains. Microorganisms 8:737. https://doi.org/10.3390/microorganisms8050737
Doyle RJ, Marquis RE (1994) Elastic, flexible peptidoglycan and bacterial cell wall properties. Trends Microbiol 2(2):57–60. https://doi.org/10.1016/0966-842X(94)90127-9
Fu P, Wang L, Chen J, Bai G, Xu L, Wang S, Guo Y (2019) Analysis of foodborne disease outbreaks in China mainland in 2015. Chin J Food Hygi 31(1):64–70. https://doi.org/10.13590/j.cjfh.2019.01.014 (in Chinese)
Fujiki J, Nakamura T, Furusawa T, Ohno H, Takahashi H, Kitana J, Usui M, Higuchi H, Tanji Y, Tamura Y, Iwano H (2018) Characterization of the lytic capability of a LysK-Like endolysin, Lys-phiSA012, derived from a polyvalent Staphylococcus aureus bacteriophage. Pharmaceuticals 11(1):25. https://doi.org/10.3390/ph11010025
Gervasi T, Horn N, Wegmann U, Dugo G, Narbad A, Mayer MJ (2014) Expression and delivery of an endolysin to combat Clostridium perfringens. Appl Microbiol Biotechnol 98:2495–2505. https://doi.org/10.1007/s00253-013-5128-y
Guldimann C, Johler S (2018) An introduction to current trends in foodborne pathogens and diseases. Curr Clin Micro Rpt 5:83–87. https://doi.org/10.1007/s40588-018-0093-y
Guo M, Feng C, Ren J, Zhuang X, Zhang Y, Zhu Y, Dong K, He P, Guo X, Qin J (2017) A novel antimicrobial endolysin, LysPA26, against Pseudomonas aeruginosa. Front Microbiol 8:293. https://doi.org/10.3389/fmicb.2017.00293
Haddad Kashani H, Fahimi H, Dasteh Goli Y, Moniri R (2017) A novel chimeric endolysin with antibacterial activity against methicillin-resistant Staphylococcus aureus. Front Cell Infet Microbiol 7:290. https://doi.org/10.3389/fcimb.2017.00290
Han F, Li M, Lin H, Wang J (2014) The novel Shewanella putrefaciens-infecting bacteriophage Spp001: genome sequence and lytic enzymes. J Ind Microbiol Biotechnol 41:1017–1026. https://doi.org/10.1007/s10295-014-1438-z
Harada LK, Silva EC, Campos WF, Del Fiol FS, Vila M, Dabrowska K, Krylov VN, Balcão VM (2018) Biotechnological applications of bacteriophages: state of the art. Microbiol Res 212-213:38–58. https://doi.org/10.1016/j.micres.2018.04.007
Kong M, Ryu S (2015) Bacteriophage PBC1 and its endolysin as an antimicrobial agent against Bacillus cereus. Appl Environ Microbiol 81(7):2274–2283. https://doi.org/10.1128/AEM.03485-14
Kumar A, Chordia N (2017) Bacterial resistance against antibiotics. Drug Resistance in Bacteria, Fungi, Malaria, and Cancer PP. 171-192. doi:https://doi.org/10.1007/978-3-319-48683-3_7
Legotsky SA, Vlasova KY, Priyma AD, Shneider MM, Pugachev VG, Totmenina OD, Kabanov AV, Miroshnikov KA, Klyachko NL (2014) Peptidoglycan degrading activity of the broad-range Salmonella bacteriophage S-394 recombinant endolysin. Biochimie 107:293–299. https://doi.org/10.1016/j.biochi.2014.09.017
Li M, Li M, Lin H, Wang J, Jin Y, Han F (2016) Characterization of the novel T4-like Salmonella enterica bacteriophage STP4-a and its endolysin. Arch Virol 161:377–384. https://doi.org/10.1007/s00705-015-2647-0
Lim JA, Shin H, Kang DH, Ryu S (2012) Characterization of endolysin from a Salmonella Typhimurium-infecting bacteriophage SPN1S. Res Microbiol 163:233–241. https://doi.org/10.1016/j.resmic.2012.01.002
Liu A, Wang Y, Cai X, Jiang S, Cai X, Shen L, Liu Y, Han G, Chen S, Wang J, Wu W, Li C, Liu S, Wang X (2019) Characterization of endolysins from bacteriophage LPST10 and evaluation of their potential for controlling Salmonella Typhimurium on lettuce. LWT-Food Sci Technol 114:108372. https://doi.org/10.1016/j.lwt.2019.108372
Lood R, Winer B, Pelzek AJ, Díez-Martínez R, Thandar M, Euler CW, Schuch R, Fischetti VA (2015) Novel phage lysin capable of killing the multidrug-resistant Gram-negative bacterium Acinetobacter baumannii in a mouse bacteremia model. Antimicrob Agents Chemother 59:1983–1991. https://doi.org/10.1128/aac.04641-14
Majowicz SE, Musto J, Scallan E, Angulo FJ, Kirk M, O'Brien SJ, Jones TF, Fazil A, Hoekstra RM (2010) The global burden of nontyphoidal Salmonella gastroenteritis. Clin Infect Dis 50(6):882. https://doi.org/10.1086/650733
Mao X, Hu J, Liu X (2011) Estimation on disease burden of foodborne non-typhoid salmonellosis in China using literature review method. China J Dis Control Prev 15: 622-625. doi: CNKI:SUN:JBKZ.0.2011-07-021 (in Chinese)
Nelson D, Schmelcher M, Rodríguez-Rubio L, Klumpp J, Pritchard DG, Dong S, Donovan DM (2012) Endolysins as antimicrobials. Adv Virus Res 83:299–365. https://doi.org/10.1016/B978-0-12-394438-2.00007-4
Oliveira H, Thiagarajan V, Walmagh M, Sillankorva S, Lavigne R, Neves-Petersen MT, Kluskens LD, Azeredo J (2014) A thermostable Salmonella phage endolysin, Lys68, with broad bactericidal properties against Gram-negative pathogens in presence of weak acids. PLoS One 9(10):e108376. https://doi.org/10.1371/journal.pone.0108376
Richard J (2017) Challenges in oral peptide delivery: lessons learnt from the clinic and future prospects. Ther Deliv 8(8):663–684. https://doi.org/10.4155/tde-2017-0024
Rodríguez-Rubio L, Martínez B, Donovan DM, Rodríguez A, García P (2013) Bacteriophage virion-associated peptidoglycan hydrolases: potential new enzybiotics. Crit Rev Microbiol 39(4):427–434. https://doi.org/10.3109/1040841X.2012.723675
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbor Laboratory Press, New York
Schmelcher M, Loessner M (2016) Bacteriophage endolysins: applications for food safety. Curr Opin Biotechnol 37:76–87. https://doi.org/10.1016/j.copbio.2015.10.005
Schmelcher M, Donovan DM, Loessner M (2012) Bacteriophage endolysins as noval antimicrobials. Future Microbiol 7(10):1147–1171. https://doi.org/10.2217/fmb.12.97
Tong J, Zhang Z, Wu Q, Huang Z, Zhao Y (2021) Antibacterial peptides from seafood: a promising weapon to combat bacterial hazards in food. Food Control 125(2):108004. https://doi.org/10.1016/j.foodcont.2021.108004
Wang W, Li M, Lin H, Wang J, Mao X (2016) The Vibrio parahaemolyticus-infecting bacteriophage qdvp001: genome sequence and endolysin with a modular structure. Arch Virol 161:2645–2652. https://doi.org/10.1007/s00705-016-2957-x
Wren BW (2010) A family of clostridial and streptococcal ligand-binding protein with conserved C-terminal repeat sequences. Mol Microbiol 5:797–803. https://doi.org/10.1111/j.1365-2958.1991.tb00752.x
Yang Q, Ding Y, Nie R, Yao L, Wang X, Zhou M, Wang J, Wang X (2020) Characterization of a novel T7-like Salmonella Typhimurium (ATCC13311) bacteriophage LPST144 and its endolysin. LWT-Food Sci and Technol 123:109034. https://doi.org/10.1016/j.lwt.2020.109034
Young R, Wang I, Roof WD (2000) Phages will out: strategies of host cell lysis. Trends Microbiol 8:120–128. https://doi.org/10.1016/S0966-842X(00)01705-4
Zhang H, Bao H, Billington C, Hudson JA, Wang R (2012) Isolation and lytic activity of the Listeria bacteriophage endolysin LysZ5 against Listeria monocytogenes in soya milk. Food Microbiol 31:133–136. https://doi.org/10.1016/j.fm.2012.01.005
Availability of data and material
All data generated or analyzed during this study are included in this published article.
Code availability
Not applicable.
Funding
This study was supported by the National Key Research and Development Program of China (No. 2017YFC1600703), Central Public-Interest Scientific Institution Basal Research Fund, YSFRI, CAFS (No. 20603022020004), and Central Public-Interest Scientific Institution Basal Research Fund, CAFS (No. 2020TD71).
Author information
Authors and Affiliations
Contributions
Y.J., D.X., and L.Y. conceived and designed the research. Y.J., D.X., L.W., M.Q., and F.L. conducted the experiments. Z.T. and L.Y. contributed analytical tools and helped with data analysis. Y.J. wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies on human or animals.
Consent to participate
Not applicable.
Consent for publication
All authors declare that this article has not been published elsewhere and agree to publish it in this journal.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jiang, Y., Xu, D., Wang, L. et al. Characterization of a broad-spectrum endolysin LysSP1 encoded by a Salmonella bacteriophage. Appl Microbiol Biotechnol 105, 5461–5470 (2021). https://doi.org/10.1007/s00253-021-11366-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-021-11366-z