Abstract
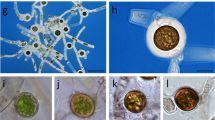
In response to environmental changes, Anabaena cylindrica differentiate three cell types: vegetative cells for photosynthesis, heterocysts for nitrogen fixation, and akinetes for stress survival. Cell-surface polysaccharides play important roles in cyanobacterial ecophysiology. In this study, specific cell-surface sugars were discovered in heterocysts, akinetes and vegetative cells of A. cylindrica using 20 fluorescein-labeled lectins. Both N-acetylglucosamine-binding lectins WGA and succinylated WGA bound specifically to the vegetative cells. Akinetes bound to three mannose-binding lectins (LCA, PSA, and ConA), and one of the galactose-binding lectins (GSL-I). Heterocyst also bound to ConA. However, the heterocysts in all4388 mutant of Anabaena sp. PCC 7120, in which the putative polysaccharide export protein gene all4388 was disrupted, exhibited diminished binding to ConA. Identification of distinct cell-surface sugar helped us to understand the role of polysaccharide for each cell type. Fluorescence-activated cell sorting may be applicable in isolating each cell type for comparative “omics” studies among the three cell types.


Similar content being viewed by others
Abbreviations
- WGA:
-
Wheat germ agglutinin
- PHA-E:
-
Phaseolus vulgaris erythroagglutinin
- ConA:
-
Concanavalin A
- LCA:
-
Len culinaris lectin
- PSA:
-
Pisum sativum agglutinin
- GSL-I:
-
Griffonia Simplicifolia lectin I
References
Bergman B, Gallon J, Rai A, Stal L (1997) N2 Fixation by non-heterocystous cyanobacteria. FEMS Microbiol Rev 19:139–185
Berman-Frank I, Lundgren P, Falkowski P (2003) Nitrogen fixation and photosynthetic oxygen evolution in cyanobacteria. Res Microbiol 154:157–164
Brooks SA (2017) Lectin histochemistry: historical perspectives, state of the art, and the future. Methods Mol Biol 1560:93–107
Cardemil L, Wolk C (1976) The polysaccharides from heterocyst and spore envelopes of a blue-green alga. Methylation analysis and structure of the backbones. J Biol Chem 251:2967–2975
Cardemil L, Wolk CP (1979) The polysaccharides from heterocyst and spore envelopes of a blue-green alga. Structure of the basic repeating unit. J Biol Chem 254:736–741
Cardemil L, Wolk C (1981) Isolated heterocysts of Anabaena variabilis synthesize envelope polysaccharide. Biochim Biophys Acta 674:265–276
Cheng K, Costerton J (1975) Ultrastructure of cell envelopes of bacteria of the bovine rumen. Appl Microbiol 29:841–849
De Philippis R, Sili C, Paperi R, Vincenzini M (2001) Exopolysaccharide-producing cyanobacteria and their possible exploitation: a review. J Appl Phycol 13:293–299
Dudman W (1977) The role of surface polysaccharides in natural environments. In: Sutherland IW (ed) Surface carbohydrates of the prokaryotic cell. Academic, London, pp 357–414
Ernst A, Black T, Cai Y, Panoff JM, Tiwari DN, Wolk CP (1992) Synthesis of nitrogenase in mutants of the cyanobacterium Anabaena sp. strain PCC 7120 affected in heterocyst development or metabolism. J Bacteriol 174:6025–6032
Fay P (1992) Oxygen relations of nitrogen fixation in cyanobacteria. Microbiol Rev 56:340–373
Gerphagnon M, Latour D, Colombet J, Sime-Ngando T (2013) Fungal parasitism: life cycle, dynamics and impact on cyanobacterial blooms. PLoS One 8:e60894
Hirosawa T, Wolk C (1979) Factors controlling the formation of akinetes adjacent to heterocysts in the cyanobacterium Cylindrospermum licheniforme Kütz. J General Microbiol 114:423–432
Hu NT, Thiel T, Giddings TH Jr, Wolk CP (1981) New Anabaena and Nostoc cyanophages from sewage settling ponds. Virology 114:236–246
Huang G, Fan Q, Lechno-Yossef S, Wojciuch E, Wolk CP, Kaneko T, Tabata S (2005) Clustered genes required for the synthesis of heterocyst envelope polysaccharide in Anabaena sp. strain PCC 7120. J Bacteriol 187:1114–1123
Kehr JC, Dittmann E (2015) Biosynthesis and function of extracellular glycans in cyanobacteria. Life (Basel) 5:164–180
Khayatan B, Meeks JC, Risser DD (2015) Evidence that a modified type IV pilus-like system powers gliding motility and polysaccharide secretion in filamentous cyanobacteria. Mol Microbiol 98:1021–1036
Kumar K, Mella-Herrera RA, Golden JW (2010) Cyanobacterial heterocysts. Cold Spring Harb Perspect Biol 2:a000315. https://doi.org/10.1101/cshperspect.a000315
Lechno-Yossef S, Fan Q, Wojciuch E, Wolk CP (2011) Identification of ten Anabaena sp. genes that under aerobic conditions are required for growth on dinitrogen but not for growth on fixed nitrogen. J Bacteriol 193:3482–3489
Maldener I, Hannus S, Kammerer M (2003) Description of five mutants of the cyanobacterium Anabaena sp. strain PCC 7120 affected in heterocyst differentiation and identification of the transposon-tagged genes. FEMS Microbiol Lett 224:205–213
Marczak M, Mazur A, Koper P, Zebracki K, Skorupska A (2017) Synthesis of rhizobial exopolysaccharides and their importance for symbiosis with legume plants. Genes (Basel) 8:E360
Meeks JC, Wycoff KL, Chapman JS, Enderlin CS (1983) Regulation of expression of nitrate and dinitrogen assimilation by Anabaena species. Appl Environ Microbiol 45:1351–1359
Nicolaisen K, Hahn A, Schleiff E (2009) The cell wall in heterocyst formation by Anabaena sp. PCC 7120. J Basic Microbiol 49:5–24
Nobles DR, Romanovicz DK, Brown RM (2001) Cellulose in cyanobacteria. Origin of vascular plant cellulose synthase? Plant Physiol 127:529–542
Paniagua-Michel J, Olmos-Soto J, Morales-Guerrero ER (2014) Algal and microbial exopolysaccharides: new insights as biosurfactants and bioemulsifiers. Adv Food Nutr Res 73:221–257
Pereira S, Zille A, Micheletti E, Moradas-Ferreira P, De Philippis R, Tamagnini P (2009) Complexity of cyanobacterial exopolysaccharides: composition, structures, inducing factors and putative genes involved in their biosynthesis and assembly. FEMS Microbiol Rev 33:917–941
Perez R, Forchhammer K, Salerno G, Maldener I (2016) Clear differences in metabolic and morphological adaptations of akinetes of two Nostocales living in different habitats. Microbiology 162:214–223
Perez R, Wormer L, Sass P, Maldener I (2018) A highly asynchronous developmental program triggered during germination of dormant akinetes of filamentous diazotrophic cyanobacteria. FEMS Microbiol Ecol 94:fix131. https://doi.org/10.1093/femsec/fix131
Schüßler A, Meyer T, Gehrig H, Kluge M (1997) Variations of lectin binding sites in extracellular glycoconjugates during the life cycle of Nostoc punctiforme, a potentially endosymbiotic cyanobacterium. Euro J Phycol 32:233–239
Singh SP, Montgomery BL (2011) Determining cell shape: adaptive regulation of cyanobacterial cellular differentiation and morphology. Trends Microbiol 19:278–285
Tamaru Y, Takani Y, Yoshida T, Sakamoto T (2005) Crucial role of extracellular polysaccharides in desiccation and freezing tolerance in the terrestrial cyanobacterium Nostoc commune. Appl Environ Microbiol 71:7327–7333
Tateno H, Nakamura-Tsuruta S, Hirabayashi J (2009) Comparative analysis of core-fucose-binding lectins from Lens culinaris and Pisum sativum using frontal affinity chromatography. Glycobiology 19:527–536
Tien CJ, Sigee DC, White KN (2005) Characterization of surface sugars on algal cells with fluorescein isothiocyanate-conjugated lectins. Protoplasma 225:225–233
Volesky B, Holan ZR (1995) Biosorption of heavy-metals. Biotechnol Prog 11:235–250
Wilde A, Mullineaux CW (2015) Motility in cyanobacteria: polysaccharide tracks and Type IV pilus motors. Mol Microbiol 98:998–1001
Wolk CP (1966) Evidence of a role of heterocysts in the sporulation of a blue-green alga. Amer J Bot 53:260–262
Wolk CP (1968) Movement of carbon from vegetative cells to heterocysts in Anabaena cylindrica. J Bacteriol 96:2138–2143
Zhou R, Wolk CP (2002) Identification of an akinete marker gene in Anabaena variabilis. J Bacteriol 184:2529–2532
Zhou R, Wolk CP (2003) A two-component system mediates developmental regulation of biosynthesis of a heterocyst polysaccharide. J Biol Chem 278:19939–19946
Acknowledgements
The authors would like to thank Dr. Peter Wolk for providing the all4388-knockout mutant FQ1595, Dr. Brözel for his advice on manuscript preparation, Dilkaran Singh’s graphing help, and acknowledge use of the South Dakota State University Functional Genomics Core Facility supported in part by NSF/EPSCoR Grant No. 0091948 and by the State of South Dakota. This work was partially supported by USDA-NIFA GRANT11665597 (to R. Z.), and by the South Dakota Agricultural Experiment Station.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Erko Stackebrandt.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1 (AVI 149313 KB)
Rights and permissions
About this article
Cite this article
Qiu, Y., Tian, S., Gu, L. et al. Identification of surface polysaccharides in akinetes, heterocysts and vegetative cells of Anabaena cylindrica using fluorescein-labeled lectins. Arch Microbiol 201, 17–25 (2019). https://doi.org/10.1007/s00203-018-1565-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-018-1565-4