Abstract
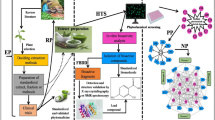
Huashanshen dripping pill (HSS), a commonly used traditional Chinese medicine, has been widely used in China due to its properties of antiasthma, expectorant, and antitussive. However, it was less in-depth understanding of the chemical composition, their absorption in plasma and potential biological mechanism. In this research, ultra performance liquid chromatography coupled with quadrupole time-of-flight mass spectrometry was developed to simultaneously identify the chemical constituents and absorption in rat plasma after oral administration of HSS. IgE-sensitized to ovalbumin was induced in BALB/c mice. ELISA and molecular docking analysis were carried out to identify their antiasthmatic mechanisms. As a result, 11 compounds were tentatively characterized by comparing the retention time, ion fragments, and the accurate mass measurement of [M + H]+ ions in the HSS. Two alkaloids including d-anisodamine and l-anisodamine were identified in the rat plasma after oral administration of HSS. The pills alleviated airway inflammation in lung tissues, and inhibited IgE, IL-4, and IL-5 in the serum. Furthermore, there were several amino acid residues involved in anisodamine-targeting receptors of hydrogen bonds and hydrophobic interactions via molecular docking indication. All in all, anisodamine would be the main component in the HSS whose antiasthmatic effects based on the inhibition of IgE, IL-4, and IL-5 production.




Similar content being viewed by others
Abbreviations
- DM:
-
dexamethasone
- ELISA:
-
enzyme-linked immunosorbent assay
- HSS:
-
huashanshen dripping pill
- IgE:
-
immunoglobulin E
- IL:
-
interleukins
- MS:
-
mass spectrometry
- OVA:
-
ovalbumin
- RT:
-
the retention time
- UHPLC-QTOF-MS:
-
ultra performance liquid chromatography coupled with quadrupole time-of-flight mass spectrometry
References
Cooper AM, Hobson PS, Jutton MR, Kao MW, Drung B, Schmidt B, Fear DJ, Beavil AJ, McDonnell JM, Sutton BJ, Gould HJ (2012) Soluble CD23 controls IgE synthesis and homeostasis in human B cells. J Immunol 188:3199–3207
Delescluse I, Mace H, Adcock JJ (2012) Inhibition of airway hyper-responsiveness by TRPV1 antagonists (SB-705498 and PF-04065463) in the unanaesthetized, ovalbumin-sensitized guinea pig. Br J Pharm 166:1822–1832
Dhaliwal B, Yuan D, Pang MO, Henry AJ, Cain K, Oxbrow A, Fabiane SM, Beavil AJ, McDonnell JM, Gould HJ, Sutton BJ (2012) Crystal structure of IgE bound to its B-cell receptor CD23 reveals a mechanism of reciprocal allosteric inhibition with high affinity receptor FcepsilonRI. Proc Natl Acad Sci USA 109:12686–12691
Foster PS, Maltby S, Rosenberg HF, Tay HL, Hogan SP, Collison AM, Yang M, Kaiko GE, Hansbro PM, Kumar RK, Mattes J (2017) Modeling TH 2 responses and airway inflammation to understand fundamental mechanisms regulating the pathogenesis of asthma. Immunol Rev 278:20–40. https://doi.org/10.1111/imr.12549
Hall S, Agrawal DK (2014) Key mediators in the immunopathogenesis of allergic asthma. Int Immunopharmacol 23:316–329
Holdom MD, Davies AM, Nettleship JE, Bagby SC, Dhaliwal B, Girardi E, Hunt J, Gould HJ, Beavil AJ, McDonnell JM, Owens RJ, Sutton BJ (2011) Conformational changes in IgE contribute to its uniquely slow dissociation rate from receptor FcvarepsilonRI. Nat Struct Mol Biol 18:571–576
Hirose K, Iwata A, Tamachi T, Nakajima H (2017) Allergic airway inflammation: key players beyond the Th2 cell pathway. Immunological Rev 278:145–161
Kistemaker LE, Bos IS, Menzen MH, Maarsingh H, Meurs H, Gosens R (2016) Combination therapy of tiotropium and ciclesonide attenuates airway inflammation and remodeling in a guinea pig model of chronic asthma. Respir Res 17:13
Kohnen-Johannsen KL, Kayser O (2019) Tropane alkaloids: chemistry, pharmacology, biosynthesis and production. Molecules 24:E796
Kruse RL, Vanijcharoenkarn K (2018) Drug repurposing to treat asthma and allergic disorders: progress and prospects. Allergy 73:313–322
Matucci A, Vultaggio A, Maggi E, Kasujee I (2018) Is IgE or eosinophils the key player in allergic asthma pathogenesis? Are we asking the right question? Respir Res 19:113
Pelaia G, Canonica GW, Matucci A, Paolini R, Triggiani M, Paggiaro P (2017) Targeted therapy in severe asthma today: focus on immunoglobulin E. Drug Des Devel Ther 11:1979–1987
Pennington LF, Tarchevskaya S, Brigger D, Sathiyamoorthy K, Graham MT, Nadeau KC, Eggel A, Jardetzky TS (2016) Structural basis of omalizumab therapy and omalizumab-mediated IgE exchange. Nat Commun 7:11610
Steinke JW, Borish L (2001) Th2 cytokines and asthma. Interleukin-4: its role in the pathogenesis of asthma, and targeting it for asthma treatment with interleukin-4 receptor antagonists. Respir Res 2:66–70
Verbout NG, Jacoby DB, Gleich GJ, Fryer AD (2009) Atropine-enhanced, antigen challenge-induced airway hyperreactivity in guinea pigs is mediated by eosinophils and nerve growth factor. Am J Physiol Lung Cell Mol Physiol 297:L228–L237
Verbout NG, Lorton JK, Jacoby DB, Fryer AD (2007) Atropine pretreatment enhances airway hyperreactivity in antigen-challenged guinea pigs through an eosinophil-dependent mechanism. Am J Physiol Lung Cell Mol Physiol 292:L1126–L1135
Wang YH, Zeng KW (2019) Natural products as a crucial source of anti-inflammatory drugs: recent trends and advancements. J Tradit Chin Med 4:257–268
Xu GN, Yang K, Xu ZP, Zhu L, Hou LN, Qi H, Chen HZ, Cui YY (2012) Protective effects of anisodamine on cigarette smoke extract-induced airway smooth muscle cell proliferation and tracheal contractility. Toxicol Appl Pharm 262:70–79
Xu ZP, Wang H, Hou LN, Xia Z, Zhu L, Chen HZ, Cui YY (2011) Modulatory effect of anisodamine on airway hyper-reactivity and eosinophilic inflammation in a murine model of allergic asthma. Int Immunopharmacol 11:260–265
Zhou M, Ma X, Sun J, Ding G, Cui Q, Miao Y, Hou Y, Jiang M, Bai G (2017) Active fragments-guided drug discovery and design of selective tropane alkaloids using ultra-high performance liquid chromatography-quadrupole time-of-flight tandem mass spectrometry coupled with virtual calculation and biological evaluation. Anal Bioanal Chem 409:1145–1157
Acknowledgements
This work was supported by grants 81673647, 81673535, and 81503086 from the National Natural Science Foundation of China.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Man, S., Cui, N., Liu, X. et al. Component analysis and antiasthmatic effects of Huashanshen dripping pill. Med Chem Res 29, 75–82 (2020). https://doi.org/10.1007/s00044-019-02460-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-019-02460-z