Abstract
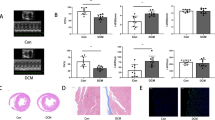
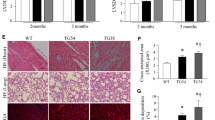
We recently reported that doxorubicin decreased the expression of calpain-1/2, while inhibition of calpain activity promoted doxorubicin-induced cardiac injury in mice. In this study, we investigated whether and how elevation of calpain-2 could affect doxorubicin-triggered cardiac injury. Transgenic mice with inducible cardiomyocyte-specific expression of calpain-2 were generated. An acute cardiotoxicity was induced in both transgenic mice and their relevant wild-type littermates by injection of a single dose of doxorubicin (20 mg/kg) and cardiac injury was analyzed 5 days after doxorubicin injection. Cardiomyocyte-specific up-regulation of calpain-2 did not induce any adverse cardiac phenotypes under physiological conditions by age 3 months, but significantly reduced myocardial injury and improved myocardial function in doxorubicin-treated mice. Cardiac protection of calpain-2 up-regulation was also observed in a mouse model of chronic doxorubicin cardiotoxicity. Up-regulation of calpain-2 increased the protein levels of mitogen activated protein kinase phosphatase-1 (MKP-1) in cultured mouse cardiomyocytes and heart tissues. Over-expression of MKP-1 prevented, whereas knockdown of MKP-1 augmented doxorubicin-induced apoptosis in cultured cardiomyocytes. Moreover, knockdown of MKP-1 offset calpain-2-elicited protective effects against doxorubicin-induced injury in cultured cardiomyocytes. Mechanistically, up-regulation of calpain-2 reduced the protein levels of phosphatase and tensin homolog and consequently promoted Akt activation, leading to increased MKP-1 protein steady-state levels by inhibiting its degradation. Collectively, this study reveals a new role of calpain-2 in promoting MKP-1 expression via phosphatase and tensin homolog/Akt signaling. This study also suggests that calpain-2/MKP-1 signaling may represent new therapeutic targets for doxorubicin-induced cardiac injury.







Similar content being viewed by others
References
Abeyrathna P, Kovacs L, Han W, Su Y (2016) Calpain-2 activates Akt via TGF-beta1-mTORC2 pathway in pulmonary artery smooth muscle cells. Am J Physiol Cell Physiol 311(1):C24–C34. https://doi.org/10.1152/ajpcell.00295.2015
Briz V, Hsu YT, Li Y, Lee E, Bi X, Baudry M (2013) Calpain-2-mediated PTEN degradation contributes to BDNF-induced stimulation of dendritic protein synthesis. J Neurosci 33(10):4317–4328. https://doi.org/10.1523/JNEUROSCI.4907-12.2013
Chang H, Zhang L, Xu PT et al (2011a) Nuclear translocation of calpain-2 regulates propensity toward apoptosis in cardiomyocytes of tail-suspended rats. J Cell Biochem 112(2):571–580. https://doi.org/10.1002/jcb.22947
Chang WT, Li J, Haung HH et al (2011b) Baicalein protects against doxorubicin-induced cardiotoxicity by attenuation of mitochondrial oxidant injury and JNK activation. J Cell Biochem 112(10):2873–2881. https://doi.org/10.1002/jcb.23201
Fan WJ, Genade S, Genis A, Huisamen B, Lochner A (2009) Dexamethasone-induced cardioprotection: a role for the phosphatase MKP-1? Life Sci 84(23–24):838–846. https://doi.org/10.1016/j.lfs.2009.03.014
Galvez AS, Diwan A, Odley AM et al (2007) Cardiomyocyte degeneration with calpain deficiency reveals a critical role in protein homeostasis. Circ Res 100(7):1071-8. https://doi.org/10.1161/01.RES.0000261938.28365.11
Glading A, Bodnar RJ, Reynolds IJ et al (2004) Epidermal growth factor activates m-calpain (calpain II), at least in part, by extracellular signal-regulated kinase-mediated phosphorylation. Mol Cell Biol 24(6):2499–2512
Goll DE, Thompson VF, Li H, Wei W, Cong J (2003) The calpain system. Physiol Rev 83(3):731–801. https://doi.org/10.1152/physrev.00029.2002
Ho WC, Pikor L, Gao Y, Elliott BE, Greer PA (2012) Calpain 2 regulates Akt-FoxO-p27(Kip1) protein signaling pathway in mammary carcinoma. J Biol Chem 287(19):15458–15465. https://doi.org/10.1074/jbc.M112.349308
Kang MY, Zhang Y, Matkovich SJ, Diwan A, Chishti AH, Dorn GW (2010) Receptor-independent cardiac protein kinase Calpha activation by calpain-mediated truncation of regulatory domains. Circ Res 107(7):903–912. https://doi.org/10.1161/CIRCRESAHA.110.220772
Kuwano Y, Gorospe M (2008) Protecting the stress response, guarding the MKP-1 mRNA. Cell Cycle 7(17):2640–2642. https://doi.org/10.4161/cc.7.17.6534
Li J, Zhu H, Shen E, Wan L, Arnold JM, Peng T (2010) Deficiency of rac1 blocks NADPH oxidase activation, inhibits endoplasmic reticulum stress, and reduces myocardial remodeling in a mouse model of type 1 diabetes. Diabetes 59(8):2033–2042. https://doi.org/10.2337/db09-1800
Li Y, Ma J, Zhu H et al (2011) Targeted inhibition of calpain reduces myocardial hypertrophy and fibrosis in mouse models of type 1 diabetes. Diabetes 60(11):2985–2994. https://doi.org/10.2337/db10-1333
Li S, Zhang L, Ni R et al (2016) Disruption of calpain reduces lipotoxicity-induced cardiac injury by preventing endoplasmic reticulum stress. Biochim Biophys Acta 1862(11):2023–2033. https://doi.org/10.1016/j.bbadis.2016.08.005
Liu Y, Shepherd EG, Nelin LD (2007) MAPK phosphatases–regulating the immune response. Nat Rev Immunol 7(3):202–212. https://doi.org/10.1038/nri2035
Liu ZF, Ji JJ, Zheng D, Su L, Peng T (2018) Calpain-2 protects against heat stress-induced cardiomyocyte apoptosis and heart dysfunction by blocking p38 mitogen-activated protein kinase activation. J Cell Physiol. https://doi.org/10.1002/jcp.27750
Lou H, Danelisen I, Singal PK (2005) Involvement of mitogen-activated protein kinases in adriamycin-induced cardiomyopathy. Am J Physiol Heart Circ Physiol 288(4):H1925–H1930. https://doi.org/10.1152/ajpheart.01054.2004
Ma J, Wei M, Wang Q et al (2012) Deficiency of Capn4 gene inhibits nuclear factor-kappaB (NF-kappaB) protein signaling/inflammation and reduces remodeling after myocardial infarction. J Biol Chem 287(33):27480–27489. https://doi.org/10.1074/jbc.M112.358929
Min K, Kwon OS, Smuder AJ et al (2015) Increased mitochondrial emission of reactive oxygen species and calpain activation are required for doxorubicin-induced cardiac and skeletal muscle myopathy. J Physiol 593(8):2017–2036. https://doi.org/10.1113/jphysiol.2014.286518
Ni R, Zheng D, Wang Q et al (2015) Deletion of capn4 protects the heart against endotoxemic injury by preventing ATP synthase disruption and inhibiting mitochondrial superoxide generation. Circ Heart Fail 8(5):988–996. https://doi.org/10.1161/CIRCHEARTFAILURE.115.002383
Peng T, Lu X, Lei M, Feng Q (2003) Endothelial nitric-oxide synthase enhances lipopolysaccharide-stimulated tumor necrosis factor-alpha expression via cAMP-mediated p38 MAPK pathway in cardiomyocytes. J Biol Chem 278(10):8099–8105. https://doi.org/10.1074/jbc.M207288200
Przyklenk K, Maynard M, Darling CE, Whittaker P (2008) Aging mouse hearts are refractory to infarct size reduction with post-conditioning. J Am Coll Cardiol 51(14):1393–1398. https://doi.org/10.1016/j.jacc.2007.11.070
Rastogi R, Jiang Z, Ahmad N et al (2013) Rapamycin induces mitogen-activated protein (MAP) kinase phosphatase-1 (MKP-1) expression through activation of protein kinase B and mitogen-activated protein kinase kinase pathways. J Biol Chem 288(47):33966–33977. https://doi.org/10.1074/jbc.M113.492702
Rojo F, Gonzalez-Navarrete I, Bragado R et al (2009) Mitogen-activated protein kinase phosphatase-1 in human breast cancer independently predicts prognosis and is repressed by doxorubicin. Clin Cancer Res 15(10):3530–3539. https://doi.org/10.1158/1078-0432.CCR-08-2070
Sanbe A, Gulick J, Hanks MC, Liang Q, Osinska H, Robbins J (2003) Reengineering inducible cardiac-specific transgenesis with an attenuated myosin heavy chain promoter. Circ Res 92(6):609–616. https://doi.org/10.1161/01.RES.0000065442.64694.9F
Sandri M, Sandri C, Gilbert A et al (2004) Foxo transcription factors induce the atrophy-related ubiquitin ligase atrogin-1 and cause skeletal muscle atrophy. Cell 117(3):399–412
Simpson L, Parsons R (2001) PTEN: life as a tumor suppressor. Exp Cell Res 264(1):29–41. https://doi.org/10.1006/excr.2000.5130
Small GW, Shi YY, Higgins LS, Orlowski RZ (2007) Mitogen-activated protein kinase phosphatase-1 is a mediator of breast cancer chemoresistance. Cancer Res 67(9):4459–4466. https://doi.org/10.1158/0008-5472.CAN-06-2644
Sugden PH (2003) Ras, Akt, and mechanotransduction in the cardiac myocyte. Circ Res 93(12):1179–1192. https://doi.org/10.1161/01.RES.0000106132.04301.F5
Takemura G, Fujiwara H (2007) Doxorubicin-induced cardiomyopathy from the cardiotoxic mechanisms to management. Prog Cardiovasc Dis 49(5):330–352. https://doi.org/10.1016/j.pcad.2006.10.002
Taneike M, Mizote I, Morita T et al (2011) Calpain protects the heart from hemodynamic stress. J Biol Chem 286(37):32170–32177. https://doi.org/10.1074/jbc.M111.248088
Taniyama Y, Walsh K (2002) Elevated myocardial Akt signaling ameliorates doxorubicin-induced congestive heart failure and promotes heart growth. J Mol Cell Cardiol 34(10):1241–1247
Thandavarayan RA, Watanabe K, Sari FR et al (2010) Modulation of doxorubicin-induced cardiac dysfunction in dominant-negative p38alpha mitogen-activated protein kinase mice. Free Radic Biol Med 49(9):1422–1431. https://doi.org/10.1016/j.freeradbiomed.2010.08.005
Wang Y, Zheng D, Wei M et al (2013) Over-expression of calpastatin aggravates cardiotoxicity induced by doxorubicin. Cardiovasc Res 98(3):381–390. https://doi.org/10.1093/cvr/cvt048
Wang Y, Lopez D, Davey PG et al (2016) Calpain-1 and calpain-2 play opposite roles in retinal ganglion cell degeneration induced by retinal ischemia/reperfusion injury. Neurobiol Dis 93:121–128. https://doi.org/10.1016/j.nbd.2016.05.007
Webster I, Smith A, Lochner A, Huisamen B (2017) The role of MKP-1 in insulin-induced cardioprotection. Cardiovasc Drugs Ther 31(3):247–254. https://doi.org/10.1007/s10557-017-6731-4
Weiss RB (1992) The anthracyclines: will we ever find a better doxorubicin? Semin Oncol 19(6):670–686
Xie P, Guo S, Fan Y, Zhang H, Gu D, Li H (2009) Atrogin-1/MAFbx enhances simulated ischemia/reperfusion-induced apoptosis in cardiomyocytes through degradation of MAPK phosphatase-1 and sustained JNK activation. J Biol Chem 284(9):5488–5496. https://doi.org/10.1074/jbc.M806487200
Yao Y, Xu X, Zhang G, Zhang Y, Qian W, Rui T (2012) Role of HMGB1 in doxorubicin-induced myocardial apoptosis and its regulation pathway. Basic Res Cardiol 107(3):267. https://doi.org/10.1007/s00395-012-0267-3
Yoshida T, Semprun-Prieto L, Sukhanov S, Delafontaine P (2010) IGF-1 prevents ANG II-induced skeletal muscle atrophy via Akt- and Foxo-dependent inhibition of the ubiquitin ligase atrogin-1 expression. Am J Physiol Heart Circ Physiol 298(5):H1565–H1570. https://doi.org/10.1152/ajpheart.00146.2010
Yuasa T, Amo-Shiinoki K, Ishikura S et al (2016) Sequential cleavage of insulin receptor by calpain 2 and gamma-secretase impairs insulin signalling. Diabetologia 59(12):2711–2721. https://doi.org/10.1007/s00125-016-4102-5
Zhang S, Meng T, Liu J, Zhang X, Zhang J (2015a) Cardiac protective effects of dexrazoxane on animal cardiotoxicity model induced by anthracycline combined with trastuzumab is associated with upregulation of calpain-2. Medicine (Baltimore) 94(4):e445. https://doi.org/10.1097/MD.0000000000000445
Zhang YY, Meng C, Zhang XM et al (2015b) Ophiopogonin D attenuates doxorubicin-induced autophagic cell death by relieving mitochondrial damage in vitro and in vivo. J Pharmacol Exp Ther 352(1):166–174. https://doi.org/10.1124/jpet.114.219261
Zheng D, Wang G, Li S, Fan GC, Peng T (2015) Calpain-1 induces endoplasmic reticulum stress in promoting cardiomyocyte apoptosis following hypoxia/reoxygenation. Biochim Biophys Acta 1852(5):882–892. https://doi.org/10.1016/j.bbadis.2015.01.019
Acknowledgements
This work was supported by Natural Science Foundation of Jiangsu Province (BK20171216 to T.P.), the Heart and Stroke Foundation of Canada (G-17-0018361 to T.P.), and Natural Science Foundation of Zhejiang Province (LY14H020005 to J.L.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zheng, D., Su, Z., Zhang, Y. et al. Calpain-2 promotes MKP-1 expression protecting cardiomyocytes in both in vitro and in vivo mouse models of doxorubicin-induced cardiotoxicity. Arch Toxicol 93, 1051–1065 (2019). https://doi.org/10.1007/s00204-019-02405-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-019-02405-w