Abstract
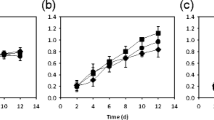
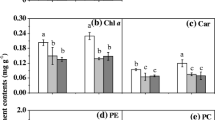
Light management strategy can be used to improve algal biomass and nutrient production. However, the response of algal metabolism to different light qualities, especially their interaction with other environmental factors, is not well understood. This study focuses on the interactive effects of light quality and culturing temperature on algal protein content and carbohydrate content of C. reinhardtii. Three LED light sources (blue light, red-orange light, and white-yellow light) were applied to grow algae in batch cultures with a light intensity of 105 μmol/m2s under the temperatures of 24 °C to 32 °C. The protein and carbohydrate content were measured in both the late exponential growth phase and the late stationary growth phase. The results revealed that there was an interactive effect of light quality and culturing temperature on the protein and carbohydrate content. The combined conditions of blue light and a temperature of 24 °C or 28 °C, which induced a larger algal cell size with a prolonged cell cycle and a low division rate, resulted in the highest protein content; the protein mass fraction and concentration were 32% and 52% higher than that under white-yellow light at 32 °C. The combined conditions of red-orange light and a temperature of 24 °C, which promoted both the cell division and size growth, enhanced the carbohydrate content; the carbohydrate mass fraction and concentration were 161% and 155% higher than that under white-yellow light at 24 °C. When there was temperature stress (32 °C) or nutrient stress, the effect of light quality reduced, and the difference of protein and carbohydrate content among the three light qualities decreased.
Key points
• Studied light quality-temperature interactive effect on protein, carbohydrate synthesis.
• Protein content was high under low cell division rate.
• Carbohydrate content was high under high cell division and cell size growth rate.





Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Abiusi F, Sampietro G, Marturano G, Biondi N, Rodolfi L, D'Ottavio M, Tredici MR (2014) Growth, photosynthetic efficiency, and biochemical composition of T. suecica F&M-M33 grown with LEDs of different colors. Biotechnol Bioeng 111(5):956–964. https://doi.org/10.1002/bit.25014
Ajayan KV, Harilal CC, Gani P (2019) Performance of reflector coated LED Bio-box on the augmentation of growth and lipid production in aerophytic trebouxiophyceaen algae Coccomyxa sp. Algal Res 38:101401. https://doi.org/10.1016/j.algal.2018.101401
Alego G, Bentley C, Smith R, Kelley D, Synder R (2018) LEDs to replace fluorescent tubes for growth of cultured algae VIMS Eastern Shore Laboratory Technical Bulletin No1. Virginia Institute of Marine Science, College of William and Mary. https://doi.org/10.25773/emkh-d241
Barro F, De La Haba P, Maldonado JM, Fontes AG (1989) Effect of light quality on growth, contents of carbohydrates, protein and pigments, and nitrate reductase activity in soybean plants. J Plant Physiol 134(5):586–591. https://doi.org/10.1016/S0176-1617(89)80152-X
Bišová K, Zachleder V (2014) Cell-cycle regulation in green algae dividing by multiple fission. J Exp Bot 65(10):2585–2602
Casal JJ (2000) Phytochromes, cryptochromes, phototropin: photoreceptor interactions in plants. Photochem Photobiol 71(1):1–11. https://doi.org/10.1562/0031-8655(2000)0710001pcppii2.0.co2
Chaplin MF, Kennedy JF (1994) Carbohydrate analysis: a practical approach. IRL Press, New York
Cheng D, Li D, Yuan Y, Zhou L, Li X, Wu T, Wang L, Zhao Q, Wei W, Sun Y (2017) Improving carbohydrate and starch accumulation in Chlorella sp. AE10 by a novel two-stage process with cell dilution. Biotechnol Biofuels 10(1):75. https://doi.org/10.1186/s13068-017-0753-9
Cookson NA, Cookson SW, Tsimring LS, Hasty J (2010) Cell cycle-dependent variations in protein concentration. Nucleic Acids Res 38(8):2676–2681. https://doi.org/10.1093/nar/gkp1069
Daniela B, Clelia S, Cyprien V, Claudia F, Claudia M, Mickael B, Elke R, Petra R (2019) Dried biofilms of desert strains of Chroococcidiopsis survived prolonged exposure to space and Mars-like conditions in low earth orbit. Astrobiology 19:1008–1017. https://doi.org/10.1089/ast.2018.1900
de Castro Araújo S, Garcia VMT (2005) Growth and biochemical composition of the diatom C. cf. wighamii brightwell under different temperature, salinity and carbon dioxide levels. I. Protein, carbohydrates and lipids. Aquaculture 246(1):405–412. https://doi.org/10.1016/j.aquaculture.2005.02.051
de Mooij T, de Vries G, Latsos C, Wijffels RH, Janssen M (2016) Impact of light color on photobioreactor productivity. Algal Res 15:32–42. https://doi.org/10.1016/j.algal.2016.01.015
del Pilar Sánchez-Saavedra M, Voltolina D (1996) Effect of blue-green light on growth rate and chemical composition of three diatoms. J Appl Phycol 8(2):131–137. https://doi.org/10.1007/bf02186316
del Pilar Sánchez-Saavedra M, Voltolina D (2006) The growth rate, biomass production and composition of Chaetoceros sp. grown with different light sources. Aquac Eng 35(2):161–165. https://doi.org/10.1016/j.aquaeng.2005.12.001
Gameiro PA, Struhl K (2018) Nutrient deprivation elicits a transcriptional and translational inflammatory response coupled to decreased protein synthesis. Cell Rep 24(6):1415–1424. https://doi.org/10.1016/j.celrep.2018.07.021
Gibson CE (1978) Field and laboratory observations on the temporal and spatial variation of carbohydrate content in planktonic blue-green algae in Lough Neagh, Northern Ireland. J Ecol 66(1):97–115. https://doi.org/10.2307/2259183
Gobble MM, Headrick D (2019) Converging innovations nourish growth of indoor farming. Res Technol Manag 62(2):7
Goold HD, Cuiné S, Légeret B, Liang Y, Brugière S, Auroy P, Javot H, Tardif M, Jones B, Beisson F, Peltier G, Li-Beisson Y (2016) Saturating light induces sustained accumulation of oil in plastidal lipid droplets in C. reinhardtii. Plant Physiol 171(4):2406–2417. https://doi.org/10.1104/pp.16.00718
Guerlava P, Izac V, Tholozan JL (1998) Comparison of different methods of cell lysis and protein measurements in C. perfringens: Application to the Cell Volume Determination. Curr Microbiol 36(3):131–135. https://doi.org/10.1007/pl00006756
Hegemann P (2008) Algal sensory photoreceptors. Annu Rev Plant Biol 59(1):167–189. https://doi.org/10.1146/annurev.arplant.59.032607.092847
Hogewoning SW, Trouwborst G, Maljaars H, Poorter H, van Ieperen W, Harbinson J (2010) Blue light dose–responses of leaf photosynthesis, morphology, and chemical composition of C. sativus grown under different combinations of red and blue light. J Exp Bot 61(11):3107–3117. https://doi.org/10.1093/jxb/erq132
Huang K, Beck CF (2003) Phototropin is the blue-light receptor that controls multiple steps in the sexual life cycle of the green alga C. reinhardtii. Proc Natl Acad Sci U S A 100(10):6269–6274. https://doi.org/10.1073/pnas.0931459100
Janssen JH, Driessen JLSP, Lamers PP, Wijffels RH, Barbosa MJ (2018) Effect of initial biomass-specific photon supply rate on fatty acid accumulation in nitrogen depleted N. gaditana under simulated outdoor light conditions. Algal Res 35:595–601. https://doi.org/10.1016/j.algal.2018.10.002
Jensen S, Knutsen G (1993) Influence of light and temperature on photoinhibition of photosynthesis in S. platensis. J Appl Phycol 5(5):495–504. https://doi.org/10.1007/bf02182508
Jones MA (2018) Using light to improve commercial value. Hortic Res 5(1):47. https://doi.org/10.1038/s41438-018-0049-7
Juneja A, Ceballos RM, Murthy GS (2013) Effects of environmental factors and nutrient availability on the biochemical composition of algae for biofuels production: a review. Energies 6(9):4607–4638. https://doi.org/10.3390/en6094607
Kim SH, Sunwoo IY, Hong HJ, Awah CC, Jeong G-T, Kim S-K (2019) Lipid and unsaturated fatty acid productions from three microalgae using nitrate and light-emitting diodes with complementary LED wavelength in a two-phase culture system. Bioprocess Biosyst Eng 42:1–10. https://doi.org/10.1007/s00449-019-02149-y
Kirst H, García-Cerdán JG, Zurbriggen A, Melis A (2012) Assembly of the light-harvesting chlorophyll antenna in the green alga C. reinhardtii requires expression of the TLA2-CpFTSY gene. Plant Physiol 158(2):930–945
Kommareddy A, Anderson G (2004) Study of light requirements of a photobioreactor. North Central ASAE/CSAE Conference Presentation MB04-111, Winnipeg, USA
Korbee N, Figueroa FL, Aguilera J (2005) Effect of light quality on the accumulation of photosynthetic pigments, proteins and mycosporine-like amino acids in the red alga P. leucosticta (Bangiales, Rhodophyta). J Photochem Photobiol B Biol 80(2):71–78
Kruger NJ (2009) The bradford method for protein quantitation. In: Walker JM (ed) The Protein Protocols Handbook. Humana Press, Totowa, pp 17–24
Kubín Š, Borns E, Doucha J, Seiss U (1983) Light absorption and production rate of C. vulgaris in light of different spectral composition. Biochem Physiol Pflanz 178(2):193–205. https://doi.org/10.1016/S0015-3796(83)80032-8
Laurens C, Simon C, Vernikos J, Gauquelin-Koch G, Blanc S, Bergouignan A (2019) Revisiting the role of exercise countermeasure on the regulation of energy balance during space flight. Front Physiol 10:321–321. https://doi.org/10.3389/fphys.2019.00321
Li X, Slavens S, Crunkleton DW, Johannes TW (2020) Interactive effect of light quality and temperature on C. reinhardtii growth kinetics and lipid synthesis. Algal Res in press:102127. https://doi.org/10.1016/j.algal.2020.102127
Matula E, Nabity J (2016) Feasibility of photobioreactor systems for use in multifunctional environmental control and life support system for spacecraft and habitat environments. In: 46th International Conference on Environmental Systems, Vienna, Austria. 46th International Conference on Environmental Systems
Matula EE, Nabity JA (2019) Failure modes, causes, and effects of algal photobioreactors used to control a spacecraft environment. Life Sci Space Res 20:35–52. https://doi.org/10.1016/j.lssr.2018.12.001
Michael CI (2015) Utilizing optical light filters and biofilm based cultivating to enhance microalgal growth. Iowa State University, Ames, IA
Mitchell CA (2012) Plant lighting in controlled environments for space and earth applications. Acta Hortic 956:23–36. https://doi.org/10.17660/ActaHortic.2012.956.1
Niederwieser T, Kociolek P, Klaus D (2018) A review of algal research in space. Acta Astronaut 146:359–367. https://doi.org/10.1016/j.actaastro.2018.03.026
Ogbonda KH, Aminigo RE, Abu GO (2007) Influence of temperature and pH on biomass production and protein biosynthesis in a putative Spirulina sp. Bioresour Technol 98(11):2207–2211. https://doi.org/10.1016/j.biortech.2006.08.028
Oldenhof H, Zachleder V, Van Den Ende H (2006) Blue- and red-light regulation of the cell cycle in C. reinhardtii (Chlorophyta). Eur J Phycol 41(3):313–320. https://doi.org/10.1080/09670260600699920
Olle M, Viršile A (2013) The effects of light-emitting diode lighting on greenhouse plant growth and quality. Agric Food Sci 22(2):223–234. https://doi.org/10.23986/afsci.7897
Paul MJ, Foyer CH (2001) Sink regulation of photosynthesis. J Exp Bot 52(360):1383–1400. https://doi.org/10.1093/jexbot/52.360.1383
Pennisi G, Blasioli S, Cellini A, Maia L, Crepaldi A, Braschi I, Spinelli F, Nicola S, Fernandez JA, Stanghellini C, Marcelis LFM, Orsini F, Gianquinto G (2019) Unraveling the role of red:blue led lights on resource use efficiency and nutritional properties of indoor grown sweet Basil. Front Plant Sci 10(305). https://doi.org/10.3389/fpls.2019.00305
Rasband W (2020) Image processing and analysis in Java. PUblisher. https://imagej.nih.gov/. Accessed 4 Dec 2020
Renaud SM, Thinh L-V, Lambrinidis G, Parry DL (2002) Effect of temperature on growth, chemical composition and fatty acid composition of tropical Australian microalgae grown in batch cultures. Aquaculture 211(1):195–214. https://doi.org/10.1016/S0044-8486(01)00875-4
Rivkin RB (1989) Influence of irradiance and spectral quality on the carbon metabolism of hytoplankton. I. Photosynthesis, chemical composition and growth. Mar Ecol Prog Ser 55:291–304
Rochet M, Legendre L, Demers S (1986) Photosynthetic and pigment responses of sea-ice microalgae to changes in light intensity and quality. J Exp Mar Biol Ecol 101(3):211–226. https://doi.org/10.1016/0022-0981(86)90264-9
Rockwell NC, Duanmu D, Martin SS, Bachy C, Price DC, Bhattacharya D, Worden AZ, Lagarias JC (2014) Eukaryotic algal phytochromes span the visible spectrum. PNAS 111(10):3871–3876. https://doi.org/10.1073/pnas.1401871111
Satthong S, Saego K, Kitrungloadjanaporn P, Nuttavut N, Amornsamankul S, Triampo W (2019) Modeling the effects of light sources on the growth of algae. Adv Differ Equ 2019(1):170. https://doi.org/10.1186/s13662-019-2112-6
Sayegh FAQ, Montagnes DJS (2011) Temperature shifts induce intraspecific variation in microalgal production and biochemical composition. Bioresour Technol 102(3):3007–3013. https://doi.org/10.1016/j.biortech.2010.10.011
Schulze PSC, Barreira LA, Pereira HGC, Perales JA, Varela JCS (2014) Light emitting diodes (LEDs) applied to microalgal production. Trends Biotechnol 32(8):422–430. https://doi.org/10.1016/j.tibtech.2014.06.001
Schulze PS, Pereira HG, Santos TF, Schueler L, Guerra R, Barreira LA, Perales JA, Varela JC (2016) Effect of light quality supplied by light emitting diodes (LEDs) on growth and biochemical profiles of N. oculata and T. chuii. Algal Res 16:387–398
Sforza E, Barbera E, Bertucco A (2015) Improving the photoconversion efficiency: an integrated photovoltaic-photobioreactor system for microalgal cultivation. Algal Res 10:202–209. https://doi.org/10.1016/j.algal.2015.05.005
Soeder CJ, Stengel E (1974) Physico-chemical factors affecting metabolism and growth rate. In: Stewart WDP (ed) Algal Physiology and Biochemistry. Univ of California Press, Oakland, CA
Sung MG, Han JI, Lee B, Chang YK (2018) Wavelength shift strategy to enhance lipid productivity of N. gaditana. Biotechnol Biofuels 11:70–70. https://doi.org/10.1186/s13068-018-1067-2
Svetoslav A (2016) Algal research in space: history, current status and future prospects. Innovare J Life Sci 4(4):1–4
Trainer VL, Moore SK, Hallegraeff G, Kudela RM, Clement A, Mardones JI, Cochlan WP (2019) Pelagic harmful algal blooms and climate change: lessons from nature’s experiments with extremes. Harmful Algae 91:101591. https://doi.org/10.1016/j.hal.2019.03.009
Uslu LH, Işık O, Sayın S, Durmaz Y, Göksan T, Gökpınar Ş (2009) The effect of temperature on protein and amino acid composition of S. platensis. J Fish Aquat Sci 26:139–142
Vadiveloo A, Moheimani NR, Cosgrove JJ, Bahri PA, Parlevliet D (2015) Effect of different light spectra on the growth and productivity of acclimated Nannochloropsis sp.(Eustigmatophyceae). Algal Res 8:121–127
Wells ML, Potin P, Craigie JS, Raven JA, Merchant SS, Helliwell KE, Smith AG, Camire ME, Brawley SH (2017) Algae as nutritional and functional food sources: revisiting our understanding. J Appl Phycol 29(2):949–982. https://doi.org/10.1007/s10811-016-0974-5
Wu H (2016) Effect of different light qualities on growth, pigment content, chlorophyll fluorescence, and antioxidant enzyme activity in the red alga P. haitanensis (Bangiales, Rhodophyta). Biomed Res Int 2016:1–8
Zaiontz C (2020) Unbalanced Factorial ANOVA. PUblisher. http://www.real-statistics.com/multiple-regression/unbalanced-factorial-anova/
Zhu CJ, Lee YK, Chao TM (1997) Effects of temperature and growth phase on lipid and biochemical composition of I. galbana TK1. J Appl Phycol 9(5):451–457. https://doi.org/10.1023/a:1007973319348
Funding
This work was supported by the NASA (grant number 15-EPSCoR2015-0026).
Author information
Authors and Affiliations
Contributions
XL conceived the research and designed the experiments. XL performed the experiments, analyzed, and interpreted the data. XL wrote the original draft. TWJ and DC: funding acquisition, supervised and edited the manuscript. SS analyzed and interpreted the data, and edited the manuscript. JM assisted the experimented and edited the manuscript. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics statement
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PDF 195 kb).
Rights and permissions
About this article
Cite this article
Li, X., Manuel, J., Slavens, S. et al. Interactive effects of light quality and culturing temperature on algal cell size, biomass doubling time, protein content, and carbohydrate content. Appl Microbiol Biotechnol 105, 587–597 (2021). https://doi.org/10.1007/s00253-020-11068-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-11068-y