Abstract
Introduction
Bone mineral density (BMD) is the major factor for determining bone strength, which is closely correlated to osteoporotic fracture risk and is largely determined by multiple genetic factors. The RANK (TNFRSF11A), receptor for RANKL, is a member of the tumor necrosis factor receptor (TNFR) superfamily and plays a central role in osteoclast development.
Methods
In order to investigate the effects of RANK polymorphism on BMD and osteoporosis, we directly sequenced the RANK gene in 24 Korean individuals and identified 25 sequence variants. Eleven of these polymorphisms were selected and genotyped in a larger-scale study of postmenopausal women (n = 560). Areal BMD (g/cm2) of the anterior–posterior lumbar spine and the nondominant proximal femur were measured using dual-energy X-ray absorptiometry.
Results
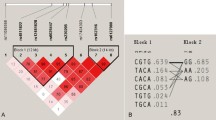
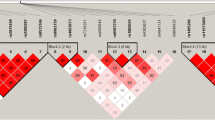
We found that two intronic polymorphisms in the RANK gene [RANK + 34863G > A (rs12458117) and RANK + 35928insdelC (new polymorphism found in this study) in intron 6] were significantly associated with the BMD of the lumbar spine, i.e., rare alleles were significantly associated with low BMD of the lumbar spine among Korean postmenopausal women (p = 0.04 and 0.02, respectively). These polymorphisms were also associated with low BMD of proximal femur sites, including Ward’s triangle, trochanter, and total femur. Our results suggest that +34863G > A and +35928insdelC polymorphisms in RANK are possible genetic factors for low BMD in postmenopausal women.

Similar content being viewed by others
References
NIH Consensus Development Panel on Osteoporosis Prevention D, and Therapy (2001) Osteoporosis prevention, diagnosis, and therapy. JAMA 285:785–795
Arden NK, Spector TD (1997) Genetic influences on muscle strength, lean body mass, and bone mineral density: a twin study. J Bone Miner Res 12:2076–2081
Eisman JA (1999) Genetics of osteoporosis. Endocr Rev 20:788–804
Kanis JA, Melton LJ 3rd, Christiansen C, Johnston CC, Khaltaev N (1994) The diagnosis of osteoporosis. J Bone Miner Res 9:1137–1141
Baron R (2004) Arming the osteoclast. Nat Med 10:458–460
Biswas RN, Baker DN, Hruska KN, Chellaiah MN (2004) Polyphosphoinositides-dependent regulation of the osteoclast actin cytoskeleton and bone resorption. BMC Cell Biol 5:19
Karst M, Gorny G, Galvin RJ, Oursler MJ (2004) Roles of stromal cell RANKL, OPG, and M-CSF expression in biphasic TGF-beta regulation of osteoclast differentiation. J Cell Physiol 200:99–106
Teitelbaum SL (2000) Bone resorption by osteoclasts. Science 289:1504–1508
Fata JE, Kong YY, Li J, Sasaki T, Irie-Sasaki J, Moorehead RA, Elliott R, Scully S, Voura EB, Lacey DL, Boyle WJ, Khokha R, Penninger JM (2000) The osteoclast differentiation factor osteoprotegerin-ligand is essential for mammary gland development. Cell 103:41–50
Hsu H, Lacey DL, Dunstan CR, Solovyev I, Colombero A, Timms E, Tan HL, Elliott G, Kelley MJ, Sarosi I, Wang L, Xia XZ, Elliott R, Chiu L, Black T, Scully S, Capparelli C, Morony S, Shimamoto G, Bass MB, Boyle WJ (1999) Tumor necrosis factor receptor family member RANK mediates osteoclast differentiation and activation induced by osteoprotegerin ligand. Proc Natl Acad Sci USA 96:3540–3545
Wong BR, Rho J, Arron J, Robinson E, Orlinick J, Chao M, Kalachikov S, Cayani E, Bartlett FS 3rd, Frankel WN, Lee SY, Choi Y (1997) TRANCE is a novel ligand of the tumor necrosis factor receptor family that activates c-Jun N-terminal kinase in T cells. J Biol Chem 272:25190–25194
Galibert L, Tometsko ME, Anderson DM, Cosman D, Dougall WC (1998) The involvement of multiple tumor necrosis factor receptor (TNFR)- associated factors in the signaling mechanisms of receptor activator of NF-kappaB, a member of the TNFR superfamily. J Biol Chem 273:34120–34127
Darnay BG, Haridas V, Ni J, Moore PA, Aggarwal BB (1998) Characterization of the intracellular domain of receptor activator of NF-kappaB (RANK). Interaction with tumor necrosis factor receptor- associated factors and activation of NF-kappab and c-Jun N-terminal kinase. J Biol Chem 273:20551–20555
Yamamoto A, Miyazaki T, Kadono Y, Takayanagi H, Miura T, Nishina H, Katada T, Wakabayashi K, Oda H, Nakamura K, Tanaka S (2002) Possible involvement of IkappaB kinase 2 and MKK7 in osteoclastogenesis induced by receptor activator of nuclear factor kappaB ligand. J Bone Miner Res 17:612–621
Darnay BG, Ni J, Moore PA, Aggarwal BB (1999) Activation of NF-kappaB by RANK requires tumor necrosis factor receptor- associated factor (TRAF) 6 and NF-kappaB-inducing kinase. Identification of a novel TRAF6 interaction motif. J Biol Chem 274:7724–7731
Zhang DE, Hetherington CJ, Chen HM, Tenen DG (1994) The macrophage transcription factor PU.1 directs tissue-specific expression of the macrophage colony-stimulating factor receptor. Mol Cell Biol 14:373–381
Johnson-Pais TL, Singer FR, Bone HG, McMurray CT, Hansen MF, Leach RJ (2003) Identification of a novel tandem duplication in exon 1 of the TNFRSF11A gene in two unrelated patients with familial expansile osteolysis. J Bone Miner Res 18:376–380
Hughes AE, Ralston SH, Marken J, Bell C, MacPherson H, Wallace RG, van Hul W, Whyte MP, Nakatsuka K, Hovy L, Anderson DM (2000) Mutations in TNFRSF11A, affecting the signal peptide of RANK, cause familial expansile osteolysis. Nat Genet 24:45–48
Cody JD, Singer FR, Roodman GD, Otterund B, Lewis TB, Leppert M, Leach RJ (1997) Genetic linkage of Paget disease of the bone to chromosome 18q. Am J Hum Genet 61:1117–1122
Good DA, Busfield F, Fletcher BH, Duffy DL, Kesting JB, Andersen J, Shaw JT (2002) Linkage of Paget disease of bone to a novel region on human chromosome 18q23. Am J Hum Genet 70:517–525
Choi JY, Shin A, Park SK, Chung HW, Cho SI, Shin CS, Kim H, Lee KM, Lee KH, Kang C, Cho DY, Kang D (2005) Genetic polymorphisms of OPG, RANK, and ESR1 and bone mineral density in Korean postmenopausal women. Calcif Tissue Int 77:152–159
Hsu YH, Niu T, Terwedow HA, Xu X, Feng Y, Li Z, Brain JD, Rosen CJ, Laird N, Xu X (2006) Variation in genes involved in the RANKL/RANK/OPG bone remodeling pathway are associated with bone mineral density at different skeletal sites in men. Hum Genet 118:568–577
Nathan H (1962) Osteophytes of the vertebral column: an anatomical study of their development according to age, race, and sex with considerations as to their etiology and significance. J Bone Joint Surg Am 44A:243–268
Kiel D (1995) Assessing vertebral fractures. National Osteoporosis Foundation Working Group on Vertebral Fractures. J Bone Miner Res 10:518–523
Livak KJ (1999) Allelic discrimination using fluorogenic probes and the 5’ nuclease assay. Genet Anal 14:143–149
Hedrick PW (1987) Gametic disequilibrium measures: proceed with caution. Genetics 117:331–341
Hedrick P, Kumar S (2001) Mutation and linkage disequilibrium in human mtDNA. Eur J Hum Genet 9:969–972
Stephens M, Smith NJ, Donnelly P (2001) A new statistical method for haplotype reconstruction from population data. Am J Hum Genet 68:978–989
Kobayashi S, Inoue S, Hosoi T, Ouchi Y, Shiraki M, Orimo H (1996) Association of bone mineral density with polymorphism of the estrogen receptor gene. J Bone Miner Res 11:306–311
Yamada Y, Miyauchi A, Takagi Y, Tanaka M, Mizuno M, Harada A (2001) Association of the C-509-> T polymorphism, alone of in combination with the T869-> C polymorphism, of the transforming growth factor-beta1 gene with bone mineral density and genetic susceptibility to osteoporosis in Japanese women. J Mol Med 79:149–156
Keen RW, Woodford-Richens KL, Lanchbury JS, Spector TD (1998) Allelic variation at the interleukin-1 receptor antagonist gene is associated with early postmenopausal bone loss at the spine. Bone 23:367–371
Murray RE, McGuigan F, Grant SF, Reid DM, Ralston SH (1997) Polymorphisms of the interleukin-6 gene are associated with bone mineral density. Bone 21:89–92
Garcia-Giralt N, Nogues X, Enjuanes A, Puig J, Mellibovsky L, Bay-Jensen A, Carreras R, Balcells S, Diez-Perez A, Grinberg D (2002) Two new single-nucleotide polymorphisms in the COL1A1 upstream regulatory region and their relationship to bone mineral density. J Bone Miner Res 17:384–393
Arko B, Prezelj J, Komel R, Kocijancic A, Hudler P, Marc J (2002) Sequence variations in the osteoprotegerin gene promoter in patients with postmenopausal osteoporosis. J Clin Endocrinol Metab 87:4080–4084
Morrison NA, Yeoman R, Kelly PJ, Eisman JA (1992) Contribution of trans-acting factor alleles to normal physiological variability: vitamin D receptor gene polymorphism and circulating osteocalcin. Proc Natl Acad Sci USA 89:6665–6669
Morrison NA, Qi JC, Tokita A, Kelly PJ, Crofts L, Nguyen TV, Sambrook PN, Eisman JA (1994) Prediction of bone density from vitamin D receptor alleles. Nature 367:284–287
Kim GS, Koh JM, Chang JS, Park BL, Kim LH, Park EK, Kim SY, Shin HD (2005) Association of the OSCAR promoter polymorphism with BMD in postmenopausal women. J Bone Miner Res 20:1342–1348
Boyle WJ, Simonet WS, Lacey DL (2003) Osteoclast differentiation and activation. Nature 423:337–342
Bachmann MF, Wong BR, Josien R, Steinman RM, Oxenius A, Choi Y (1999) TRANCE, a tumor necrosis factor family member critical for CD40 ligand-independent T helper cell activation. J Exp Med 189:1025–1031
Anderson DM, Maraskovsky E, Billingsley WL, Dougall WC, Tometsko ME, Roux ER, Teepe MC, DuBose RF, Cosman D, Galibert L (1997) A homologue of the TNF receptor and its ligand enhance T-cell growth and dendritic-cell function. Nature 390:175–179
Yan T, Riggs BL, Boyle WJ, Khosla S (2001) Regulation of osteoclastogenesis and RANK expression by TGF-beta1. J Cell Biochem 83:320–325
Arai F, Miyamoto T, Ohneda O, Inada T, Sudo T, Brasel K, Miyata T, Anderson DM, Suda T (1999) Commitment and differentiation of osteoclast precursor cells by the sequential expression of c-Fms and receptor activator of nuclear factor kappaB (RANK) receptors. J Exp Med 190:1741–1754
Kido S, Inoue D, Iliura K, Javier W, Ito Y, Matsumoto T (2003) Expression of RANK is dependent upon differentiation into the macrophage/osteoclast lineage: induction by 1alpha, 25-dihydroxyvitamin D3 and TPA in a human myelomonocytic cell line, HL60. Bone 32:621–629
Kang HS, Park EK, Kim KH, Park JY, Choi JY, Shin HI, Jun CD, Kang SS, Kim SY (2004) Receptor activator of nuclear factor-kappaB is induced by a rottlerin-sensitive and p38 MAP kinase-dependent pathway during monocyte differentiation. Mol Cells 17(3):438–445
Ast G (2004) How did alternative splicing evolve? Nat Rev Genet 5:773–782
MacGregor A, Snieder H, Spector TD (2000) Genetic factors and osteoporotic fractures in elderly people. Twin data support genetic contribution to risk of fracture. BMJ 320:1669–1670; author reply 1670–1661
Deng HW, Chen WM, Recker S, Stegman MR, Li JL, Davies KM, Zhou Y, Deng H, Heaney R, Recker RR (2000) Genetic determination of Colles’ fracture and differential bone mass in women with and without Colles’ fracture. J Bone Miner Res 15:1243–1252
Acknowledgements
This study was supported in part by a grant from the Korea Health 21 R&D Project, Ministry of Health & Welfare, Republic of Korea (Project No.: 01-PJ3-PG6-01GN11-0002) and in part by Kyungpook National University Hospital Grant (2003).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Ad. Table 1
Frequencies of the human RANK gene polymorphisms (DOC 56.5 KB).
Ad. Table 2
Frequencies of the human RANK gene polymorphisms according to the evaluation machines (DOC 47 KB).
Ad. Table 3
Primer sequences for discovery of RNAK polymorphism (DOC 33.5 KB).
Ad. Table 4
Sequences of amplifying and TaqMan probe for RNAK single nucleotide polymorphism (SNP) genotyping (DOC 33 KB).
Rights and permissions
About this article
Cite this article
Koh, JM., Park, B.L., Kim, D.J. et al. Identification of novel RANK polymorphisms and their putative association with low BMD among postmenopausal women. Osteoporos Int 18, 323–331 (2007). https://doi.org/10.1007/s00198-006-0244-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-006-0244-5