Abstract
Summary
We tested the feasibility of a fracture prevention decision aid in an online patient portal. The decision aid was acceptable for patients and successfully decreased decisional conflict. This study suggests the possible utility of leveraging the patient portal to enhance patient education and decision making in osteoporosis care.
Introduction
Although interventions have improved osteoporosis screening and/or treatment for certain populations of high-risk patients, recent national studies suggest that large-scale uptake of these interventions has been limited. We aimed to determine the feasibility and potential efficacy of a patient portal-based osteoporosis decision aid (DA).
Methods
We conducted a pilot randomized controlled trial of primary care patients aged ≥55 who were enrolled in a patient portal and had a T-score of <−1. Intervention subjects were provided a link to a patient DA. The DA contained a 10-year fracture risk calculator, summary of medication risks and benefits (prescription and nonprescription), and an elicitation of values. Subjects completed questionnaires assessing the primary outcomes of decisional conflict and preparation for decision making and secondary outcomes related to feasibility and planning for a larger trial. Charts were reviewed for physician-subject interactions and medication uptake.
Results
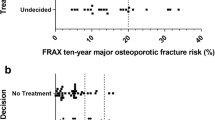
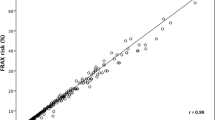
The DA was acceptable to subjects, but 17 % of the patients in the decision aid arm incorrectly entered their T-scores into FRAX-based risk calculator. Decisional conflict was lower post-intervention for those who were randomized to the decision aid arm compared to controls (17.8 vs. 47.1, p < .001), and there was a significant difference in the percentage of patients who made a treatment decision at 3 months. No significant differences were observed in medication uptake.
Conclusions
A portal-based osteoporosis DA was acceptable and improved several measures of decision quality. Given its effect on improving the quality of patients’ decisions, future studies should examine whether it improves physician guideline adherence or medication adherence uptake among treated patients.



Similar content being viewed by others
References
National Osteoporosis Foundation (2014) Clinician’s guide to prevention and treatment of osteoporosis. National Osteoporosis Foundation, Washington D.C
Watts NB, Bilezikian JP, Camacho PM, Greenspan SL, Harris ST, Hodgson SF, Kleerekoper M, Luckey MM, McClung MR, Pollack RP, Petak SM, Osteoporosis Task Force AACE (2010) American Association of Clinical Endocrinologists medical guidelines for clinical practice for the diagnosis and treatment of postmenopausal osteoporosis. Endocr Pract 16(Suppl 3):1–37. doi:10.4158/EP.16.S3.1
Neuner JM, Binkley N, Sparapani RA, Laud PW, Nattinger AB (2006) Bone density testing in older women and its association with patient age. J Am Geriatr Soc 54(3):485–489. doi:10.1111/j.1532-5415.2005.00622.x
Andrade SE, Majumdar SR, Chan KA, Buist DS, Go AS, Goodman M, Smith DH, Platt R, Gurwitz JH (2003) Low frequency of treatment of osteoporosis among postmenopausal women following a fracture. Arch Intern Med 163(17):2052–2057. doi:10.1001/archinte.163.17.2052
Lo J, Pressman A, Omar M, Ettinger B (2006) Persistence with weekly alendronate therapy among postmenopausal women. Osteoporosis Int 17(6):922–928. doi:10.1007/s00198-006-0085-2
Kothawala P, Badamgarav E, Ryu S, Miller RM, Halbert RJ (2007) Systematic review and meta-analysis of real-world adherence to drug therapy for osteoporosis. Mayo Clin Proc 82(12):1493–1501. doi:10.1016/s0025-6196(11)61093-8
Solomon DH, Johnston SS, Boytsov NN, McMorrow D, Lane JM, Krohn KD (2014) Osteoporosis medication use after hip fracture in U.S. patients between 2002 and 2011. J Bone Miner Res 29(9):1929–1937. doi:10.1002/jbmr.2202
Balasubramanian A, Tosi LL, Lane JM, Dirschl DR, Ho P-R, O’Malley CD (2014) Declining rates of osteoporosis management following fragility fractures in the U.S., 2000 through 2009. J Bone Joint Surg 96(7):e52. doi:10.2106/jbjs.l.01781
Saag KG, Gehlbach SH, Curtis JR, Youket TE, Worley K, Lange JL (2006) Trends in prevention of glucocorticoid-induced osteoporosis. J Rheum 33(8):1651–1657
Kamel H, Hussain M, Tariq S, Perry HI, Morley J (2000) Failure to diagnose and treat osteoporosis in elderly patients hospitalized with hip fracture. Am J Med 109:326–328. doi:10.1016/S0002-9343(00)00457-5
Neuner JM, Zhang X, Sparapani R, Laud PW, Nattinger AB (2007) Racial and socioeconomic disparities in bone density testing before and after hip fracture. J Gen Intern Med 22:123–145. doi:10.1007/s11606-007-0217-1
Neuner JM, Zimmer JK, Hamel MB (2003) Diagnosis and treatment of osteoporosis in patients with vertebral compression fractures. J Am Geriatr Soc 51(4):483–491. doi:10.1046/j.1532-5415.2003.51156.x
Stacey D, Légaré F, Col NF, Bennett CL, Barry MJ, Eden KB, Holmes-Rovner M, Llewellyn-Thomas H, Lyddiatt A, Thomson R (2014) Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev (1). doi:10.1002/14651858.CD001431.pub4
Agency for Healthcare Research and Quality (2012) Healthy bones: a decision aid for women after menopause. http://www.effectivehealthcare.ahrq.gov/ehc/decisionaids/osteoporosis/. Accessed: 2013
Ottawa Hospital Research Institute (2011) Osteoporosis: Should I have a dual x-ray absorptiometry (DEXA) test? http://204.187.39.28/AZsumm.php?ID=1128. Accessed February 2013
Neuner J, Fedders M, Caravella M, Bradford L, Schapira M (2015) Meaningful use and the patient portal: patient enrollment, use, and satisfaction with patient portals at a later-adopting center. Am J Med Qual 30(2):105–113. doi:10.1177/1062860614523488
Charts to download (2011) World Health Organization Collaborating Centre for Metabolic Bone Diseases. http://www.shef.ac.uk/FRAX/charts.aspx. Accessed 2013
Elwyn G, O’Connor A, Stacey D, Volk R, Edwards A, Coulter A, Thomson R, Barratt A, Barry M, Bernstein S, Butow P, Clarke A, Entwistle V, Feldman-Stewart D, Holmes-Rovner M, Llewellyn-Thomas H, Moumjid N, Mulley A, Ruland C, Sepucha K, Sykes A, Whelan T (2006) Developing a quality criteria framework for patient decision aids: online international Delphi consensus process. Brit Med J 333(7565):417. doi:10.1136/bmj.38926.629329.AE
Graham I, O’Connor A (1996) Preparation for decision making scale. http://decisionaid.ohri.ca/docs/develop/Tools/PrepDM.pdf. Accessed February 2013
O’Connor A (1993) User manual- decisional conflict scale. http://decisionaid.ohri.ca/docs/develop/User_Manuals/UM_Decisional_Conflict.pdf. Accessed February 2013
Fowler FJ Jr, Gallagher PM, Drake KM, Sepucha KR (2013) Decision dissonance: evaluating an approach to measuring the quality of surgical decision making. Joint Comm J Quality Patient Saf 39(3):136–144
Zikmund-Fisher BJ, Couper MP, Singer E, Levin CA, Fowler FJ, Ziniel S, Ubel PA, Fagerlin A (2010) The DECISIONS study: a nationwide survey of United States adults regarding 9 common medical decisions. Med Decis Mak 30(5 suppl):20S–34S. doi:10.1177/0272989x09353792
O’Connor AM (1995) Validation of a decisional conflict scale. Med Decis Mak 15(1):25–30. doi:10.1177/0272989X9501500105
Neuner JM, Schapira MM (2014) Patient perceptions of osteoporosis treatment thresholds. J Rheumatol 41(3):516–522. doi:10.3899/jrheum.130548
Montori VM, Shah ND, Pencille LJ, Branda ME, Van Houten HK, Swiglo BA, Kesman RL, Tulledge-Scheitel SM, Jaeger TM, Johnson RE, Bartel GA, Melton Iii LJ, Wermers RA (2011) Use of a decision aid to improve treatment decisions in osteoporosis: the osteoporosis choice randomized trial. Am J Med 124(6):549–556. doi:10.1016/j.amjmed.2011.01.013
Acknowledgments
A special thanks to Glenn Bushee for his invaluable assistance in the development of the decision aid and to the Agency for Healthcare Research and Quality and staff at John M. Eisenberg Center for Clinical Decisions and Communications Science for making their decision aid publically available.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Funding
This study was funded by the Clinical and Translational Science Institute of Southeast Wisconsin (project number 5,520,204).
Conflicts of interest
None.
Rights and permissions
About this article
Cite this article
Smallwood, A.J., Schapira, M.M., Fedders, M. et al. A pilot randomized controlled trial of a decision aid with tailored fracture risk tool delivered via a patient portal. Osteoporos Int 28, 567–576 (2017). https://doi.org/10.1007/s00198-016-3767-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-016-3767-4