Abstract
Objectives
Linezolid is a commonly used antibiotic in the clinical treatment of gram-positive bacterial infections. The impacts of drug interactions on the pharmacokinetics of linezolid are often overlooked. This manuscript aims to review the medications that affect the pharmacokinetics of linezolid.
Methods
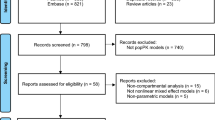
In accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, we queried the PubMed, Embase, and Cochrane Library for publications from database establishment to November 3, 2023, using the search terms: “Linezolid” and “interaction,” or “interact,” or “drug-drug interaction,” or “co-treatment,” or “cotreatment,” or “combined,” or “combination.”
Results
A total of 24 articles were included. Among the reported medication interactions, rifampicin, levothyroxine, venlafaxine, and phenobarbital could reduce the concentration of linezolid; clarithromycin, digoxin, cyclosporine, proton pump inhibitors, and amiodarone could increase the concentration of linezolid, while aztreonam, phenylpropanolamine, dextromethorphan, antioxidant vitamins, and magnesium-containing antacids had no significant effects on linezolid pharmacokinetics. The ratio of mean (ROM) of linezolid AUC in co-treatment with rifampicin to monotherapy was 0.67 (95%CI 0.58–0.77) and 0.63 (95%CI 0.43–0.91), respectively, in 2 studies, and co-treatment with 500 mg clarithromycin to monotherapy was 1.81 (95%CI 1.49–2.13).
Conclusions
This systematic review found that numerous drugs have an impact on the pharmacokinetics of linezolid, and the purported main mechanism may be that linezolid is the substrate of P-glycoprotein. In clinical practice, it is prudent to pay attention to the changes in linezolid pharmacokinetics caused by interactions. Conducting therapeutic drug monitoring (TDM) is beneficial to improve efficacy and reduce adverse reactions of linezolid.

Similar content being viewed by others
Data availability
All the data used for this publication is presented in the main article.
References
Stevens DL, Dotter B, Madaras-Kelly K (2004) A review of linezolid: the first oxazolidinone antibiotic. Expert Rev Anti Infect Ther 2(1):51–59
Linezolid injection drug label information. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=d05541c6-fd63-cc99-e053-2995a90a4ec7#. Accessed 10 May 2023
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:n71
Stang A (2010) Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 25(9):603–605
National Heart, Lung and Blood Institute. Quality assessment tool for case series studies. https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools. Accessed on 19 Aug 2023
Jonathan Sterne JH, Reeves B, Savovi´c J, Turner L. RoB 2 for crossover trials. https://www.riskofbias.info/welcome/rob-2-0-tool/rob-2-for-crossover-trials. Accessed on 21 Aug 2023
Sisson TL, Jungbluth GL, Hopkins NK (1999) A pharmacokinetic evaluation of concomitant administration of linezolid and aztreonam. J Clin Pharmacol 39(12):1277–1282
Hendershot PE, Antal EJ, Welshman IR, Batts DH, Hopkins NK (2001) Linezolid: pharmacokinetic and pharmacodynamic evaluation of coadministration with pseudoephedrine HCl, phenylpropanolamine HCl, and dextromethorpan HBr. J Clin Pharmacol 41(5):563–572
Grunder G, Zysset-Aschmann Y, Vollenweider F, Maier T, Krähenbühl S, Drewe J (2006) Lack of pharmacokinetic interaction between linezolid and antacid in healthy volunteers. Antimicrob Agents Chemother 50(1):68–72
Hoyo I, Martínez-Pastor J, Garcia-Ramiro S, Climent C, Brunet M, Cuesta M, Mensa J, Soriano A (2012) Decreased serum linezolid concentrations in two patients receiving linezolid and rifampicin due to bone infections. Scand J Infect Dis 44(7):548–550
Cojutti P, Maximova N, Crichiutti G, Isola M, Pea F (2015) Pharmacokinetic/pharmacodynamic evaluation of linezolid in hospitalized paediatric patients: a step toward dose optimization by means of therapeutic drug monitoring and Monte Carlo simulation. J Antimicrob Chemother 70(1):198–206
Blassmann U, Roehr AC, Frey OR, Koeberer A, Briegel J, Huge V, Vetter-Kerkhoff C (2016) Decreased linezolid serum concentrations in three critically ill patients: clinical case studies of a potential drug interaction between linezolid and rifampicin. Pharmacology 98(1–2):51–55
Pai MP, Cojutti PG, Gerussi V, Della Siega P, Tascini C, Pea F (2022) Linezolid population pharmacokinetics to improve dosing in cardiosurgical patients: factoring a new drug-drug interaction pathway. Clin Infect Dis 25:ciac917
Abdelgawad N, Wasserman S, Abdelwahab MT, Davis A, Stek C, Wiesner L, Black J, Meintjes G, Wilkinson RJ, Denti P (2023) Linezolid population pharmacokinetic model in plasma and cerebrospinal fluid among patients with tuberculosis meningitis. J Infect Dis 22:jiad413
Bock M, Van Hasselt JGC, Schwartz F, Wang H, Høiby N, Fuursted K, Ihlemann N, Gill S, Christiansen U, Bruun NE, Elming H, Povlsen JA, Køber L, Høfsten DE, Fosbøl EL, Pries-Heje MM, Christensen JJ, Rosenvinge FS, Torp-Pedersen C, Helweg-Larsen J, Tønder N, Iversen K, Bundgaard H, Moser C (2023) Rifampicin reduces plasma concentration of linezolid in patients with infective endocarditis. J Antimicrob Chemother 12:dkad316
Gordi T, Tan LH, Hong C et al (2003) The pharmacokinetics of linezolid are not affected by concomitant intake of the antioxidant vitamins C and E. J Clin Pharmacol 43(10):1161–1167
Gandelman K, Zhu T, Fahmi OA, Glue P, Lian K (2011) Obach RS. Damle B -J Clin Pharmacol 51(2):229–236
Pea F, Viale P, Cojutti P, Del Pin B, Zamparini E, Furlanut M (2012) Therapeutic drug monitoring may improve safety outcomes of long-term treatment with linezolid in adult patients. J Antimicrob Chemother 67(8):2034–2042
Bolhuis MS, van Altena R, van Soolingen D et al (2013) Clarithromycin increases linezolid exposure in multidrug-resistant tuberculosis patients. Eur Respir J 42(6):1614–1621
Hashimoto S, Honda K, Fujita K, Miyachi Y, Isoda K, Misaka K, Suga Y, Kato S, Tsuchiya H, Kato Y, Okajima M, Taniguchi T, Shimada T, Sai Y (2018) Effect of coadministration of rifampicin on the pharmacokinetics of linezolid: clinical and animal studies. J Pharm Health Care Sci 12(4):27
Puplà Bartoll A, Bellés Medall MD, Pérez Catalán IE, Roig Martí C, Bodega Azuara J, Gil Candel M, Esteve López V, Ferrando PR (2023) Ajuste posológico de linezolid por interacción con rifampicina en endocarditis infecciosa [Dose adjustment of linezolid due to interaction with rifampicin in infective endocarditis]. Rev Esp Quimioter 36(4):436–438
Egle H, Trittler R, Kümmerer K, Lemmen SW (2005) Linezolid and rifampin: drug interaction contrary to expectations? Clin Pharmacol Ther 77(5):451–453
Gebhart BC, Barker BC, Markewitz BA (2007) Decreased serum linezolid levels in a critically ill patient receiving concomitant linezolid and rifampin. Pharmacotherapy 27(3):476–479
Pea F, Furlanut M, Cojutti P et al (2010) Therapeutic drug monitoring of linezolid: a retrospective monocentric analysis. Antimicrob Agents Chemother 54(11):4605–4610
Bolhuis MS, van Altena R, Uges DR, van der Werf TS, Kosterink JG, Alffenaar JW (2010) Clarithromycin significantly increases linezolid serum concentrations. Antimicrob Agents Chemother 54(12):5418–5419
Pea F, Cadeo B, Cojutti PG, Pecori D, Bassetti M (2014) Linezolid underexposure in a hypothyroid patient on levothyroxine replacement therapy: a case report. Ther Drug Monit 36(5):687–689
Cojutti P, Crapis M, Bassetti M, Hope W, Pea F (2015) Linezolid underexposure in a patient co-treated with venlafaxine. Eur J Clin Pharmacol 71(10):1285–1286
Ashizawa N, Tsuji Y, Kawago K, Higashi Y, Tashiro M, Nogami M, Gejo R, Narukawa M, Kimura T, Yamamoto Y (2016) Successful treatment of methicillin-resistant Staphylococcus aureus osteomyelitis with combination therapy using linezolid and rifampicin under therapeutic drug monitoring. J Infect Chemother 22(5):331–334
Töpper C, Steinbach CL, Dorn C, Kratzer A, Wicha SG, Schleibinger M, Liebchen U, Kees F, Salzberger B, Kees MG (2016) Variable linezolid exposure in intensive care unit patients-possible role of drug-drug interactions. Ther Drug Monit 38(5):573–578
Yulin Z, Lingti K, Shan G, Yong Z (2020) A possible interaction between linezolid and digoxin: a case report of therapeutic drug monitoring. Saudi Pharm J 28(11):1408–1410
Friedrich JO, Adhikari NK, Beyene J (2011) Ratio of means for analyzing continuous outcomes in meta-analysis performed as well as mean difference methods. J Clin Epidemiol 64(5):556–564
Estimating the sample mean and standard deviation (SD) from the five-number summary and its application in meta-analysis. https://www.math.hkbu.edu.hk/~tongt/papers/median2mean.html. Accessed 23 Aug 2023
Allegra S, Di Paolo A, Cusato J, Fatiguso G, Arrigoni E, Danesi R, Corcione S, DʼAvolio A (2019) Different underlying mechanism might explain the absence of a significant difference in area under the concentration-time curve of linezolid for different ABCB1 genotypes. Ther Drug Monit 41(2):254–255
Siegmund W, Altmannsberger S, Paneitz A, Hecker U, Zschiesche M, Franke G, Meng W, Warzok R, Schroeder E, Sperker B, Terhaag B, Cascorbi I, Kroemer HK (2002) Effect of levothyroxine administration on intestinal P-glycoprotein expression: consequences for drug disposition. Clin Pharmacol Ther 72(3):256–264
Levin GM, Nelson LA, DeVane CL, Preston SL, Eisele G, Carson SW (2001) A pharmacokinetic drug-drug interaction study of venlafaxine and indinavir. Psychopharmacol Bull 35(2):62–71
Eberl S, Renner B, Neubert A, Reisig M, Bachmakov I, König J, Dörje F, Mürdter TE, Ackermann A, Dormann H, Gassmann KG, Hahn EG, Zierhut S, Brune K, Fromm MF (2007) Role of p-glycoprotein inhibition for drug interactions: evidence from in vitro and pharmacoepidemiological studies. Clin Pharmacokinet 46(12):1039–1049
Fenner KS, Troutman MD, Kempshall S et al (2009) Drug-drug interactions mediated through P-glycoprotein: clinical relevance and in vitro-in vivo correlation using digoxin as a probe drug. Clin Pharmacol Ther 85(2):173–181
Katoh M, Nakajima M, Yamazaki H, Yokoi T (2001) Inhibitory effects of CYP3A4 substrates and their metabolites on P-glycoprotein-mediated transport. Eur J Pharm Sci 12(4):505–513
Pauli-Magnus C, Rekersbrink S, Klotz U, Fromm MF (2001) Interaction of omeprazole, lansoprazole and pantoprazole with P-glycoprotein. Naunyn Schmiedebergs Arch Pharmacol 364(6):551–557
Kono Y, Kawahara I, Shinozaki K, Nomura I, Marutani H, Yamamoto A, Fujita T (2021) Characterization of P-glycoprotein inhibitors for evaluating the effect of P-glycoprotein on the intestinal absorption of drugs. Pharmaceutics 13(3):388
Juliano RL, Ling V (1976) A surface glycoprotein modulating drug permeability in Chinese hamster ovary cell mutants. Biochim Biophys Acta 455(1):152–162
Ling V (1997) Multidrug resistance: molecular mechanisms and clinical relevance. Cancer Chemother Pharmacol 40(Suppl):S3–S8
Gottesman MM, Pastan I, Ambudkar SV (1996) P-glycoprotein and multidrug resistance. Curr Opin Genet Dev 6:610–617
Taubert M, Zander J, Frechen S, Scharf C, Frey L, Vogeser M, Fuhr U, Zoller M (2017) Optimization of linezolid therapy in the critically ill: the effect of adjusted infusion regimens. J Antimicrob Chemother 72(8):2304–2310
Collett A, Tanianis-Hughes J, Warhurst G (2004) Rapid induction of P-glycoprotein expression by high permeability compounds in colonic cells in vitro: a possible source of transporter mediated drug interactions? Biochem Pharmacol 68:783–790
Cheli S, Fusi M, De Silvestri A, Bonini I, Clementi E, Cattaneo D, Montrasio C, Baldelli S (2021) In linezolid underexposure, pharmacogenetics matters: the role of CYP3A5. Biomed Pharmacother 139:111631
Obach RS (2022) Linezolid metabolism is catalyzed by cytochrome P450 2J2, 4F2, and 1B1. Drug Metab Dispos 50(4):413–421
Author information
Authors and Affiliations
Contributions
Qiang Xu and Lu Li designed the study. Qiang Xu, Yanlei Sang, and Anna Gao investigated the reviews and data. Qiang Xu wrote the original draft; Lu Li reviewed and edited the manuscript. All authors agreed to submit the final version to the journal and agreed to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This systematic review was registered in PROSPERO (CRD42023453429) on 21/08/2023.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, Q., Sang, Y., Gao, A. et al. The effects of drug-drug interaction on linezolid pharmacokinetics: A systematic review. Eur J Clin Pharmacol (2024). https://doi.org/10.1007/s00228-024-03652-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00228-024-03652-2