Abstract
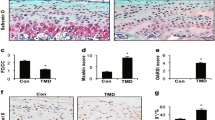
The purpose of this study is to evaluate whether the effects of botulinum neurotoxin (botox) injection into the masseter in the mandibular condylar cartilage (MCC) and subchondral bone could be rescued by compressive loading of the temporomandibular joint (TMJ). Twenty-four 6-week-old female mice (C57BL/6J) were used. Mice were divided in three groups: (1) Botox (n = 8); (2) Botox plus loading (n = 8); (3) Pure control (n = 8). Bone labels (3 and 1 day before sacrifice) and the proliferation marker EdU (2 and 1 day before sacrifice) were intraperitoneally injected into all groups of mice. Condyles were dissected and examined by micro-CT and histology. Sagittal sections of condyles were stained for TRAP, alkaline phosphatase, EdU, TUNEL, and toluidine blue. In addition, immunostaining for pSmad, VEGF, and Runx2 was performed. Bone volume fraction, tissue density, and trabecular thickness were significantly decreased on the subchondral bone of botox-injected side when compared to control side and control mice, 4 weeks after injection. Furthermore, histological analysis revealed decrease in mineralization, matrix deposition, TRAP activity, EdU, and TUNEL-positive cells in the MCC of the botox-injected side, 4 weeks after injection. However, compressive loading reversed the reduced bone volume and density and the cellular changes in the MCC caused by Botox injection. TMJ compressive loading rescues the negative effects of botox injection into the masseter in the MCC and subchondral bone.





Similar content being viewed by others
References
Connelly ST, Myung J, Gupta R, Tartaglia GM, Gizdulich A, Yang J, Silva R (2017) Clinical outcomes of Botox injections for chronic temporomandibular disorders: do we understand how Botox works on muscle, pain, and the brain? Int J Oral Maxillofac Surg 46:322–327
Pedemonte C, Pérez Gutiérrez H, González E, Vargas I, Lazo D (2015) Use of onabotulinumtoxinA in post-traumatic oromandibular dystonia. J Oral Maxillofac Surg 73:152–157
Sidebottom AJ, Patel AA, Amin J (2013) Botulinum injection for the management of myofascial pain in the masticatory muscles. A prospective outcome study. Br J Oral Maxillofac Surg 51:199–205
Ivask O, Leibur E, Akermann S, Tamme T, Voog-Oras U (2016) Intramuscular botulinum toxin injection additional to arthrocentesis in the management of temporomandibular joint pain. Oral Surg Oral Med Oral Pathol Oral Radiol 122:e99–e106
Park MY, Ahn KY, Jung DS (2003) Botulinum toxin type A treatment for contouring of the lower face. Dermatol Surg 29:477–483 (discussion 483)
Kim NH, Chung JH, Park RH, Park JB (2005) The use of botulinum toxin type A in aesthetic mandibular contouring. Plast Reconstr Surg 115:919–930
Xie Y, Zhou J, Li H, Cheng C, Herrler T, Li Q (2014) Classification of masseter hypertrophy for tailored botulinum toxin type A treatment. Plast Reconstr Surg 134:209e–218e
Rossetto O, Pirazzini M, Montecucco C (2014) Botulinum neurotoxins: genetic, structural and mechanistic insights. Nat Rev Micro 12:535–549
Rafferty KL, Liu ZJ, Ye W, Navarrete AL, Nguyen TT, Salamati A, Herring SW (2012) Botulinum toxin in masticatory muscles: short- and long-term effects on muscle, bone, and craniofacial function in adult rabbits. Bone 50:651–662
Raphael KG, Tadinada A, Bradshaw JM, Janal MN, Sirois DA, Chan KC, Lurie AG (2014) Osteopenic consequences of botulinum toxin injections in the masticatory muscles: a pilot study. J Oral Rehabil 41:555–563
Tsai CY, Huang RY, Lee CM, Hsiao WT, Yang LY (2010) Morphologic and bony structural changes in the mandible after a unilateral injection of botulinum neurotoxin in adult rats. J Oral Maxillofac Surg 68:1081–1087
Tsai CY, Yang LY, Chen KT, Chiu WC (2010) The influence of masticatory hypofunction on developing rat craniofacial structure. Int J Oral Maxillofac Surg 39:593–598
Tsai CY, Chiu WC, Liao YH, Tsai CM (2009) Effects on craniofacial growth and development of unilateral botulinum neurotoxin injection into the masseter muscle. Am J Orthod Dentofacial Orthop 135:142.e141–146 (discussion 142–143)
Kim JY, Kim ST, Cho SW, Jung HS, Park KT, Son HK (2008) Growth effects of botulinum toxin type A injected into masseter muscle on a developing rat mandible. Oral Dis 14:626–632
Dutra EH, MH OB, Lima A, Kalajzic Z, Tadinada A, Nanda R, Yadav S (2016) Cellular and matrix response of the mandibular condylar cartilage to botulinum toxin. PLoS ONE 11:e0164599
Kaul R, O’Brien MH, Dutra E, Lima A, Utreja A, Yadav S (2016) The effect of altered loading on mandibular condylar cartilage. PLoS ONE 11:e0160121
Kawamoto T (2003) Use of a new adhesive film for the preparation of multi-purpose fresh-frozen sections from hard tissues, whole-animals, insects and plants. Arch Histol Cytol 66:123–143
Chen H, Ghori-Javed FY, Rashid H, Adhami MD, Serra R, Gutierrez SE, Javed A (2014) Runx2 regulates endochondral ossification through control of chondrocyte proliferation and differentiation. J Bone Miner Res 29:2653–2665
Chen J, Sorensen KP, Gupta T, Kilts T, Young M, Wadhwa S (2009) Altered functional loading causes differential effects in the subchondral bone and condylar cartilage in the temporomandibular joint from young mice. Osteoarthritis Cartilage 17:354–361
Utreja A, Dyment NA, Yadav S, Villa MM, Li Y, Jiang X, Nanda R, Rowe DW (2016) Cell and matrix response of temporomandibular cartilage to mechanical loading. Osteoarthritis Cartilage 24:335–344
Sobue T, Yeh WC, Chhibber A, Utreja A, Diaz-Doran V, Adams D, Kalajzic Z, Chen J, Wadhwa S (2011) Murine TMJ loading causes increased proliferation and chondrocyte maturation. J Dent Res 90:512–516
Shen G, Darendeliler MA (2005) The adaptive remodeling of condylar cartilage—a transition from chondrogenesis to osteogenesis. J Dent Res 84:691–699
Pirttiniemi P, Kantomaa T, Sorsa T (2004) Effect of decreased loading on the metabolic activity of the mandibular condylar cartilage in the rat. Eur J Orthod 26:1–5
Retting KN, Song B, Yoon BS, Lyons KM (2009) BMP canonical Smad signaling through Smad1 and Smad5 is required for endochondral bone formation. Development 136:1093–1104
Hayman AR (2008) Tartrate-resistant acid phosphatase (TRAP) and the osteoclast/immune cell dichotomy. Autoimmunity 41:218–223
Karsenty G, Wagner EF (2002) Reaching a genetic and molecular understanding of skeletal development. Dev Cell 2:389–406
Carlevaro MF, Cermelli S, Cancedda R, Descalzi Cancedda F (2000) Vascular endothelial growth factor (VEGF) in cartilage neovascularization and chondrocyte differentiation: auto-paracrine role during endochondral bone formation. J Cell Sci 113(Pt 1):59–69
Ferrara N, Alitalo K (1999) Clinical applications of angiogenic growth factors and their inhibitors. Nat Med 5:1359–1364
Takeda S, Bonnamy JP, Owen MJ, Ducy P, Karsenty G (2001) Continuous expression of Cbfa1 in nonhypertrophic chondrocytes uncovers its ability to induce hypertrophic chondrocyte differentiation and partially rescues Cbfa1-deficient mice. Genes Dev 15:467–481
Ding M, Lu Y, Abbassi S, Li F, Li X, Song Y, Geoffroy V, Im HJ, Zheng Q (2012) Targeting Runx2 expression in hypertrophic chondrocytes impairs endochondral ossification during early skeletal development. J Cell Physiol 227:3446–3456
Gibson G (1998) Active role of chondrocyte apoptosis in endochondral ossification. Microsc Res Tech 43:191–204
Drissi MH, Li X, Sheu TJ, Zuscik MJ, Schwarz EM, Puzas JE, Rosier RN, O’Keefe RJ (2003) Runx2/Cbfa1 stimulation by retinoic acid is potentiated by BMP2 signaling through interaction with Smad1 on the collagen X promoter in chondrocytes. J Cell Biochem 90:1287–1298
Franceschi RT, Xiao G (2003) Regulation of the osteoblast-specific transcription factor, Runx2: responsiveness to multiple signal transduction pathways. J Cell Biochem 88:446–454
Zelzer E, Glotzer DJ, Hartmann C, Thomas D, Fukai N, Soker S, Olsen BR (2001) Tissue specific regulation of VEGF expression during bone development requires Cbfa1/Runx2. Mech Dev 106:97–106
Ai WJ, Li J, Lin SM, Li W, Liu CZ, Lv WM (2015) R-Smad signaling-mediated VEGF expression coordinately regulates endothelial cell differentiation of rat mesenchymal stem cells. Stem Cells Dev 24:1320–1331
Acknowledgements
The authors have no affiliations with or involvement in any organization or entity with any financial interest in the subject matter or materials discussed in this manuscript. Research reported in this publication was supported by the National Institute of Dental and Craniofacial Research of the National Institute of Health under the Award Number KO8DE025914 and by the American Association of Orthodontic Foundation and startup funds provided to SY.
Funding
National Institute of Craniofacial and Dental Research (KO8DE025914) and American Association of Orthodontic Foundation.
Author information
Authors and Affiliations
Contributions
EHD—conceived and designed study, collected, assembled and interpreted data, drafted and approved article. MHO—conceived and designed study, collected, assembled and interpreted data, drafted and approved article. CL—assisted in processing and interpreting the histological images. AT—collected, assembled and interpreted data, revised and approved article. RN—collected, assembled and interpreted data, revised and approved article. SY—obtained funding, conceived and designed study, collected, assembled and interpreted data, drafted and approved article.
Corresponding author
Ethics declarations
Conflict of interest
Eliane H. Dutra, Mara H. O’Brien, Candice Logan, Aditya Tadinada, Ravindra Nanda, Sumit Yadav declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
All institutional and national guidelines for the care and use of laboratory animals were followed.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dutra, E.H., O’Brien, M.H., Logan, C. et al. Loading of the Condylar Cartilage Can Rescue the Effects of Botox on TMJ. Calcif Tissue Int 103, 71–79 (2018). https://doi.org/10.1007/s00223-017-0385-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-017-0385-x