Abstract
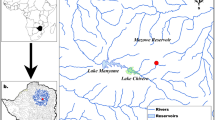
In the last decades, mega-diverse rivers worldwide (e.g., the Amazon and their tributaries) have experienced several human-driven transformations, although impact assessments oftentimes lack baseline data or reference values to better estimate observed disturbances. Herein, we assessed THg and δ15N in fish muscle tissue to determine biomagnification processes, also including seasonal assessments, concerning Hg in fish from the Belo Monte reservoir, located on the Xingu River, prior to its impoundment. A total of 312 samples from 19 fish species belonging to distinct feeding guilds were sampled. None presented THg concentrations above the threshold levels of 0.5 μg g−1 established by the World Health Organization. However, biomagnification was observed (R = 0.8; p < 0.001), also confirmed by a trophic magnification factor of 1.76 for the entire food web. The THg content in the water was less than 0.001 mg L−1 and only Hemiodus unimaculatus presented seasonal variations for THg. Therefore, at the time of this study, the Xingu River presented low Hg contamination levels in both water and target fish.


Similar content being viewed by others
References
Almeida MD, Lacerda LD, Bastos WR, Herrmann JC (2005) Mercury loss from soils following conversion from forest to pasture in Rondônia, Western Amazon, Brazil. Environ Pollut 137:179–186. https://doi.org/10.1016/j.envpol.2005.02.026
ANVISA (Agência Nacional de Vigilância Sanitária) (2013) Resolução - Rdc No 42, De 29 de Agosto de 2013. Diário Oficial da União 33–35
Arrifano GPF, Martín-Doimeadios RCR, Jiménez-Moreno M et al (2018) Large-scale projects in the Amazon and human exposure to mercury: the case-study of the Tucuruí Dam. Ecotoxicol Environ Saf 147:299–305. https://doi.org/10.1016/j.ecoenv.2017.08.048
Azevedo LS, Pestana IA, da Costa Nery AF et al (2020) Mercury concentration in six fish guilds from a floodplain lake in western Amazonia: interaction between seasonality and feeding habits. Ecol Indic 111:106056. https://doi.org/10.1016/j.ecolind.2019.106056
Azevedo-Silva CE, Almeida R, Carvalho DP et al (2016) Mercury biomagnification and the trophic structure of the ichthyofauna from a remote lake in the Brazilian Amazon. Environ Res 151:286–296. https://doi.org/10.1016/j.envres.2016.07.035
Béliveau A, Lucotte M, Davidson R et al (2017) Reduction of soil erosion and mercury losses in agroforestry systems compared to forests and cultivated fields in the Brazilian Amazon. J Environ Manag 203:522–532. https://doi.org/10.1016/j.jenvman.2017.07.037
Beltrán-Pedreros S, Forsberg BR, Beltran-pedreros S et al (2017) Mercury bioaccumulation in fish of commercial importance from different trophic categories in an Amazon floodplain lake. Neotrop Ichthyol 9:901–908. https://doi.org/10.1590/S1679-62252011000400022
Berzas-Nevado JJ, Rodríguez Martín-Doimeadios RC, Guzmán Bernardo FJ et al (2010) Mercury in the Tapajós River basin, Brazilian Amazon: A review. Environ Int 36:593–608. https://doi.org/10.1016/j.envint.2010.03.011
Bonotto DM, Wijesiri B, Vergotti M et al (2018) Assessing mercury pollution in Amazon River tributaries using a Bayesian Network approach. Ecotoxicol Environ Saf 166:354–358. https://doi.org/10.1016/j.ecoenv.2018.09.099
Brito BC, Forsberg BR, Kasper D et al (2017) The influence of inundation and lake morphometry on the dynamics of mercury in the water and plankton in an Amazon floodplain lake. Hydrobiologia 790:35–48. https://doi.org/10.1007/s10750-016-3017-y
Clayden MG, Kidd KA, Wyn B, Kirk JL, Muir DCG, O’Driscoll NJ (2013) Mercury biomagnification through food webs is affected by physical and chemical characteristics of Lakes. Environ Sci Pollut Res 47:12047–12053. https://doi.org/10.1557/proc-639-g11.59
Fearnside PM (2016) Environmental and social impacts of hydroelectric dams in Brazilian Amazonia: implications for the aluminum industry. World Dev 77:48–65. https://doi.org/10.1016/j.worlddev.2015.08.015
Feng Y, Lu D, Moran E et al (2017) Examining spatial distribution and dynamic change of urban land covers in the Brazilian Amazon using multitemporal multisensor high spatial resolution satellite imagery. Remote Sens 9:1–20. https://doi.org/10.3390/rs9040381
Ferreira da Silva S, Oliveira Lima M (2020) Mercury in fish marketed in the Amazon triple frontier and health risk assessment. Chemosphere 248:125989. https://doi.org/10.1016/j.chemosphere.2020.125989
Fisk AT, Hobson KA, Norstrom RJ (2001) Influence of chemical and biological factors on trophic transfer of persistent organic pollutants in the Northwater Polynya marine food web. Environ Sci Technol 35:732–738. https://doi.org/10.1021/es001459w
Fitzgerald DB, Sabaj Perez MH, Sousa LM et al (2018) Diversity and community structure of rapids-dwelling fishes of the Xingu River: implications for conservation amid large-scale hydroelectric development. Biol Conserv 222:104–112. https://doi.org/10.1016/j.biocon.2018.04.002
IPCS (International Programme on Chemical Safety) (1990) Methylmercury. World Health Organization, coord, Geneva
Jackson AT, Adite A, Roach KA, Winemiller KO (2013) Primary production, food web structure, and fish yields in constructed and natural wetlands in the floodplain of an African river. Can J Fish Aquat Sci 70:1–11. https://doi.org/10.1139/cjfas-2012-0403
Jardine TD, Kidd KA, O’Driscoll N (2013) Food web analysis reveals effects of pH on mercury bioaccumulation at multiple trophic levels in streams. Aquat Toxicol 132–133:46–52. https://doi.org/10.1016/j.aquatox.2013.01.013
Kasper D, Palermo EFA, Branco CWC, Malm O (2012) Evidence of elevated mercury levels in carnivorous and omnivorous fishes downstream from an Amazon reservoir. Hydrobiologia 694:87–98. https://doi.org/10.1007/s10750-012-1133-x
Kasper D, Forsberg BR, Amaral JHF et al (2014) Reservoir stratification affects methylmercury levels in river water, plankton, and fish downstream from Balbina hydroelectric dam, Amazonas, Brazil. Environ Sci Technol 48:1032–1040. https://doi.org/10.1021/es4042644
Kidd KA, Burkhard LP, Babut M, Borgå K, Muir DCG, Perceval O, Ruedel H, Woodburn K, Embry MR (2018) Practical advice for selecting or determining trophic magnification factors for application under the European Union Water Framework Directive. Integr Environ Assess Manag 15:266–277. https://doi.org/10.1002/ieam.4102
Lee S, Lee E, An K (2018) Lotic ecosystem health assessments using an integrated analytical approach of physical habitat, chemical water quality, and fish multi-metric health metrics. Pol J Environ Stud 27:2113–2131. https://doi.org/10.15244/pjoes/78044
Lees AC, Peres CA, Fearnside PM et al (2016) Hydropower and the future of Amazonian biodiversity. Biodivers Conserv 25:451–466. https://doi.org/10.1007/s10531-016-1072-3
MMA (Ministério do Meio Ambiente) (2005). Resolução nº 357, de 17 de março de 2005. Available in www.mma.gov.br/conama.
Palermo EFA, Kasper D, Reis TS et al (2004) Mercury level increase in fish tissues downstream the Tucuruí Reservoir. Brazil RMZ-M&G. https://doi.org/10.13140/2.1.2890.4644
Pérez MS (2015) Where the Xingu bends and will soon break. Am Sci 103(6):395–403. https://doi.org/10.1511/2015.117.395
Pestana IA, Azevedo LS, Bastos WR, Magalhães de Souza CM (2019) The impact of hydroelectric dams on mercury dynamics in South America: a review. Chemosphere 219:546–556. https://doi.org/10.1016/j.chemosphere.2018.12.035
Pouilly M, Pérez T, Rejas D et al (2012) Mercury bioaccumulation patterns in fish from the Iténez river basin, Bolivian Amazon. Ecotoxicol Environ Saf 83:8–15. https://doi.org/10.1016/j.ecoenv.2012.05.018
Sampaio da Silva D, Lucotte M, Roulet M et al (2005) Trophic structure and bioaccumulation of mercury in fish of three natural lakes of the Brazilian Amazon. Water Air Soil Pollut 165:77–94. https://doi.org/10.1007/s11270-005-4811-8
Siqueira GW, Aprile F, Irion G, Braga ES (2018) Mercury in the Amazon basin: human influence or natural geological pattern? J S Am Earth Sci 86:193–199. https://doi.org/10.1016/j.jsames.2018.06.017
Souza-Araujo J, Giarrizzo T, Lima MO, Souza MBG (2016) Mercury and methyl mercury in fishes from Bacajá River (Brazilian Amazon): evidence for bioaccumulation and biomagnification. J Fish Biol 89:249–263. https://doi.org/10.1111/jfb.13027
Tuomola L, Niklasson T, de Castro E, Silva E, Hylander LD (2008) Fish mercury development in relation to abiotic characteristics and carbon sources in a six-year-old, Brazilian reservoir. Sci Total Environ 390:177–187. https://doi.org/10.1016/j.scitotenv.2007.09.030
Winemiller KO, McIntyre PB, Castello L et al (2016) Balancing hydropower and biodiversity in the Amazon, Congo, and Mekong. Science 351:128–129
Acknowledgements
TG is funded by National Council for Scientific and Technological Development (CNPq # 311078/2019-2) and Norte Energia S.A. (P&D-02-2020). This study was funded by Norte Energia S.A. and Fundacão de Amparo e Desenvolvimento da Pesquisa (FADESP). TG is funded by National Council for Scientific and Technological Development (CNPq # 311078/2019-2) and Norte Energia S.A. (P&D-02-2020). This study was funded by Norte Energia S.A. and Fundacão de Amparo e Desenvolvimento da Pesquisa (FADESP).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Souza-Araujo, J., Andrades, R., Hauser-Davis, R.A. et al. Before the Dam: A Fish-Mercury Contamination Baseline Survey at the Xingu River, Amazon Basin Before the Belo Monte Dam. Bull Environ Contam Toxicol 108, 861–866 (2022). https://doi.org/10.1007/s00128-021-03371-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-021-03371-9