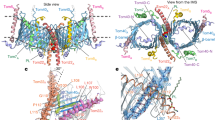
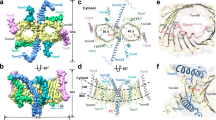
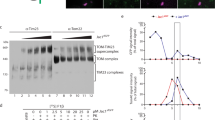
Abstract
Tom40, the central component of the preprotein translocase of the mitochondrial outer membrane (TOM complex), forms the import pore that facilitates the translocation of preproteins across the outer membrane. Though the function of Tom40 has been intensively studied, the details of the interactions between presequence peptides and Tom40 remain unclear. In this study, we expressed rat Tom40 in Escherichia coli and purified it from inclusion bodies before investigating the refolded protein by fluorescence spectroscopy and circular dichroism (CD) spectroscopy. The far-UV CD spectra of the refolded Tom40 in various concentrations of urea revealed that the refolded protein has a well-defined structure consisting mainly of β-sheet. Moreover, the specific binding of presequence peptides to Tom40, which was demonstrated by fluorescence quenching, showed that the refolded purified protein is functional and that the interaction between Tom40 and presequence peptides is mainly electrostatic in nature.




Similar content being viewed by others
References
Ahting U, Thun C, Hegerl R, Typke D, Nargang FE, Neupert W, Nussberger S (1999) The TOM core complex: the general protein import pore of the outer membrane of mitochondria. J Cell Biol 147:959–968. https://doi.org/10.1083/jcb.147.5.959
Bay DC, Hafez M, Young MJ, Court DA (2012) Phylogenetic and coevolutionary analysis of the β-barrel protein family comprised of mitochondrial porin (VDAC) and Tom40. Biochim Biophys Acta 1818:1502–1519. https://doi.org/10.1016/j.bbamem.2011.11.027
Braun P, von Heijne G (1999) The aromatic residues Trp and Phe have different effects on the positioning of a transmembrane helix in the microsomal membrane. Biochemistry 38:9778–9782. https://doi.org/10.1021/bi990923a
Dekker PJ, Ryan MT, Brix J, Müller H, Hönlinger A, Pfanner N (1998) Preprotein translocase of the outer mitochondrial membrane: molecular dissection and assembly of the general import pore complex. Mol Cell Biol 18:6515–6524. https://doi.org/10.1128/MCB.18.11.6515
dePlanque MR, Goormaghtigh E, Greathouse DV, Koeppe RE, Kruijtzer JA, Liskamp RM, deKruijff B, Killian JA (2001) Sensitivity of single membrane-spanning a-helical peptides to hydrophobic mismatch with a lipid bilayer: effects on backbone structure, orientation and extent of membrane incorporation. Biochemistry 40:5000–5010. https://doi.org/10.1021/bi000804r
Eftink MR (1994) The use of fluorescence methods to monitor unfolding transitions in proteins. Biophys J 66:482–501. https://doi.org/10.1016/S0006-3495(94)80799-4
Eftink MR, Ghiron CA (1981) Fluorescence quenching studies with proteins. Anal Biochem 114:199–227. https://doi.org/10.1016/0003-2697(81)90474-7
Feng W, Zhang Y, Deng H, Li SJ (2016) Interaction of divalent metal ions with human translocase of inner membrane of mitochondria Tim23. Biochem Biophys Res Commun 475(1):76–80. https://doi.org/10.1016/j.bbrc.2016.05.039
Gabriel K, Egan B, Lithgow T (2003) Tom40, The import channel of the mitochondrial outer membrane, plays an active role in sorting imported proteins. EMBO J 22:2380–2386. https://doi.org/10.1093/emboj/cdg229
Geissler A, Chacinska A, Truscott KN, Wiedemann N, Brandner K, Sickmann A, Meyer HE, Meisinger C, Pfanner N, Rehling P (2002) The mitochondrial presequence translocase: an essential role of Tim50 in directing preproteins to the import channel. Cell 111:507–518. https://doi.org/10.1016/S0092-8674(02)01073-5
Gessmann D, Flinner N, Pfannstiel J, Schlösinger A, Schleiff E, Nussberger S, Mirus O (2011) Structural elements of the mitochondrial preprotein-conducting channel Tom40 dissolved by bioinformatics and mass spectrometry. Biochim Biophys Acta 1807:1647–1657. https://doi.org/10.1016/j.bbabio.2011.08.006
Hill K, Model K, Ryan MT, Dietmeier K, Martin F, Wagner R, Pfanner N (1998) Tom40 forms the hydrophilic channel of the mitochondrial import pore for preproteins. Nature 395:516–521. https://doi.org/10.1038/26780
Johnson WC Jr (1988) Secondary structure of proteins through circular dichroism spectroscopy. Ann Rev Biophys Biophys Chem 17:145–166. https://doi.org/10.1146/annurev.bb.17.060188.001045
Jones BE, Beechem JM, Matthews CR (1995) Local and global dynamics during the folding of Escherichia coli dihydrofolate reductase by time-resolved fluorescence spectroscopy. Biochemistry 34:1867–1877. https://doi.org/10.1021/bi00006a007
Kanamori T, Nishikawa S, Shin I, Schultz PG, Endo T (1997) Probing the environment along the protein import pathways in yeast mitochondria by site-specific photo crosslinking. Proc Natl Acad Sci USA 94:485–490
Kuszak AJ, Jacobs D, Gurnev PA, Shiota T, Louis JM, Lithgow T, Bezrukov SM, Rostovtseva TK, Buchanan SK (2015) Evidence of distinct channel conformations and substrate binding affinities for the mitochondrial outer membrane protein translocase pore Tom40. J Biol Chem 290(43):26204–26217. https://doi.org/10.1074/jbc.M115.642173
Lackey SW, Taylor RD, Go NE, Wong A, Sherman EL, Nargang FE (2014) Evidence supporting the 19 β-strand model for Tom40 from cysteine scanning and protease site accessibility studies. J Biol Chem 289(31):21640–21650. https://doi.org/10.1074/jbc.M114.578765
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. https://doi.org/10.1038/227680a0
Lakowicz JR (2006) Quenching of fluorescence. Principles of fluorescence spectroscopy, vol 3. Springer, Boston, pp 288–289
Lakowicz JR, Weber G (1973) Quenching of fluorescence by oxygen. A probe for structural fluctuations in macromolecules. Biochemistry 12:4161–4170. https://doi.org/10.1021/bi00745a021
Li SJ, Kawazaki M, Ogasahara K, Nakagawa A (2006) The intracellular region of ClC-3 chloride channel is in a partially folded state and a monomer. J Biochem 139:813–820. https://doi.org/10.1093/jb/mvj099
Manavalan P, Johnson WC Jr (1983) Sensitivity circular dichroism to protein tertiary structure class. Nature 305:831–832. https://doi.org/10.1038/305831a0
Pfanner N, Wiedemann N, Meisinger C, Lithgow T (2004) Assembling the mitochondrial outer membrane. Nat Struct Mol Biol 11:1044–1048. https://doi.org/10.1038/nsmb852
Pusnik M, Charrière F, Mäser P, Waller RF, Dagley MJ, Lithgow T, Schneider A (2009) The single mitochondrial porin of Trypanosoma brucei is the main metabolite transporter in the outer mitochondrial membrane. Mol Biol Evol 26:671–680. https://doi.org/10.1093/molbev/msn288
Rapaport D, Neupert W, Lill R (1997) Mitochondrial protein import. Tom40 plays a major role in targeting and translocation of preproteins by forming a specific binding site for the presequence. J Biol Chem 272:18725–18731. https://doi.org/10.1074/jbc.272.30.18725
Rapaport D, Mayer A, Neupert W, Lill R (1998) Cis and trans sites of the TOM complex of mitochondria in unfolding and initial translocation of preproteins. J Biol Chem 273:8806–8813. https://doi.org/10.1074/jbc.273.15.8806
Reithmeier RA (1995) Characterization and modeling of membrane proteins using sequence analysis. Curr Opin Struct Biol 5:491–500. https://doi.org/10.1016/0959-440X(95)80034-4
Ross PD, Subramanian S (1981) Thermodynamics of protein association reactions: forces contributing to stability. Biochemistry 20:3096–3102. https://doi.org/10.1021/bi00514a017
Ryan MT, Wagner R, Pfanner N (2000) The transport machinery for the import of preproteins across the outer mitochondrial membrane. Int J Biochem Cell Biol 32:13–21. https://doi.org/10.1016/S1357-2725(99)00114-4
Suzuki H, Okazawa Y, Komiya T, Saeki K, Mekada E, Kitada S, Ito A, Mihara K (2000) Characterization of rat TOM40, a central component of the preprotein translocase of the mitochondrial outer membrane. J Biol Chem 275:37930–37936. https://doi.org/10.1074/jbc.M006558200
Ware WR (1962) Oxygen quenching solution: an experimental study discussion process. J Phys Chem 66:455–458. https://doi.org/10.1021/j100809a020
Yuen CT, Davidson AR, Deber CM (2000) Role of aromatic residues at the lipid-water interface in micelle-bound bacteriophage M13 major coat protein. Biochemistry 39:16155–16162. https://doi.org/10.1021/bi0016117
Zeth K (2010) Structure and evolution of mitochondrial outermembrane proteins of β-barrel topology. Biochim Biophys Acta 1797:1292–1299. https://doi.org/10.1016/j.bbabio.2010.04.019
Zhang Y, Xu Y, Zhao Q, Ji Z, Li Q, Li SJ (2011) Expression and structural characterization of human translocase of inner membrane of mitochondria Tim50. Protein Expr Purif 80(1):130–137. https://doi.org/10.1016/j.pep.2011.06.012
Zhang Y, Deng H, Zhang X, Li SJ (2012a) Interaction of divalent metal ions with human translocase of inner membrane of mitochondria Tim50. Biochem Biophys Res Commun 428:365–370. https://doi.org/10.1016/j.bbrc.2012.10.060
Zhang Y, Deng H, Zhao Q, Li SJ (2012b) Interaction of presequence with human translocase of the inner membrane of mitochondria Tim50. J Phys Chem B 116(9):2990–2998. https://doi.org/10.1021/jp2108279
Zhang Y, Xu Y, Zhao Q, Ji Z, Deng H, Li SJ (2012c) The structural characteristics of human preprotein translocase of the inner mitochondrial membrane Tim 23: implications for its physiological activities. Protein Expr Purif 82(2):255–262. https://doi.org/10.1016/j.pep.2012.01.009
Zhang Y, Deng H, Zhao Q, Li SJ (2013) Interaction of presequence peptides with human translocase of inner membrane of mitochondria Tim23. Biochem Biophys Res Commun 437(2):292–299. https://doi.org/10.1016/j.bbrc.2013.06.070
Acknowledgements
This work was supported by National Natural Science Foundation of China (No. 30970579, 31271464 and 21707165), and the Ph.D. Programs Foundation of Ministry of Education of China (No. 20110031110004 and 20120031110028), and the Basic Science and Advance Technology Research Program of Tianjin (No. 14JCYBJC23400).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Feng, W., Li, J., Liang, Y. et al. Structural characterisation of a full-length mitochondrial outer membrane TOM40 preprotein translocase: implications for its interaction with presequence peptides. Eur Biophys J 48, 35–43 (2019). https://doi.org/10.1007/s00249-018-1329-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-018-1329-8