Abstract
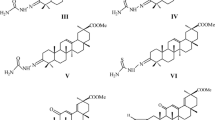
New 3-amino substituted derivatives, containing linear, aromatic, and heterocyclic fragments, as well as conjugates with biogenic amines—spermine and spermidine, were synthesized from 3,11-dioxo analogs of fusidic acid and its methyl ester. Antitumor activity of the compounds was studied in vitro towards the 60 cell lines of nine different types of human tumors of the NCI collection. Introduction of pyrrolidine, n-butylamine, benzylamine, and ethylenediamine substituents into the molecules were found to provide a pronounced selective effect on five cell lines of leukemia: HL-60, K-562, MOLT-4, RPMI-8226, and SR, inhibiting their growth from 70% to complete death of cancer cells. Methylfusidate derivative with a spermine fragment was shown to exhibit the widest spectrum of antiproliferative action among the obtained compounds, inhibiting the growth of leukemia, NSC lung cancer, colon cancer, and melanoma cell lines of 68–92%.


Similar content being viewed by others
References
Abdel-Magid AF, Carson KG, Harris BD, Maryanoff CA, Shah RD (1996) Reductive amination of aldehydes and ketones with sodium triacetoxyborohydride. Studies on direct and indirect reductive amination procedure. J Org Chem 61:3849–3862
Agostinelli E, Arancia G, Vedova LD, Belli F, Marra M, Salvi M, Toniello A (2004) The biological functions of polyamine oxidation products by amine oxidases: Perspectives of clinical applications. Amino Acids 27:347–358
Bachrach U, Heimer YM (1989) The Physiology of Polyamines. CRC Press, Florida
Boyd MR, Paul KD (1995) Some practical considerations and applications of the National Cancer Institute in vitro anticancer drug discovery screen. Drug Res Rep 34:91–109
Buyel JF (2018) Plants as sources of natural and recombinant anti-cancer agents. Biotechnol Adv 36:506–520
Cicek-Saydam C, Cavusoglu C, Burhanoglu D, Hilmioglu S, Ozkalay N, Bilgic A (2001) In vitro susceptibility of Mycobacterium tuberculosis to fusidic acid. Clin Microbiol Infect 7:700–702
Faber V, Dalgleish AG, Newell A, Malkovsky M (1987) Inhibition of HIV replication in vitro by fusidic acid. Lancet 10:827–828
Gerner EW, Meyskens FL Jr. (2004) Polyamines and cancer: old molecules, new understanding. Nat Rev Cancer 4:781–792
Gilchrist SE, Lange D, Letchford K, Bach H, Fazli L, Burt HM (2013) Fusidic acid and rifampicin co-loaded PLGA nanofibers for the prevention of orthopedic implant associated infections. J Control Release 170:64–73
Golledge C (1999) Fusidic acid in other infections. Int J Antimicrob Agents 12:11–15
Grever MR, Schepartz SA, Chabner BA (1992) The National Cancer Institute: cancer drug discovery and development program. Semin Oncol 19:622–638
Kilic FS, Erol K, Batu O, Yildirim E, Usluer G (2002) The effects of fusidic acid on the inflammatory response in rats. Pharm Res 45:265–267
Kucers A, Bennett NMck (1997) The use of antibiotics. Butterworth–Heinemann, London
Ni J, Guo M, Cao Y, Lei L, Liu K, Wang B, Lu F, Zhai R, Gao X, Yan C, Wang H, Bi Y (2019) Discovery, synthesis of novel fusidic acid derivatives possessed amino-terminal groups at the 3-hydroxyl position with anticancer activity. Eur J Med Chem 162:122–131
Loncle C, Salmi C, Letourneux Y, Brunel JM (2007) Synthesis of new 7-aminosterol squalamine analogues with high antimicrobial activities through a stereoselective titanium reductive amination reaction. Tetrahedron 63:12968–12974
Markman JL, Rekechenetskiy A, Holler E, Ljubimova JY (2013) Nanomedicine therapeutic approaches to overcome cancer drug resistance. Adv Drug Deliv Rev 65:1866–1879
Mazumder A, Cerella C, Diederich M (2018) Natural scaffolds in anticancer therapy and precision medicine. Biotechnol Adv 36:1563–1585
Monks A, Scudiero D, Skehan P, Shoemaker R, Paull KD, Vistica D, Hose C, Langley J, Cronise P, Vaigro-Wolff A, Gray-Goodrich M, Campbell H, Mayo J, Boyd MJ (1991) Feasibility of a highflux anticancer drug screen using a diverse panel of cultured human tumor cell lines. Nat Cancer Inst 183:757–766
Reynolds JIF (1996) The extra pharmacopeia. Royal Pharmaceutical Society, London
Salama AA, AbouLaila M, Moussa AA, Nayel MA, El-Sify A, Terkawi MA, Hassan HY, Yokoyama N, Igarashi I (2013) Evaluation of in vitro and in vivo inhibitory effects of fusidic acid on Babesia and Theileria parasites. Vet Parasitol 191:1–10
Salimova EV, Mamaev AG, Tretyakova EV, Kukovinets OS, Mavzyutov AR, Shvets KYu, Parfenova LV (2018a) Synthesis and biological activity of fusidic acid cyanoethyl derivatives. Russ J Org Chem 54:1411–1418
Salimova EV, Mamaev AG, Tretyakova EV, Kukovinets OS, Parfenova LV (2018b) Reductive amination of fusidane triterpenoid ketones. Mediterr J Chem 7:198–203
Singh K, Espinoza-Moraga M, Njoroge M, Kaur G, Okombo J, De Kock C, Smith PJ, Wittlin S, Chibale K (2017) Synthesis and biological characterisation of ester and amide derivatives of Fusidic acid as antiplasmodial agents. BMC Lett 27:658–661
Skehan P, Storeng R, Scudiero D, Monks A, McMahon J, Vistica D, Warren JR, Bokesch H, Kenney S, Boyd MR (1990) New colorimetric cytotoxicity assay for anticancer-drug screening. J Natl Cancer Inst 82:1107–1112
Speck-Planche A (2019) Multiple perspectives in anti-cancer drug discovery: from old targets and natural products to innovative computational approaches. Anti-Cancer Agents Med Chem 19:146–147
Thomas T, Thomas TJ (2001) Polyamines in cell growth and cell death: molecular mechanisms and therapeutic applications. Cell Mol Life Sci 58:244–258
Tripathi VC, Satish S, Horam S, Raj S, lal A, Arockiaraj J, Pasupuleti M, Dikshit DK (2018) Natural products from polar organisms: Structural diversity, bioactivities and potential pharmaceutical applications. Polar Sci 18:147–166
Tyrrell DA (1969) The possible chemotherapy of respiratory virus diseases. Sci Basis Med Ann Rev 1969:294–319
Woster P, Casero R (2011) Polyamine Drug Discovery. CRC Press, Florida
Yatin M (2002) Polyamines in living organisms. J Cell Mol Biol 1:57–67
Acknowledgements
This work was financially supported by the Russian Foundation of Basic Research (research project No. 17-43-020021 r_a). The part of the research was carried out in accordance with federal program No. АААА-А19-119022290012-3. The structural studies of compounds (3)–(20) were carried out at the Center for Collective Use “Agidel” at the Institute of Petrochemistry and Catalysis, Russian Academy of Sciences. We thank National Cancer Institute for the screening of cytotoxicity of compounds 3–20.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
About this article
Cite this article
Salimova, E.V., Tret’yakova, E.V. & Parfenova, L.V. Synthesis and cytotoxic activity of 3-amino substituted fusidane triterpenoids. Med Chem Res 28, 2171–2183 (2019). https://doi.org/10.1007/s00044-019-02445-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-019-02445-y