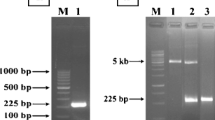
Abstract
Sakacin P, a bacteriocin from Lactobacillus sakei, shows strong activity against food-borne pathogens such as Listeria monocytogenes. In L. sakei, the structural gene (sppA) encoding sakacin P is controlled by a strict regulatory mechanism, and the quantity of secreted sakacin P is limited. In this study, the sppA gene was synthesized by splicing overlap extension PCR and cloned into Escherichia coli. After the induction with isopropyl-β-d-thiogalactopyranoside, the recombinant sakacin P was successfully expressed. The collected cells were sonicated, and the activity was detected by agar diffusion method. The results also showed that the low-temperature induction can improve the activity of sakacin P.







Similar content being viewed by others
References
Baneyx F, Mujacic M (2004) Recombinant protein folding and misfolding in Escherichia coli. Nat Biotechnol 22(11):1399–1408
BD Diagnostic Systems (2003) Difco & BBL manual: manual of microbiological culture media, 11th edn. BD Diagnostic Systems, Franklin Lakes, pp 150–152
Beaulieu L, Groleau D, Miguez CB, Jetté JF, Aomari H, Subirade M (2005) Production of pediocin PA-1 in the methylotrophic yeast Pichia pastoris reveals unexpected inhibition of its biological activity due to the presence of collagen-like material. Protein Expr Purif 43(2):111–125
Biet F, Berjeaud JM, Worobo RW, Cenatiempo Y, Fremaux C (1998) Heterologous expression of the bacteriocin mesentericin Y105 using the dedicated transport system and the general secretion pathway. Microbiology 144(Pt10):2845–2854
Drider D, Fimland G, Héchard Y, McMullen LM, Prévost H (2006) The continuing story of class IIa bacteriocins. Microbiol Mol Biol R 70(2):564–582
Georgopoulos A (1978) A simple micro agar diffusion method for the determination of antibiotic concentrations in blood and other body fluids. Zentralbl Bakteriol Orig A 242(3):387–393
Ghrairi T, Frère J, Berjeaud JM, Manai M (2005) Lactococcin MMT24, a novel two-peptide bacteriocin produced by Lactococcus lactis isolated from rigouta cheese. Int J Food Microbiol 105(3):389–398
Gutiérrez J, Criado R, Citti R, Martín M, Herranz C, Nes IF, Cintas LM, Hernández PE (2005) Cloning, production and functional expression of enterocin P, a sec-dependent bacteriocin produced by Enterococcus faecium P13, in Escherichia coli. Int J Food Microbiol 103(3):239–250
Horton RM, Cai ZL, Ho SN, Pease LR (1990) Gene splicing by overlap extension: tailor-made genes using the polymerase chain reaction. Biotechniques 8(5):528–535
Hühne K, Axelsson L, Holck A, Kröckel L (1996) Analysis of the sakacin P gene cluster from Lactobacillus sake Lb674 and its expression in sakacin-negative Lb. sake strains. Microbiology 142(Pt 6):1437–1448
Katla T, Møretrø T, Sveen I, Aasen IM, Axelsson L, Rørvik LM, Naterstad K (2002) Inhibition of Listeria monocytogenes in chicken cold cuts by addition of sakacin P and sakacin P-producing Lactobacillus sakei. J Appl Microbiol 93(2):191–196
Kawai Y, Arakawa K, Itoh A, Saitoh B, Ishiii Y, Nishimura J, Kitazawa H, Itoh T, Saito T (2003) Heterologous expression of gassericin A, a bacteriocin produced by Lactobacillus gasseri LA39. Anim Sci J 74:45–51
Klaenhammer TR (1993) Genetics of bacteriocins produced by lactic acid bacteria. FEMS Microbiol Rev 12(1–3):39–85
Mathiesen G, Huehne K, Kroeckel L, Axelsson L, Eijsink VG (2005) Characterization of a new bacteriocin operon in sakacin P-producing Lactobacillus sakei, showing strong translational coupling between the bacteriocin and immunity genes. Appl Environ Microbiol 71(7):3565–3574
Millette M, Dupont C, Archambault D, Lacroix M (2007) Partial characterization of bacteriocins produced by human Lactococcus lactis and Pediococccus acidilactici isolates. J Appl Microbiol 102(1):274–282
Moon GS, Pyun YR, Kim WJ (2006) Expression and purification of a fusion-typed pediocin PA-1 in Escherichia coli and recovery of biologically active pediocin PA-1. Int J Food Microbiol 108(1):136–140
Møretrø T, Naterstad K, Wang E, Aasen IM, Chaillou S, Zagorec M, Axelsson L (2005) Sakacin P non-producing Lactobacillus sakei strains contain homologues of the sakacin P gene cluster. Res Microbiol 156(9):949–960
Moretro T, Aasen IM, Storro I, Axelsson L (2000) Production of sakacin P by Lactobacillus sakei in a completely defined medium. J Appl Microbiol 88(3):536–545
Nes IF, Holo H (2000) Class II antimicrobial peptides from lactic acid bacteria. Biopolymers 55(1):50–61
Nigutová K, Serencová L, Piknová M, Javorský P, Pristas P (2008) Heterologous expression of functionally active enterolysin A, class III bacteriocin from Enterococcus faecalis, in Escherichia coli. Protein Expr Purif 60(1):20–24
Peng L, Xu Z, Fang X, Wang F, Cen P (2004) High-level expression of soluble human β-defensin-2 in Escherichia coli. Process Biochem 39(12):2199–2205
Richard C, Drider D, Elmorjani K, Marion D, Prévost H (2004) Heterologous expression and purification of active divercin V41, a class IIa bacteriocin encoded by a synthetic gene in Escherichia coli. J Bacteriol 186(13):4276–4284
Rodríguez JM, Martínez MI, Horn N, Dodd HM (2003) Heterologous production of bacteriocins by lactic acid bacteria. Int J Food Microbiol 80(2):101–116
Sakayori Y, Muramatsu M, Hanada S, Kamagata Y, Kawamoto S, Shima J (2003) Characterization of Enterococcus faecium mutants resistant to mundticin KS, a class IIa bacteriocin. Microbiology 149(Pt 10):2901–2908
Sambrook J, Russell D (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbor Laboratory Press, New York
Schägger H, Jagow G (1987) Tricine-sodium dodecyl sulphate-polyacrylamide gel electrophoresis for the separation of proteins in the range from 1 to 100 kDa. Anal Biochem 166(2):368–379
Shlyapnikov YM, Andreev YA, Kozlov SA, Vassilevski AA (2008) Bacterial production of latarcin 2a, a potent antimicrobial peptide from spider venom. Protein Expr Purif 60(1):89–95
Tichaczek PS, Vogel RF, Hammes WP (1994) Cloning and sequencing of sakP encoding sakacin P, the bacteriocin produced by Lactobacillus sake LTH 673. Microbiology 140(Pt 2):361–367
Urso R, Rantsiou K, Cantoni C, Comi G, Cocolin L (2006) Sequencing and expression analysis of the sakacin P bacteriocin produced by a Lactobacillus sakei strain isolated from naturally fermented sausages. Appl Microbiol Biotechnol 71(4):480–485
Van Reenen VA, Chikindas ML, Van Zyl WH, Dicks MT (2003) Characterization and heterologous expression of class IIa bacteriocin, plantaricin 423 from Lactobacillus plantarum 423, in Saccharomyces cerevisiae. Int J Food Microbiol 81(1):29–40
Vaughan A, Eijsink VG, Sinderen D (2003) Functional characterization of a composite bacteriocin locus from malt isolate Lactobacillus sakei 5. Appl Environ Microbiol 69(12):7194–7203
Young L, Dong Q (2004) Two-step total gene synthesis method. Nucleic Acids Res 32(7):e59
Acknowledgments
This work was financially supported by the National Natural Science Foundation of Chinese (No. 20706023), National Science Fund for Distinguished Young Scholars (31125021), Research Program of State Key Laboratory of Food Science and Technology (SKLFMB-200802), Fundamental Research Funds for the Central Universities (JUSRP11017 and JUSRP31002), and Innovative Research Team in University (IRT0627).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, H., Tian, F., Li, S. et al. Cloning and heterologous expression of a bacteriocin sakacin P from Lactobacillus sakei in Escherichia coli . Appl Microbiol Biotechnol 94, 1061–1068 (2012). https://doi.org/10.1007/s00253-012-3872-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-3872-z