Abstract
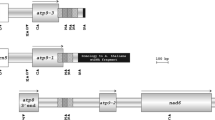
In order to determine the molecular basis of cytoplasmic male sterility (CMS) in alloplasmic lines of eggplant, the genomic structures and transcription patterns of mitochondrial ATP synthase subunit (atp) and cytochrome oxidase subunit (cox) genes were studied for wild and cultivated eggplants. Alloplasmic eggplant lines with cytoplasms of wild Solanum species showing either anther indehiscent type of CMS or non-pollen production type of CMS were studied with the cultivated eggplant Solanum melongena, used as a control. Southern hybridization of the mitochondrial genes indicated the difference between the two types of CMS and showed complete identity within each type. The cytoplasmic patterns of all wild species differed from that of the cultivated eggplant. Thus, the cytoplasm of the six wild eggplants and the one cultivated eggplant was classified into three groups. Male sterile plants of both types of CMS showed novel transcription patterns of atp1, whereas a different transcription pattern of cox2 was observed only in the anther indehiscent type. Based on these differences, we determined the DNA sequences of about a 4 kbp segment in the atp1 region. Although the coding and 3′ flanking regions were almost identical among the cytoplasms, the 5′ flanking region was completely different and novel open reading frames (orfs) were found for each of the CMS types and the cultivated eggplant. The cytoplasm of Solanum kurzii inducing the anther indehiscent type CMS had orf312, and those of Solanum aethiopicum and Solanum grandifolium of non-pollen production type CMS had orf218. The correspondence between the transcription patterns of these orfs and phenotypic expression of male sterility strongly suggests that these orfs are causal genes for each type of CMS.






Similar content being viewed by others
References
Bergman P, Edqvist J, Farbos I, Glimelius K (2002) Male-sterile tobacco displays abnormal mitochondrial atp1 transcript accumulation and reduced floral ATP/ADP ratio. Plant Mol Biol 42:531–544
Budar F, Berthomé R (2007) Cytoplasmic male sterility and mitochondrial gene mutations in plants. In: Logan DC (ed) Plant mitochondria. Blackwell Publishing, Oxford, pp 278–307
Chanut FA, Grabau EA, Gesterland RF (1993) Complex organization of the soybean mitochondrial genome: recombination repeats and multiple transcripts at the atpA loci. Curr Genet 23:234–247
Chase CD, Ortega VM (1992) Organization of ATPA coding and 3′ flanking sequences associated with cytoplasmic male sterility in Phaseolus vulgaris L. Curr Genet 22:147–153
Fang MR, Mao R, Xie W (1985) Breeding of cytoplasmically inherited male sterile lines of eggplant (Solanum melongena L.). Acta Hort Sinica 12:261–266
Hanson MR, Bentolila S (2004) Interactions of mitochondrial and nuclear genes that affect male gametophyte development. Plant Cell 16:S154–S169
Isshiki S, Kawajiri N (2002) Effect of cytoplasm of Solanum violaceum Ort. on fertility of eggplant (S. melongena L.). Sci Hortic 93:9–18
Kakizaki Y (1931) Hybrid vigor in eggplant and its practical utilization. Genetics 16:1–25
Khan MMR, Isshiki S (2008) Development of a male sterile eggplant by utilizing the cytoplasm of Solanum virginianum and a biparental transmission of chloroplast DNA in backcrossing. Sci Hortic 117:316–320
Khan MMR, Isshiki S (2009) Functional male-sterility expressed in eggplant (Solanum melongena L.) containing the cytoplasm of S. kurzii Brace & Prain. J Hortic Sci Biotech 84:92–96
Khan MMR, Isshiki S (2010a) Development of the male sterile line of eggplant using the cytoplasm of Solanum aethiopicum aculeatum group. J Jpn Soc Hortic Sci 79:348–353
Khan MMR, Isshiki S (2010b) Development of a cytoplasmic male sterile line of eggplant (Solanum melongena L.) with the cytoplasm of S. anguivi. Plant Breed 130:256–260
Kikuchi K, Honda I, Matsuo S, Fukuda M, Saito T (2008) Stability of fruit set of newly selected parthenocarpic eggplant lines. Sci Hortic 115:111–116
Kim DH, Kang JG, Kim BD (2007) Isolation and characterization of the cytoplasmic male sterility associated orf456 gene of chili pepper (Capsicum annuum L.). Plant Mol Biol 63:519–532
Laughnan SG, Newton KJ (2012) Plant mitochondrial mutations. In: Bock R, Knoop V (eds) Genomics of chloroplasts and mitochondria. Springer, Dordrecht, pp 267–291
Leino M, Landgren M, Glimelius K (2005) Alloplamic effects on mitochondrial transcriptional activity and RNA turnover result in accumulated transcripts of Arabidopsis orfs in cytoplasmic male-sterile Brassica napus. Plant J 42:469–480
Liu YG, Mitsukawa N, Oosumi T, Whittier RF (1995) Efficient isolation and mapping of Arabidopsis thaliana T-DNA insert junctions by thermal asymmetric interlaced PCR. Plant J 8:457–463
Pelletier G, Budar F (2007) The molecular biology of cytoplasmically inherited male sterility and prospects for its engineering. Curr Opin Biotech 18:121–125
Rotino GL, Perri E, Zottini M, Sommer H, Spena A (1997) Genetic engineering of parthenocarpic plants. Nat Biotech 15:1398–1401
Saito T, Matsunaga H, Saito A, Hamato N, Koga T, Suzuki T, Yoshida T (2009) A novel source of cytoplasmic male sterility and a fertility restoration gene in eggplant (Solanum melongena L.) lines. J Jpn Soc Hortic Sci 78:425–430
Acknowledgments
This work was supported by the Program for Basic and Applied Researches for Innovations in Bio-oriented Industry (BRAIN). We are grateful to Y. Yamashita for the assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Havey.
Rights and permissions
About this article
Cite this article
Yoshimi, M., Kitamura, Y., Isshiki, S. et al. Variations in the structure and transcription of the mitochondrial atp and cox genes in wild Solanum species that induce male sterility in eggplant (S. melongena). Theor Appl Genet 126, 1851–1859 (2013). https://doi.org/10.1007/s00122-013-2097-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-013-2097-6