Abstract
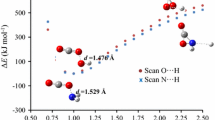
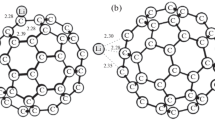
In this work, we analyze the interaction of carbon dioxide with different carboxylate anion derivatives in the gas phase at the ab initio CCSD(T)//MP2 level using the aug-cc-pVTZ basis set. The systems considered here include the formate, acetate, propionate, bicarbonate, carbamate and glycinate anions. The study is relevant to get a better understanding of the interactions involved in novel carbon capture processes through either ionic liquids or amino acid salts. We describe the formation of covalent and non-covalent adducts and show that the formation energies are significantly larger than those previously reported for amines, which are used in conventional carbon capture processes. The nature of the interactions is analyzed using the natural bond orbitals methodology. The binding energy in the non-covalent processes does not depend much on the derivative, but covalent adducts display a rough correlation with nucleophilic/electrophilic indices provided distortion effects on the monomers are taken into account. In the case of glycinate, interactions with the amino and carboxylic moieties involve comparable energetics and the existence of several minima in the potential energy surface might be a factor contributing to the good CO2 capture capacity exhibited by this species in recent experimental studies.









Similar content being viewed by others
References
Paris Agreement, United Nations Treaty. https://treaties.un.org/pages/ViewDetails.aspx?src=TREATY&mtdsg_no=XXVII-7-d&chapter=27&clang=_en. Accessed 25 Feb 2019
Vitillo JG, Smit B, Gagliardi L (2017) Chem Rev 117:9521–9523
Liu Q, Wu L, Jackstell R, Beller M (2015) Nat Commun 6:5933
Rochelle GT (2009) Science 325:1652–1654
Ramdin M, de Loos TW, Vlugt TJ (2012) Ind Eng Chem Res 51:8149–8177
Aghaie M, Rezaei N, Zendehboudi S (2018) Renew Sustain Energy Rev 96:502–525
Jutz F, Andanson J-M, Baiker A (2010) Chem Rev 111:322–353
Jockenhövel T, Schneider R, Rode H (2009) Energy Proc 1:1043–1050
Guo D, Thee H, Tan CY, Chen J, Fei W, Kentish S, Stevens GW, da Silva G (2013) Energy Fuels 27:3898–3904
Zhang Z, Li Y, Zhang W, Wang J, Soltanian MR, Olabi AG (2018) Renew Sustain Energy Rev 98:179–188
Hu G, Smith KH, Wu Y, Mumford KA, Kentish SE, Stevens GW (2018) Chin J Chem Eng
Shiflett MB, Yokozeki A (2008) J Chem Eng Data 54:108–114
Carvalho PJ, Álvarez VH, Schröder B, Gil AM, Marrucho IM, Aznar M, Santos LM, Coutinho JA (2009) J Phys Chem B 113:6803–6812
Barber PS, Griggs CS, Gurau G, Liu Z, Li S, Li Z, Lu X, Zhang S, Rogers RD (2013) Angew Chem Int Ed Engl 52:12350–12353
Ma J, Zhou Z, Zhang F, Fang C, Wu Y, Zhang Z, Li A (2011) Environ Sci Technol 45:10627–10633
Meredith JC, Johnston KP, Seminario JM, Kazarian SG, Eckert CA (1996) J Phys Chem 100:10837–10848
Girard E, Tassaing T, Marty J-D, Destarac M (2016) Chem Rev 116:4125–4169
Raveendran P, Wallen SL (2002) J Am Chem Soc 124:7274–7275
Ingrosso F, Ruiz-Lopez MF (2018) J Phys Chem A 122:1764–1770
Ingrosso F, Ruiz-Lopez MF (2017) ChemPhysChem 18:2560–2572
San-Fabian E, Ingrosso F, Lambert A, Bernal-Uruchurtu MI, Ruiz-Lopez MF (2014) Chem Phys Lett 601:98–102
Munoz-Losa A, Martins-Costa MTC, Ingrosso F, Ruiz-Lopez MF (2014) Mol Simul 40:154–159
Azofra LM, Altarsha M, Ruiz-Lopez MF, Ingrosso F (2013) Theoret Chem Acc 132:1326
Altarsha M, Ingrosso F, Ruiz-Lopez MF (2012) ChemPhysChem 13:3397–3403
Trung NT, Nguyen MT (2013) Chem Phys Lett 581:10–15
Kim N-S, Jeong S-K, Yoon S-H, Park G-S (2011) Bull Korean Chem Soc 32:4441–4443
Shi W, Myers CR, Luebke DR, Steckel JA, Sorescu DC (2011) J Phys Chem B 116:283–295
Shi W, Thompson RL, Albenze E, Steckel JA, Nulwala HB, Luebke DR (2014) J Phys Chem B 118:7383–7394
Steckel JA (2012) J Phys Chem A 116:11643–11650
Tian Q, Li R, Sun H, Xue Z, Mu T (2015) J Mol Liq 208:259–268
Simon NM, Zanatta M, Neumann J, Girard AL, Marin G, Stassen H, Dupont J (2018) ChemPhysChem 19:2879–2884
Hussain MA, Soujanya Y, Sastry GN (2011) Environ Sci Technol 45:8582–8588
Kim N, Yoon S, Park G (2013) Tetrahedron 69:6693–6697
Azofra LM (2015) Chem Phys 453:1–6
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark MJ, Heyd J, Brothers EN, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell AP, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam NJ, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, Gaussian 09. Gaussian Inc, Wallingford
Bartlett RJ (1989) J Phys Chem 93:1697–1708
Moller C, Plesset MS (1934) Phys Rev 46:0618–0622
Dunning THJ (1989) J Chem Phys 90:1007–1023
Lee TJ, Taylor PR (1989) Int J Quantum Chem 36:199–207
Boys SF, Bernardi F (1970) Mol Phys 19:553–566
Reed AE, Curtiss LA, Weinhold F (1988) Chem Rev 88:899–926
Caldwell G, Renneboog R, Kebarle P (1989) Can J Chem 67:611–618
O’Hair RAJ, Bowie JH, Gronert S (1992) Int J Mass Spectrom Ion Process 117:23–36
Remko M, Smieško M, van Duijnen PT (2000) Mol Phys 98:709–714
Chattaraj PK, Maiti B (2001) J Phys Chem A 105:169–183
Domingo LR, Pérez P (2011) Org Biomol Chem 9:7168–7175
Contreras R, Andres J, Safont V, Campodonico P, Santos J (2003) J Phys Chem A 107:5588–5593
Geerlings P, De Proft F, Langenaeker W (2003) Chem Rev 103:1793–1874
Leitzke A, Flyunt R, Theruvathu JA, von Sonntag C (2003) Org Biomol Chem 1:1012–1019
Ingrosso F, Altarsha M, Dumarçay F, Kevern G, Barth D, Marsura A, Ruiz-López MF (2016) Chem Eur J 22:2972–2979
Corradini D, Coudert FX, Vuilleumier R (2016) Nat Chem 8:454–460
da Silva EF, Svendsen HF (2004) Ind Eng Chem Res 43:3413–3418
Davran-Candan T (2014) J Phys Chem A 118:4582–4590
Firaha DS, Kirchner B (2016) Chemsuschem 9:1591–1599
Yang X, Rees RJ, Conway W, Puxty G, Yang Q, Winkler DA (2017) Chem Rev 117:9524–9593
Orestes E, Ronconi CM, de Mesquita Carneiro JW (2014) Phys Chem Chem Phys 16:17213–17219
Siggel MRF, Thomas TD (1992) J Am Chem Soc 114:5795–5800
Kumar P, Hogendoorn J, Versteeg G, Feron P (2003) AIChE J 49:203–213
Acknowledgements
FI gratefully acknowledges the support from the EMERGENCE call of the Chemistry Institute of the CNRS (Project RéScMol). FI and MFRL are grateful to the French CINES (Project lct2550) for providing computational resources. WH is grateful for the IT department at USEK for technical aid to use HPC on Microsoft Azure platform and to AUF for supporting this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Published as part of the special collection of articles derived from the 11th Congress on Electronic Structure: Principles and Applications (ESPA-2018).
Rights and permissions
About this article
Cite this article
Harb, W., Ingrosso, F. & Ruiz-López, M.F. Molecular insights into the carbon dioxide–carboxylate anion interactions and implications for carbon capture. Theor Chem Acc 138, 85 (2019). https://doi.org/10.1007/s00214-019-2472-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-019-2472-8