Abstract
Summary
Bone marrow adipose tissue has not been studied in patients with inactive inflammatory bowel disease. We found that these patients have preserved marrow adiposity even with low bone mass. Factors involved in bone loss in active disease may have long-lasting effects but do not seem to affect bone marrow adiposity.
Introduction
Reduced bone mass is known to occur at varying prevalence in patients with inflammatory bowel diseases (IBD) because of inflammation, malnutrition, and steroid therapy. Osteoporosis may develop in these patients as the result of an imbalanced relationship between osteoblasts and adipocytes in bone marrow. This study aimed to evaluate for the first time bone mass and bone marrow adipose tissue (BMAT) in a particular subgroup of IBD patients characterized by long-term, steroid-free remission.
Methods
Patients with Crohn’s disease (CD; N = 21) and ulcerative colitis (UC; N = 15) and controls (C; N = 65) underwent dual X-ray energy absorptiometry and nuclear magnetic resonance spectroscopy of the L3 lumbar vertebra for BMAT assessment.
Results
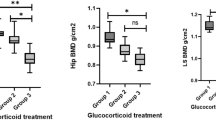
Both the CD and UC subgroups showed significantly higher proportions of patients than controls with Z-score ≤−2.0 at L1–L4 (C 1.54%; CD 19.05%; UC 20%; p = 0.02), but not at other sites. The proportions of CD patients with a T-score ˂−1.0 at the femoral neck (C 18.46%; CD 47.62%; p = 0.02) and total hip (C 16.92%; CD 42.86%; p = 0.03) were significantly higher than among controls. There were no statistically significant differences between IBD patients and controls regarding BMAT at L3 (C 28.62 ± 8.15%; CD 29.81 ± 6.90%; UC 27.35 ± 9.80%; p = 0.67).
Conclusions
IBD patients in long-term, steroid-free remission may have a low bone mass in spite of preserved BMAT. These findings confirm the heterogeneity of bone disorders in IBD and may indicate that factors involved in bone loss in active disease may have long-lasting effects on these patients.



Similar content being viewed by others
References
Ali T, Lam D, Bronze MS et al (2009) Osteoporosis in inflammatory bowel disease. Am J Med 122:599–604
Ghosh S, Cowen S, Hannan WJ et al (1994) Low bone mineral density in Crohn’s disease, but not in ulcerative colitis, at diagnosis. Gastroenterology 107:1031–1039
Tilg H, Moschen AR, Kaser A et al (2008) Gut, inflammation and osteoporosis: basic and clinical concepts. Gut 57:684–694
Jahnsen J, Falch JA, Aadland E et al (1997) Bone mineral density is reduced in patients with Crohn’s disease but not in patients with ulcerative colitis: a population based study. Gut 40:313–319
Ardizzone S, Bollani S, Bettica P et al (2000) Altered bone metabolism in inflammatory bowel disease: there is a difference between Crohn’s disease and ulcerative colitis. J Intern Med 247:63–70
Silvennoinen A, Karttunen TJ, Niemelã SE et al (1995) A controlled study of bone mineral density in patients with inflammatory bowel disease. Gut 37:71–76
Kanis JA (1994) Assessment of fracture risk and its application to screening for postmenopausal osteoporosis: synopsis of a WHO report. WHO Study Group. Osteoporos Int 4:368–381
Pigot F, Roux C, Chaussade S et al (1992) Low bone mineral density in patients with inflammatory bowel disease. Dig Dis Sci 37:1396–1403
Roux C, Abitbol V, Chaussade S et al (1995) Bone loss in patients with inflammatory bowel disease: a prospective study. Osteoporos Int 5:156–160
Targownik LE, Bernstein CN, Leslie WD (2014) Risk factors and management of osteoporosis in inflammatory bowel disease. Curr Opin Gastroenterol 30:168–174
Pietschmann P, Resch H, Woloszczuk W et al (1990) A circadian rhythm of serum osteocalcin levels in postmenopausal osteoporosis. Eur J Clin Investig 20:310–312
Yavropoulou MP, Tomos K, Tsekmekidou X et al (2011) Response of biochemical markers of bone turnover to oral glucose load in diseases that affect bone metabolism. Eur J Endocrinol 164:1035–1041
Sanchez Cano D, Ruiz-Villaverde R, Olvera Porcel MC et al (2011) Evaluation of bone mineral density, bone turnover markers, the OPG/RANKL system and sTNF-RI in Crohn’s disease. Gastroenterol Hepatol 34:3–9
de Paula FJ, Rosen CJ (2013) Bone remodeling and energy metabolism: new perspectives. Bone Res 1:72–84
Bianco P, Riminucci M, Gronthos S et al (2001) Bone marrow stromal stem cells: nature, biology, and potential applications. Stem Cells 19:180–192
Li X, Kuo D, Schafer AL et al (2011) Quantification of vertebral bone marrow fat content using 3 tesla MR spectroscopy: reproducibility, vertebral variation, and applications in osteoporosis. J Magn Reson Imaging 33:974–979
Bredella MA, Fazeli PK, Miller KK et al (2009) Increased bone marrow fat in anorexia nervosa. J Clin Endocrinol Metab 94:2129–2136
Liu Y, Tang GY, Tang RB et al (2010) Assessment of bone marrow changes in postmenopausal women with varying bone densities: magnetic resonance spectroscopy and diffusion magnetic resonance imaging. Chin Med J 123:1524–1527
Tang GY, Lv ZW, Tang RB et al (2010) Evaluation of MR spectroscopy and diffusion-weighted MRI in detecting bone marrow changes in postmenopausal women with osteoporosis. Clin Radiol 65:377–381
Silverberg MS, Satsangi J, Ahmad T et al (2005) Toward an integrated clinical, molecular and serological classification of inflammatory bowel disease: report of a Working Party of the 2005 Montreal World Congress of Gastroenterology. Can J Gastroenterol 19(Suppl A):5a–36a
Best WR, Becktel JM, Singleton JW et al (1976) Development of a Crohn’s disease activity index. National Cooperative Crohn’s Disease Study. Gastroenterology 70:439–444
Schousboe JT, Shepherd JA, Bilezikian JP et al (2013) Executive summary of the 2013 ISCD position development conference on bone densitometry. J Clin Densitom 16:455–467
de Paula FJA, de Araújo IM, Carvalho AL et al (2015) The relationship of fat distribution and insulin resistance with lumbar spine bone mass in women. PLoS One 10:e0129764
Zhang Q, Riddle RC, Clemens TL (2015) Bone and the regulation of global energy balance. J Intern Med 277:681–689
Rosen CJ, Bouxsein ML (2006) Mechanisms of disease: is osteoporosis the obesity of bone? Nat Clin Pract Rheumatol 2:35–43
Cosman F, de Beur SJ, LeBoff MS et al (2014) Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int 25:2359–2381
van Hogezand RA, Hamdy NA (2006) Skeletal morbidity in inflammatory bowel disease. Scand J Gastroenterol Suppl 243:59–64
Bernstein CN, Leslie WD, Leboff MS (2003) AGA technical review on osteoporosis in gastrointestinal diseases. Gastroenterology 124:795–841
Compston J, Reid DM, Boisdron J et al (2008) Recommendations for the registration of agents for prevention and treatment of glucocorticoid-induced osteoporosis: an update from the Group for the Respect of Ethics and Excellence in Science. Osteoporos Int 19:1247–1250
Seguro LP, Rosario C, Shoenfeld Y (2013) Long-term complications of past glucocorticoid use. Autoimmun Rev 12:629–632
Cohen A, Shen W, Dempster DW et al (2015) Marrow adiposity assessed on transiliac crest biopsy samples correlates with noninvasive measurement of marrow adiposity by proton magnetic resonance spectroscopy (1H-MRS) at the spine but not the femur. Osteoporos Int 26:2471–2478
Bredella MA, Daley SM, Kalra MK et al (2015) Marrow adipose tissue quantification of the lumbar spine by using dual-energy CT and single-voxel 1H MR spectroscopy: a feasibility study. Radiology 277:230–235
Maciel JG, de Araújo IM, Carvalho AL et al (2016) Marrow fat quality differences by sex in healthy adults. J Clin Densitom. doi:10.1016/j.jocd.2016.08.002
Blebea JS, Houseni M, Torigian DA et al (2007) Structural and functional imaging of normal bone marrow and evaluation of its age-related changes. Semin Nucl Med 37:185–194
Liney GP, Bernard CP, Manton DJ et al (2007) Age, gender, and skeletal variation in bone marrow composition: a preliminary study at 3.0 tesla. J Magn Reson Imaging 26:787–793
Schwartz AV (2015) Marrow fat and bone: review of clinical findings. Front Endocrinol 6:1–6
Devlin MJ, Cloutier AM, Thomas NA et al (2010) Caloric restriction leads to high marrow adiposity and low bone mass in growing mice. J Bone Miner Res 25:2078–2088
Fazeli PK, Bredella MA, Freedman L et al (2012) Marrow fat and preadipocyte factor-1 levels decrease with recovery in women with anorexia nervosa. J Bone Miner Res 27:1864–1871
de Araújo IM, Salmon CE, Nahas AK et al (2017) Marrow adipose tissue spectrum in obesity and type 2 diabetes mellitus. Eur J Endocrinol 176:21–30
Schett G, Kiechl S, Redlich K et al (2004) Soluble RANKL and risk of nontraumatic fracture. JAMA 291:1108–1113
Vega D, Maalouf NM, Sakhaee K (2007) CLINICAL review #: the role of receptor activator of nuclear factor-kappaB (RANK)/RANK ligand/osteoprotegerin: clinical implications. J Clin Endocrinol Metab 92:4514–4521
Cawthorn WP, Scheller EL, Leaman BS et al (2014) Bone marrow adipose tissue is an endocrine organ that contributes to increased circulating adiponectin during caloric restriction. Cell Metab 20:368–375
Pironi L, Callegari C, Cornia GL et al (1988) Lactose malabsorption in adult patients with Crohn’s disease. Am J Gastroenterol 83:1267–1271
Dinca M, Fries W, Luisetto G et al (1999) Evolution of osteopenia in inflammatory bowel disease. Am J Gastroenterol 94:1292–1297
Arj A, Razavi Zade M, Yavari M et al (2016) Proton pump inhibitors use and change in bone mineral density. Int J Rheum Dis 19:864–868
Acknowledgements
The authors are indebted to Marta T. Nakao and Rodrigo A. Pessini for their competent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional Ethics Committee (HCRP Case No. 3836/2011) and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Funding
This study was funded by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP).
Conflicts of interest
None.
Rights and permissions
About this article
Cite this article
Bastos, C.M., Araújo, I.M., Nogueira-Barbosa, M.H. et al. Reduced bone mass and preserved marrow adipose tissue in patients with inflammatory bowel diseases in long-term remission. Osteoporos Int 28, 2167–2176 (2017). https://doi.org/10.1007/s00198-017-4014-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-017-4014-3