Abstract
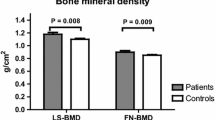
There is limited literature from India with regard to the prevalence and magnitude of renal tubular and bone manifestations in Wilson’s disease (WD). Thus, we studied the prevalence of renal tubular acidosis among Indian patients with WD and also evaluated bone health and body composition in them. It was a cross-sectional study conducted at a south Indian tertiary care center. Twenty-five consecutive patients with WD aged more than 12 years attending the hepatology and neurology departments and 50 age, sex and BMI-matched controls were recruited. After clinical assessment, they underwent biochemical testing to assess renal tubular dysfunction. Bone mineral density (BMD) and body composition were assessed using a dual energy X-ray absorptiometry (DXA) scanner. Fifty-six percent (14/25) of patients with WD had renal tubular acidosis (RTA). Of them, 24% were diagnosed to have distal RTA. RTA was more common in hepatic WD patients who had prolonged duration of illness. Patients with WD had significantly lower BMD as compared to control subjects (p < 0.05). Low BMI, low IGF-1 and a shorter duration of therapy were key determinants of low bone mass in them (p < 0.05). Patients with WD had significantly more body fat (p = 0.01) and lower lean muscle mass (p = 0.03) when compared to age, sex and BMI-matched controls. In conclusion, renal tubular acidosis was common in patients with Wilson’s disease. These patients had a lower bone mineral density, higher body fat percentage and lower lean muscle mass as compared to controls.
Similar content being viewed by others
References
Patil M, Sheth KA, Krishnamurthy AC, Devarbhavi H (2013) A review and current perspective on Wilson disease. J Clin Exp Hepatol 3(4):321–336
Tarnacka B, Rodo M, Cichy S, Członkowska A (2000) Procreation ability in Wilson’s disease. Acta Neurol Scand 101(6):395–398
Kapoor N, Chapla A, Furler J, Paul TV, Harrap S, Oldenburg B et al (2019) Genetics of obesity in consanguineous populations—a road map to provide novel insights in the molecular basis and management of obesity. EBioMedicine 40:33–34
Mamidala MP, Kalikiri MK, Praveen Kumar PTV, Rajesh N, Vallamkonda OR, Rajesh V (2015) Consanguinity in India and its association with autism spectrum disorder. Autism Res 8(2):224–228
Kelly C, Pericleous M (2018) Wilson disease: more than meets the eye. Postgrad Med J 94(1112):335–347
Weiss KH, Van de Moortele M, Gotthardt DN, Pfeiffenberger J, Seessle J, Ullrich E et al (2015) Bone demineralisation in a large cohort of Wilson disease patients. J Inherit Metab Dis 38(5):949–956
Domrongkitchaiporn S, Pongsakul C, Stitchantrakul W, Sirikulchayanonta V, Ongphiphadhanakul B, Radinahamed P et al (2001) Bone mineral density and histology in distal renal tubular acidosis. Kidney Int 59(3):1086–1093
Binu AJ, Cherian KE, Kapoor N, Thomas N, Paul TV (2018) Referral pattern for DXA scanning in a tertiary care centre from southern India. Arch Osteoporos 13(1):133
Ghosh L, Shah M, Pate S, Mannari J, Sharma K (2012) Wilson’s disease presenting with hypokalemia, hypoparathyroidism and renal failure. J Assoc Phys India 60:57–59
Roberts EA (2018) Update on the diagnosis and management of Wilson disease. Curr Gastroenterol Rep 20(12):56
Ala A, Walker AP, Ashkan K, Dooley JS, Schilsky ML (2007) Wilson’s disease. Lancet Lond Engl 369(9559):397–408
Marshall WA, Tanner JM (1969) Variations in pattern of pubertal changes in girls. Arch Dis Child 44(235):291–303
Marshall WA, Tanner JM (1970) Variations in the pattern of pubertal changes in boys. Arch Dis Child 45(239):13–23
Bagga A, Sinha A (2007) Evaluation of renal tubular acidosis. Indian J Pediatr 74(7):679–686
Walton RJ, Bijvoet OL (1975) Nomogram for derivation of renal threshold phosphate concentration. Lancet Lond Engl 2(7929):309–310
Teixeira S, Domingos J, Carvalho R, Miranda HP, Magalhaes M (2011) Prevalence of metabolic bone disease in Wilson. In: BioScientifica. https://www.endocrine-abstracts.org/ea/0026/ea0026p535. Accessed 19 Jan 2019
Krishnamoorthy S, Audinarayana N (2001) Trends in consanguinity in south India. J Biosoc Sci 33(2):185–197
Tesar V, Mokrejsová M, Marecek Z, Petrtýl J (1991) Distal renal tubular acidosis in patients with Wilson’s disease. Sb Lek 93(9–10):315–323
Dastur DK, Manghani DK, Wadia NH (1968) Wilson’s disease in India. I. Geographic, genetic, and clinical aspects in 16 families. Neurology 18(1):21–31
Boga S, Ala A, Schilsky ML (2017) Hepatic features of Wilson disease. Handb Clin Neurol 142:91–99
Santhosh S, Shaji RV, Eapen CE, Jayanthi V, Malathi S, Chandy M et al (2006) ATP7B mutations in families in a predominantly southern Indian cohort of Wilson’s disease patients. Indian J Gastroenterol 25(6):277–282
Reddy GS, Jones G, Kooh SW, Fraser D (1982) Inhibition of 25-hydroxyvitamin D3-1-hydroxylase by chronic metabolic acidosis. Am J Physiol 243(4):E265–E271
Palkar AV, Shrivastava MS, Padwal NJ, Padhiyar RN, Moulick N (2011) Renal tubular acidosis due to Wilson’s disease presenting as metabolic bone disease. BMJ Case Rep [Internet]. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3158360/. Accessed 19 Jan 2019
Kim CJ (2011) Vitamin D dependent rickets type I. Korean J Pediatr 54(2):51–54
Cunningham J, Fraher LJ, Clemens TL, Revell PA, Papapoulos SE (1982) Chronic acidosis with metabolic bone disease. Effect of alkali on bone morphology and vitamin D metabolism. Am J Med 73(2):199–204
Quemeneur A-S, Trocello J-M, Ea H-K, Ostertag A, Leyendecker A, Duclos-Vallée J-C et al (2014) Bone status and fractures in 85 adults with Wilson’s disease. Osteoporos Int 25(11):2573–2580
Chenbhanich J, Thongprayoon C, Atsawarungruangkit A, Phupitakphol T, Cheungpasitporn W (2018) Osteoporosis and bone mineral density in patients with Wilson’s disease: a systematic review and meta-analysis. Osteoporos Int 29(2):315–322
Handzlik-Orlik G, Holecki M, Wilczyński K, Duława J (2016) Osteoporosis in liver disease: pathogenesis and management. Ther Adv Endocrinol Metab 7(3):128–135
Selimoglu MA, Ertekin V, Doneray H, Yildirim M (2008) Bone mineral density of children with Wilson disease: efficacy of penicillamine and zinc therapy. J Clin Gastroenterol 42(2):194–198
Raj JP, Oommen AM, Paul TV (2015) Dietary calcium intake and physical activity levels among urban south Indian postmenopausal women. J Fam Med Prim Care 4(3):461–464
Balk EM, Adam GP, Langberg VN, Earley A, Clark P, Ebeling PR et al (2017) Global dietary calcium intake among adults: a systematic review. Osteoporos Int 28(12):3315–3324
Paul TV, Thomas N, Seshadri MS, Oommen R, Jose A, Mahendri NV (2008) Prevalence of osteoporosis in ambulatory postmenopausal women from a semiurban region in southern India: relationship to calcium nutrition and vitamin D status. Endocr Pract 14(6):665–671
Harinarayan CV, Ramalakshmi T, Prasad UV, Sudhakar D, Srinivasarao PVLN, Sarma KVS et al (2007) High prevalence of low dietary calcium, high phytate consumption, and vitamin D deficiency in healthy south Indians. Am J Clin Nutr 85(4):1062–1067
Peng S, Plank LD, McCall JL, Gillanders LK, McIlroy K, Gane EJ (2007) Body composition, muscle function, and energy expenditure in patients with liver cirrhosis: a comprehensive study. Am J Clin Nutr 85(5):1257–1266
Rizzoli R, Bischoff-Ferrari H, Dawson-Hughes B, Weaver C (2014) Nutrition and bone health in women after the menopause. Womens Health Lond Engl 10(6):599–608
Acknowledgements
We would like to thank the study participants for their participation in the study. This study was funded by the fluid research grant, Christian Medical College, Vellore. The funding body had no role in design, collection and analysis, interpretation of data, writing of the manuscript or decision to submit for publication.
Author information
Authors and Affiliations
Contributions
NK, KGS, MT, CEE and TVP coordinated and conducted the study. NK, KEC and TVP wrote the first draft. CEE and NT revised the draft. All authors reviewed and approved the final version of the draft.
Corresponding author
Ethics declarations
Conflict of interest
Nitin Kapoor, Kripa Elizabeth Cherian, Kattiparambil G. Sajith, Maya Thomas, Chundamannil E. Eapen, Nihal Thomas, Thomas V. Paul confirm that they have no conflict of interest related to this study.
Human and Animal Rights and Informed Consent
All tests on participants were performed in accordance with ethical standards stipulated by the CMC Research ethics committee. This study protocol was approved by the institutional review board prior to initiation of this study vide IRB Min No. 7917, dated 04.07.2016. No amimals were used in this study. All subjects were recruited following a detailed written informed consent, and in those below 18 years of age, an assent form was signed by their parents.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kapoor, N., Cherian, K.E., Sajith, K.G. et al. Renal Tubular Function, Bone Health and Body Composition in Wilson’s Disease: A Cross-Sectional Study from India. Calcif Tissue Int 105, 459–465 (2019). https://doi.org/10.1007/s00223-019-00588-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-019-00588-z