Abstract
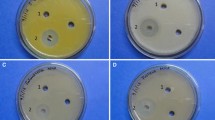
The discovery of new antimicrobials is the prime target in the fight against antimicrobial resistance. The continuous search for new lead compounds from bacteria of untapped and extreme ecosystems such as mangroves is currently being undertaken. This study describes the metabolite profiling of the Streptomyces euryhalinus culture extract. Previously, Streptomyces euryhalinus was isolated from the mangrove forest of Indian Sundarbans as a novel microorganism. The antimicrobial mechanism of action of Streptomyces euryhalinus culture extract against bacteria and fungi has been analyzed in this study. The gas chromatography–mass spectrometry profile of the ethyl acetate extract bacterial culture displayed the presence of several bioactive compounds with antibacterial, antifungal and antioxidant properties. The bacterial extract showed significant antimicrobial activity in terms of zone of inhibition, minimum inhibitory concentration, minimum bactericidal concentration, and minimum fungicidal concentration. Moreover, substantial capacity to alter or damage the inner membrane as well as the outer membrane of the gram-positive and gram-negative bacteria was exhibited by the bacterial extract. This membrane alteration or damaging potential of the extract is the mechanism of action. Biofilm formation inhibition property of the extract also signified its antimicrobial action, and possible use against resistant bacteria. The extract has shown notable activity against the virulence factors like prevention of hemolysis in bacteria and inhibition of secreted aspartyl proteinase in fungi. These functions of the bacterial extract have revealed the extent of its action in the prevention of infection by terminating the secretory virulence factors and by damaging the tissue.






Similar content being viewed by others
References
Andreote FD, Jiménez DJ, Chaves D, Dias Franco AC, Luvizotto DM, Dini-Andreote F, Fasanella CC, Lopez MV, Baena S, Taketani RG, de Melo IS (2012) The microbiome of Brazilian mangrove sediments as revealed by metagenomics. PloSOne 7:e38600
Arumugam M, Mitra A, Jaisankar P, Dasgupta S, Sen T, Gachhui R, Mukhopadhyay UK, Mukherjee J (2010) Isolation of an unusual metabolite 2-allyloxyphenol from a marine actinobacterium, its biological activities and applications. Appl Microbiol Biotechnol 86:109–117
Arumugam M, Mitra A, Pramanik M, Saha M, GachhuiMukjerjeea RJ (2011) Streptomyces sundarbansensis sp. Nov., a novel actinomycete that produces 2-allyloxyphenol. Int J Syst Evol Microbiol 61:2664–2669
Biswas K, Chowdhury JD, Mahansaria R, Mukherjee J (2017) Streptomyces euryhalinus sp. nov., a new actinomycete isolated from a mangrove forest. J Antibiot 70:747–753
Borg M, Rüchel R (1988) Expression of extracellular acid proteinase by proteolytic Candida spp. during experimental infection of oral mucosa. Infect Immun 56:626–631
Cooper MA, Shlaes D (2011) Fix the antibiotics pipeline. Nature 472:32
Debbab A, Aly AH, Proksch P (2011) Bioactive secondary metabolites from endophytes and associated marine derived fungi. Fungal Divers 49:1–12
Denyer SP (1990) Mechanisms of action of biocides. Int Biodeterior 26:89–100
Devi KP, Nisha SA, Sakthivel R, Pandian SK (2010) Eugenol (an essential oil of clove) acts as an antibacterial agent against Salmonella typhi by disrupting the cellular membrane. J Ethnopharmacol 130:107–115
Feller IC, Lovelock CE, Berger U, McKee KL, Joye SB, Ball MC (2010) Biocomplexity in mangrove ecosystems. Annu Rev Mar Sci 2:395–417
Ghosh A, Dey N, Bera A et al (2010) Culture independent molecular analysis of bacterial communities in the mangrove sediment of Sundarban. India Saline Syst 6:1
Gopal B, Chauhan M (2006) Biodiversity and its conservation in the Sundarban mangrove ecosystem. Aquat Sci 68:338–354
Hidayathulla S, Shahat AA, Ahamad SR, Al Moqbil AAN, Alsaid MS, Divakar DD (2018) GC/MS analysis and characterization of 2-Hexadecen-1-oland beta sitosterol from Schimpera arabica extract for its bioactive potential as antioxidant and antimicrobial. J Appl Microbiol 124:1082–1091
Huang Z, Yang J, Cai X et al (2012) A new furanocoumarin from the mangrove endophytic fungus Penicillium sp. (ZH16). Nat Prod Res 26:1291–1295
Ibrahim HR, Sugimoto Y, Aoki T (2000) Ovotransferrin antimicrobial peptide (OTAP-92) kills bacteria througha membrane damage mechanism. Biochim Biophys Acta 1523:196–205
Je JY, Kim Sk (2006) Antimicrobial action of novel chitin derivative. Biochem Biophys Acta 1760:104–109
Kaur G, Balamurugan P, Vasudevan S, Jadav S, Princy SA (2017) Antimicrobial and antibiofilm potential of acyclic amines and diamines against multi-drug resistant Staphylococcus aureus. Front Microbiol 8:1767
Khan I, Bahuguna A, Kumar P, Bajpai VK, Kang SC (2017) Antimicrobial potential of carvacrol against uropathogenic Escherichia coli via membrane disruption, depolarization, and reactive oxygen species generation. Front Microbiol 8:2421
Kovács A, Vasas A, Hohmann J (2008) Natural phenanthrenes and their biological activity. Phytochemistry 69:1084–1110
Kupferwasser LI, Yeaman MR, Nast CC, Kupferwasser D, Xiong YQ, Palma M, Cheung AL, Bayer AS (2003) Salisylic acid attenuates virulence in endovascular infection by targeting global regulatory pathways in Staphylococcus aureus. J Clin Invest 112:222–223
Li N, Sheng-Nan T, Cui J, Guo N, Wang W, Yuan-gang Z, Jin S, Xian-xiu X, Liu Q, Yu-jie F (2014) PA-1, a novel synthesized pyrrolizidine alkaloid, inhibits the growth of Escherichia coli and Staphylococcus aureus by damaging the cell membrane. J Antibiot 67:689–696
MacDonald F, Odds FC (1983) Virulence for mice of a proteinase-secreting strain of Candida albicans and a proteinase-deficient mutant. J Gen Microbiol 129:431–438
Meire P, Ysebaert T, Van Damme S et al (2005) The Scheldt estuary: a description of a changing ecosystem. Hydrobiologia 540:1–11
Michalska M, Wolf P (2015) Pseudomonas Exotoxin A: optimized by evolution for effective killing. Front Microbiol 6:963
O’Neill J (2014) Review on antimicrobial resistance antimicrobial resistance: tackling a crisis for the health and wealth of nations. Review on Antimicrobial Resistance, London
Padmavathia AR, Bakkiyaraja D, Thajuddinb N, Pandian SK (2015) Effect of 2, 4-di-tert-butylphenol on growth and biofilm formation by an opportunistic fungus Candida albicans. Biofouling 31:565–574
Saha M, Ghosh D Jr, Ghosh D, Garai D, Jaisankar P, Sarkar KK, Dutta PK, Das S, Jha T, Mukherjee J (2005) Studies on the production and purification of an antimicrobial compound and taxonomy of the producer isolated from the marine environment of the Sundarbans. Appl Microbiol Biotechnol 66:497–505
Sana S, Datta S, Biswas D, Sengupta D (2018) Assessment of synergistic antibacterial activity of combined biosurfactants revealed by bacterial cell envelop damage. Biochim Biophys Acta Biomembr 1860:579–585
Sarker SD, Nahar L, Kumarasamy Y (2007) Microtitreplate-based antibacterial assay incorporating resazurin as an indicator of cell growth, and its application in the in vitro antibacterial screening of phytochemicals. Methods 42:321–324
Schaller M, Korting HC, Schafer W, Bastert J, Chen W, Hube B (1999) Secreted aspartic proteinase (Sap) activity contributes to tissue damage in a model of human oral Candidosis. Mol Microbiol 34:169–180
Schaller M, Krnjaic N, Niewerth M, Hamm G, Hube B, Korting HC (2003) Effect of antimycotic agents on the activity of aspartyl proteinases secreted by Candida albicans. J Med Microbiol 52:247–249
Simoes M, Bennett RN, Rosa EAS (2009) Understanding antimicrobial activities of phytochemicals against multidrug resistant bacteria and biofilms. Nat Prod Rep 26:746–757
Sundararaman M, Kumar RR, Venkatesan P, Ilangovan A (2013) 1-Alkyl-(N, N-dimethylamino) pyridinium bromides: inhibitory effect on virulence factors of Candida albicans and on the growth of bacterial pathogens. J Med Microbial 62:241–248
Tao Y, Qian L-H, Xie J (2011) Effect of chitosan on membrane permeability and cell morphology of Pseudomonas aeruginosa and Staphyloccocus aureus. Carbohydr Polym 86:969–974
Tomasz A (1994) Multiple-antibiotic resistant pathogenic bacteria. N Engl J Med 330:1247–1251
Varsha KK, Devendra L, Shilpa G, Priya S, Pandey A, Nampoothiri KM (2015) 2,4-Di-tert-butyl phenol as the antifungal, antioxidant bioactive purified from a newly isolated Lactococcus sp. Int J Food Microbiol 211:44–50
Wei YH, Liou GY, Liu HY, Lee FL (2011) Sympodiomycopsis kandeliae sp. nov., a basidiomycetous anamorphic fungus from mangroves, and reclassification of Sympodiomycopsis lanaiensis as Jaminaea lanaiensis comb. nov. Int J Syst Evol Micrbiol 61:469–473
Wiegand I, Hilpert K, Hancock REW (2008) Agar and broth dilution methods to determine the minimal inhibitory concentration (MIC) of antimicrobial substances. Nat Protoc 3:163–175
Wu S, Liu G, Zhang D, Li C, Sun C (2015) Purification and biochemical characterization of analkaline protease from marinebacteria Pseudoalteromonas sp. J Basic Microbiol 55:1427–1434
Wu S, Liu G, Jin W, Xiu P, Sun C (2016) Antibiofilm and anti-infection of a marine bacterial exopolysaccharide against Pseudomonas aeruginosa. Front Microbiol 7:102
Xu MJ, Liu XJ, Zhao YL, Liu D, Xu ZH, Lang XM, Ao P, Lin WH, Yang SL, Zhang ZG, Xu J (2012) Identification and characterization of an anti-fibrotic benzopyran compound isolated from mangrove-derived Streptomyces xiamenensis. Mar Drugs 10:639–646
Xu DB, Ye WW, Han Y, Deng Z-X, Hong K (2014) Natural products from mangrove actinomycetes. Mar Drugs 12:2590–2613
Yadav MK, Chae SW, Im GJ, Chung JW, Song JJ (2015) Eugenol: a phyto-compound effective against methicillin-resistant and methicillin-sensitive Staphylococcus aureus clinical strain biofilms. PLoS ONE 10(3):e0119564
Acknowledgements
We are thankful to Council of Scientific and Industrial Research for funding this research (Grant No. 09/096(0717)/2012-EMR-I).
Author information
Authors and Affiliations
Contributions
KB conceptualized and performed all experiments, and framed the article, DB supported to perform the cell membrane related experiments, interpretation of data and reviewed the article, MS helped to analyses the data and reviewed the article, JM conceptualized the study, supervised the study, and reviewed the article, SK supported in antibiofilm assay and hemolysis assay, and reviewed the article.
Corresponding author
Ethics declarations
Conflict of interest
We declare no conflict of interest.
Accession numbers
Chinese Centre for Industrial Culture Collection 11032T, Leibniz Institute DSMZ–German Collection of Microorganisms and Cell Cultures DSM 103378T.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Biswas, K., Bhattarcharya, D., Saha, M. et al. Evaluation of antimicrobial activity of the extract of Streptomyces euryhalinus isolated from the Indian Sundarbans. Arch Microbiol 204, 34 (2022). https://doi.org/10.1007/s00203-021-02698-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-021-02698-5