Abstract
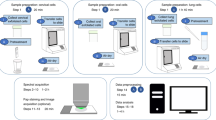
Fine needle aspirates (FNAs) of suspicious breast lesions are often used to aid the diagnosis of female breast cancer. Biospectroscopy tools facilitate the acquisition of a biochemical cell fingerprint representative of chemical bonds present in a biological sample. The mid-infrared (IR; 4,000–400 cm−1) is absorbed by the chemical bonds present, allowing one to derive an absorbance spectrum. Complementary to IR spectroscopy, Raman spectroscopy measures the scattering by chemical bonds following excitation by a laser to generate an intensity spectrum. Our objective was to apply these methods to determine whether a biospectroscopy approach could objectively segregate different categories of FNAs. FNAs of breast tissue were collected (n = 48) in a preservative solution and graded into categories by a cytologist as C1 (non-diagnostic), C2 (benign), C3 (suspicious, probably benign) or C5 (malignant) [or C4 (suspicious, probably malignant); no samples falling within this category were identified during the collection period of the study]. Following washing, the cellular material was transferred onto BaF2 (IR-transparent) slides for interrogation by Raman or Fourier-transform IR (FTIR) microspectroscopy. In some cases where sufficient material was obtained, this was transferred to low-E (IR-reflective) glass slides for attenuated total reflection–FTIR spectroscopy. The spectral datasets produced from these techniques required multivariate analysis for data handling. Principal component analysis followed by linear discriminant analysis was performed independently on each of the spectral datasets for only C2, C3 and C5. The resulting scores plots revealed a marked overlap of C2 with C3 and C5, although the latter pair were both significantly segregated (P < 0.001) in the Raman spectra. Good separation was observed between C3 and C5 in all three spectral datasets. Analysis performed on the average spectra showed the presence of three distinct cytological groups. Our findings suggest that biospectroscopy tools coupled with multivariate analysis may support the current FNA tests whilst increasing the sensitivity and associated reliability for improved diagnostics.

Average IR spectra derived from different categories of FNA specimens






Similar content being viewed by others
Abbreviations
- ν asPO −2 :
-
Asymmetric phosphate stretching vibrations
- ATR-FTIR:
-
Attenuated total reflection Fourier-transform infrared
- BRCA1 :
-
Breast cancer 1
- BRCA2 :
-
Breast cancer 2
- FNA:
-
Fine needle aspirate
- FTIR:
-
Fourier-transform infrared
- IR:
-
Infrared
- LD:
-
Linear discriminant
- LDA:
-
Linear discriminant analysis
- PCA:
-
Principal component analysis
- ν sPO −2 :
-
Symmetric phosphate stretching vibrations
- TP53 :
-
Tumour suppressor protein 53
References
Grover PL, Martin FL (2002) The initiation of breast and prostate cancer. Carcinogenesis 23:1095–1102
Martin FL, Carmichael PL, Crofton-sleigh C, Venitt S, Phillips DH, Grover PL (1996) Genotoxicity of human mammary lipid. Cancer Res 56:5342–5346
Blanks RG, Moss SM, McGahan CE, Quinn MJ, Babb PJ (2000) Effect of NHS breast screening programme on mortality from breast cancer in England and Wales, 1990–8: comparison of observed with predicted mortality. BMJ 321:665–669
Liston J, Wilson R (2010) Clinical guidelines for breast cancer screening assessment, 3rd edn. NHSBSP Publication No. 49
Britton PD, Mccann J (1999) Needle biopsy in the NHS Breast Screening Programme 1996/97: how much and how accurate? Breast 8:5–11
Dixon JM, Thomas J (2006) In: Dixon JM (ed) ABC of breast diseases, 3rd edn. Blackwell, Oxford, pp 1–7
Edmonds P, Jardines L (1999) In: Eskin BA, Asbell SO, Jardines L (eds) Breast disease for primary care physicians. Parthenon Publishing Group, Lancashire, pp 75–82
Kelly JG, Martin-Hirsch PL, Martin FL (2009) Discrimination of base differences in oligonucleotides using mid-infrared spectroscopy and multivariate analysis. Anal Chem 81:5314–5319
Martin FL, Kelly JG, Llabjani L, Martin-Hirsch PL, Patel IP, Trevisan J, Fullwood NJ, Walsh MJ (2010) Distinguishing cell types or populations based on the derivation and computational analyses of their infrared spectra. Nature Prot 5:1748–1760
Stone N, Matousek P (2008) Advanced transmission Raman spectroscopy: a promising tool for breast disease diagnosis. Cancer Res 68:4424–4430
Fabian H, Thi NAN, Eiden M, Lasch P, Schmitt J, Naumann D (2006) Diagnosing benign and malignant lesions in breast tissue sections by using IR-microspectroscopy. Biochim Biophys Acta 1758:874–882
Griffiths PR, Haseth JA (2008) Fourier-transform infrared spectroscopy, 2nd edn. Springer, Berlin
Stuart B (2005) Infrared spectroscopy: fundamentals and applications, 1st edn. Wiley, Chichester
Stone N, Kendall C, Smith J, Crow P, Barr H (2004) Raman spectroscopy for identification of epithelial cancers. Faraday Discuss 126:141–157
Schuster KC, Reese I, Urlaub E, Gapes JR, Lendl B (2000) Multidimensional information on the chemical composition of single bacterial cells by confocal Raman microspectroscopy. Anal Chem 72:5529–5534
Chan JW, Taylor DS, Zwerdling T, Lane SM, Ihara K, Huser T (2006) Micro-Raman spectroscopy detects individual neoplastic and normal hematopoietic cells. Biophys J 90:648–656
Jess PRT, Smith DDW, Mazilu M, Dholakia K, Riches AC, Herrington CS (2007) Early detection of cervical neoplasia by Raman spectroscopy. Int J Cancer 121:2723–2728
Kast RE, Serhatkulu GK, Cao A, Pandya AK, Dai H, Thakur JS, Naik VM, Naik R, Klein MD, Auner GW, Rabah R (2008) Raman spectroscopy can differentiate malignant tumors from normal breast tissue and detect early neoplastic changes in a mouse model. Biopolymers 89:235–241
Lyng FM, Faoláin EO, Conroy J, Meade AD, Knief P, Duffy B, Hunter MB, Bryne JM, Kelehan P, Bryne HJ (2007) Vibrational spectroscopy for cervical cancer pathology, from biochemical analysis to diagnostic tool. Exp Mol Pathol 82:121–129
German MJ, Hammiche A, Ragavan N, Tobin MJ, Cooper LJ, Matanhelia SS, Hindley AC, Nicholson CM, Fullwood NJ, Pollock HM, Martin FL (2006) Infrared spectroscopy with multivariate analysis potentially facilitates the segregation of different types of prostate cell. Biophys J 90:3783–3795
Llabjani V, Trevisan J, Jones KC, Shore RF, Martin FL (2010) Binary mixture effects of PBDE congeners (47, 153, 183, or 209) and PCB congeners (126 or 153) in MCF-7 cells: biochemical alterations assessed by IR spectroscopy and multivariate analysis. Environ Sci Technol 44:3992–3998
Taylor SE, Cheung KT, Patel II, Trevisan J, Stringfellow HF, Ashton KM, Wood NJ, Keating PJ, Martin-Hirsch PL, Martin FL (2011) Infrared spectroscopy with multivariate analysis to interrogate endometrial tissue: a novel and objective diagnostic approach. Br J Cancer 104:790–797
Cheung KT, Trevisan J, Kelly JG, Ashton KM, Stringfellow HF, Taylor SE, Singh MN, Martin-Hirsch PL, Martin FL (2011) Fourier-transform infrared spectroscopy discriminates a spectral signature of endometriosis independent of inter-individual variation. Analyst 136:2047–2055
Martin FL (2011) Shining a new light into molecular workings. Nat Methods 8:385–387
Acknowledgements
This work has been funded by the Rosemere Cancer Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kelly, J.G., Ahmadzai, A.A., Hermansen, P. et al. A biospectroscopic interrogation of fine needle aspirates points towards segregation between graded categories: an initial study towards diagnostic screening. Anal Bioanal Chem 401, 957–967 (2011). https://doi.org/10.1007/s00216-011-5137-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-011-5137-6