Abstract
Purpose
Combined intravenous and intraventricular administration of vancomycin into the cerebrospinal fluid (CSF) has been increasingly utilized for neurosurgical patients, but little is known about the population pharmacokinetics of vancomycin in the plasma and CSF. The aim of our study was to identify significant factors associated with plasma and CSF vancomycin concentrations to guide clinicians with vancomycin dosing.
Methods
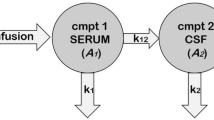
Patients with an indwelling ventricular drainage catheter who received intravenous and intraventricular vancomycin were enrolled in this study. Blood and CSF samples were collected at scheduled times and vancomycin concentrations determined. A three-compartmental model (central, peripheral and CSF compartments) was proposed to describe the in vivo behavior of vancomycin. CSF outflow resulted in vancomycin loss, and the clearance of CSF compartment (CLCSF) was used to describe this loss. The nonlinear mixed-effects modeling method was applied to structure the population model, and the stepwise incorporation of seven covariates into the final model was attempted. Simulation was performed with the goal of CSF concentrations reaching or exceeding the minimum inhibitory concentration during therapy.
Results
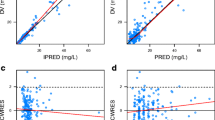
Serum creatinine clearance had a significant influence on clearance of the central compartment. CLCSF had a positive correlation with drainage amount and a negative correlation with elapsed time. Model validation (bootstrap and visual predictive check) demonstrated the stability and performance of the proposed population model. A simple-to-use dosage regimen table was created based on the simulation results.
Conclusions
The proposed final model may be used to guide clinicians with vancomycin dosing in this specific patient population.



Similar content being viewed by others
References
Rybak M, Lomaestro B, Rotschafer JC, Moellering R Jr, Craig W, Billeter M, Dalovisio JR, Levine DP (2009) Therapeutic monitoring of vancomycin in adult patients: A consensus review of the American Society of Health-System Pharmacists, the Infectious Diseases Society of America, and the Society of Infectious Diseases Pharmacists. Am J Health Syst Pharm 66:82–98. doi:10.2146/ajhp080434
Tunkel AR, Hartman BJ, Kaplan SL, Kaufman BA, Roos KL, Scheld WM, Whitley RJ (2004) Practice guidelines for the management of bacterial meningitis. Clin Infect Dis 39:1267–1284. doi:10.1086/425368
Martin JH, Norris R, Barras M, Roberts J, Morris R, Doogue M, Jones GR (2010) Therapeutic monitoring of vancomycin in adult patients: A consensus review of the American Society of Health-System Pharmacists, the Infectious Diseases Society of America, and the Society of Infectious Diseases Pharmacists. Clin Biochem Rev 31:21–24
Wang Q, Shi Z, Wang J, Shi G, Wang S, Zhou J (2008) Postoperatively administered vancomycin reaches therapeutic concentration in the cerebral spinal fluid of neurosurgical patients. Surg Neurol 69:126–129; discussion 129. doi:10.1016/j.surneu.2007.01.073
Li X, Wu Y, Sun S, Mei S, Wang J, Wang Q, Zhao Z (2015) Population pharmacokinetics of vancomycin in postoperative neurosurgical patients. J Pharm Sci 104:3960–3967. doi:10.1002/jps.24604
Li X, Wu Y, Sun S, Zhao Z, Wang Q (2016) Population pharmacokinetics of vancomycin in postoperative neurosurgical patients and the application in dosing recommendation. J Pharm Sci 105:3425–3431. doi:10.1016/j.xphs.2016.08.012
Al-Jeraisy M, Phelps SJ, Christensen ML, Einhaus S (2004) Intraventricular vancomycin in pediatric patients with cerebrospinal fluid shunt infections. J Pediatr Pharmacol Ther 9:36–42. doi:10.5863/1551-6776-9.1.36
Remes F, Tomas R, Jindrak V, Vanis V, Setlik M (2013) Intraventricular and lumbar intrathecal administration of antibiotics in postneurosurgical patients with meningitis and/or ventriculitis in a serious clinical state. J Neurosurg 119:1596–1602. doi:10.3171/2013.6.JNS122126
Chen K, Wu Y, Wang Q, Wang J, Li X, Zhao Z, Zhou J (2015) The methodology and pharmacokinetics study of intraventricular administration of vancomycin in patients with intracranial infections after craniotomy. J Crit Care 30(218):e211–e215. doi:10.1016/j.jcrc.2014.09.020
Matsunaga N, Hisata K, Shimizu T (2015) An investigation into the vancomycin concentration in the cerebrospinal fluid due to vancomycin intraventricular administration in newborns: A study of 13 cases. Medicine (Baltimore) 94:e922. doi:10.1097/MD.0000000000000922
Popa D, Loewenstein L, Lam SW, Neuner EA, Ahrens CL, Bhimraj A (2016) Therapeutic drug monitoring of cerebrospinal fluid vancomycin concentration during intraventricular administration. J Hosp Infect 92:199–202. doi:10.1016/j.jhin.2015.10.017
Lin WW, Wu W, Jiao Z, Lin RF, Jiang CZ, Huang PF, Liu YW, Wang CL (2016) Population pharmacokinetics of vancomycin in adult Chinese patients with post-craniotomy meningitis and its application in individualised dosage regimens. Eur J Clin Pharmacol 72:29–37. doi:10.1007/s00228-015-1952-6
Donadello K, Roberts JA, Cristallini S, Beumier M, Shekar K, Jacobs F, Belhaj A, Vincent JL, de Backer D, Taccone FS (2014) Vancomycin population pharmacokinetics during extracorporeal membrane oxygenation therapy: A matched cohort study. Crit Care 18:632. doi:10.1186/s13054-014-0632-8
Guilhaumou R, Marsot A, Dupouey J, Galambrun C, Boulamery A, Coze C, Simon N, Andre N (2016) Pediatric patients with solid or hematological tumor disease: Vancomycin population pharmacokinetics and dosage optimization. Ther Drug Monit 38:559–566. doi:10.1097/FTD.0000000000000318
Wu Y, Kang J, Wang Q (2017) Drug concentrations in the serum and cerebrospinal fluid of patients treated with norvancomycin after craniotomy. Eur J Clin Microbiol Infect Dis 36:305–311. doi:10.1007/s10096-016-2803-9
Mangin O, Urien S, Mainardi JL, Fagon JY, Faisy C (2014) Vancomycin pharmacokinetic and pharmacodynamic models for critically ill patients with post-sternotomy mediastinitis. Clin Pharmacokinet 53:849–861. doi:10.1007/s40262-014-0164-z
Escobar L, Andresen M, Downey P, Gai MN, Regueira T, Borquez T, Lipman J, Roberts JA (2014) Population pharmacokinetics and dose simulation of vancomycin in critically ill patients during high-volume haemofiltration. Int J Antimicrob Agents 44:163–167. doi:10.1016/j.ijantimicag.2014.03.009
Thomson AH, Staatz CE, Tobin CM, Gall M, Lovering AM (2009) Development and evaluation of vancomycin dosage guidelines designed to achieve new target concentrations. J Antimicrob Chemother 63:1050–1057. doi:10.1093/jac/dkp085
Li LJ, Shang DW, Li WB, Guo W, Wang XP, Ren YP, Li AN, Fu PX, Ji SM, Lu W, Wang CY (2012) Population pharmacokinetics of clozapine and its primary metabolite norclozapine in Chinese patients with schizophrenia. Acta Pharmacol Sin 33:1409–1416. doi:10.1038/aps.2012.71
Byon W, Smith MK, Chan P, Tortorici MA, Riley S, Dai H, Dong J, Ruiz-Garcia A, Sweeney K, Cronenberger C (2013) Establishing best practices and guidance in population modeling: An experience with an internal population pharmacokinetic analysis guidance. CPT Pharmacometrics Syst Pharmacol 2:e51. doi:10.1038/psp.2013.26
Appleman MD, Citron DM (2010) Efficacy of vancomycin and daptomycin against Staphylococcus aureus isolates collected over 29 years. Diagn Microbiol Infect Dis 66:441–444. doi:10.1016/j.diagmicrobio.2009.11.008
Oyaert M, Spriet I, Allegaert K, Smits A, Vanstraelen K, Peersman N, Wauters J, Verhaegen J, Vermeersch P, Pauwels S (2015) Factors impacting unbound vancomycin concentrations in different patient populations. Antimicrob Agents Chemother 59:7073–7079. doi:10.1128/AAC.01185-15
Lu C, Zhang Y, Chen M, Zhong P, Chen Y, Yu J, Wu X, Wu J, Zhang J (2016) Population pharmacokinetics and dosing regimen optimization of Meropenem in cerebrospinal fluid and plasma in patients with meningitis after neurosurgery. Antimicrob Agents Chemother 60:6619–6625. doi:10.1128/AAC.00997-16
Mei S, Luo X, Li X, Li Q, Huo J, Yang L, Zhu L, Feng W, Zhou J, Shi G, Zhao Z (2016) Development and validation of an LC-MS/MS method for the determination of tigecycline in human plasma and cerebrospinal fluid and its application to a pharmacokinetic study. Biomed Chromatogr 30:1992–2002. doi:10.1002/bmc.3776
Lutsar I, McCracken GH Jr, Friedland IR (1998) Antibiotic pharmacodynamics in cerebrospinal fluid. Clin Infect Dis 27:1117–1127 quiz 1128-1119
Acknowledgements
The authors acknowledge Dr. Yuanxing Wu and Guangqiang Chen for their help with sample collection. This work was supported by grants from the National Natural Science Foundation of China (81503157), the Organization Department of Beijing Municipal Committee (2014000021469G258), and the Capital Medical University (16JL72).
Author information
Authors and Affiliations
Contributions
Q Wang and Z Zhao conceived and designed the study. X Li designed the statistical analysis and drafted the manuscript. X Li and S Sun performed the analysis and wrote the manuscript. X Ling and K Chen performed the experiments. Q Wang and Z Zhao supervised the quality of the study. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest to disclose.
Electronic supplementary material
ESM 1
(DOCX 23 kb).
Rights and permissions
About this article
Cite this article
Li, X., Sun, S., Ling, X. et al. Plasma and cerebrospinal fluid population pharmacokinetics of vancomycin in postoperative neurosurgical patients after combined intravenous and intraventricular administration. Eur J Clin Pharmacol 73, 1599–1607 (2017). https://doi.org/10.1007/s00228-017-2313-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-017-2313-4