Abstract
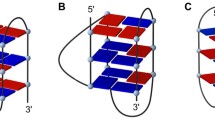
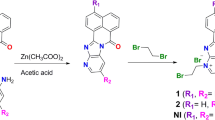
Neomycin is known to preferentially bind to A-form nucleic acid structures including triplex DNA, DNA and RNA hybrid, and duplex RNA. Tethering a DNA intercalator moiety to the C5” position of the ring III of neomycin is a practical approach to develop potent binders targeting various nucleic acid secondary structures via a synergistic effect; however, the minimal stacking surface of the intercalating moiety required to exhibit the effect remains unclear. In the present work, we synthesized four nucleobase and neomycin conjugates via click chemistry. All four conjugates stabilized a DNA oligonucleotide triplex in the thermal denaturation experiments monitored by UV. The guanine-neomycin conjugate (6b) showed a better triplex stabilization effect than neomycin. All the conjugates, as well as neomycin, exhibited no thermal stabilization effect on a human telomeric DNA G-quadruplex. These results suggest that the synergistic effect of binding is vastly dependent on the surface area of the stacking moiety of the conjugates. In addition, tethering a nucleobase to the C5” position of neomycin enhanced the cytotoxicity of neomycin toward MCF-7 and HeLa cancer cells but decreased the antibacterial effect of neomycin against several Gram-negative and Gram-positive bacterial species.





Similar content being viewed by others
Abbreviations
- Neo:
-
neomycin
- rRNA:
-
ribosomal RNA
- TAR:
-
trans-activation response element
- AMEs:
-
aminoglycoside-modifying enzymes
- BQQ:
-
benzo[f]quino[3,4-b]quinoxaline
- DMSO:
-
dimethyl sulfoxide
- MTT:
-
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- IC50 :
-
half maximal inhibitory concentration
- MIC:
-
minimum inhibitory concentration
- NMR:
-
nuclear magnetic resonance
- UHPLC:
-
ultra-high-performance liquid chromatography
- IR:
-
infrared
- ESI:
-
electrospray ionization
- HRMS:
-
high-resolution mass spectrometry
- TOF:
-
time of flight
- FBS:
-
fetal bovine serum
- DMEM:
-
Dulbecco’s modified eagle’s medium
- KB test:
-
Kirby–Bauer test
- OD:
-
optical density
References
Ambrus A, Chen D, Dai J, Bialis T, Jones RA, Yang D (2006) Human telomeric sequence forms a hybrid-type intramolecular g-quadruplex structure with mixed parallel/antiparallel strands in potassium solution. Nucleic Acids Res 34:2723–2735
Arya DP, Micovic L, Charles I, Coffee Jr. RL, Willis B, Xue L (2003a) Neomycin binding to Watson-Hoogsteen (W-H) DNA triplex groove: a model. J Am Chem Soc 125:3733–3744
Arya DP, Xue L, Tennant P (2003b) Combining the best in triplex recognition: synthesis and nucleic acid binding of a BQQ-neomycin conjugate. J Am Chem Soc 125:8070–8071
Arya DP, Xue L, Willis B (2003c) Aminoglycoside (neomycin) preference is for a-form nucleic acids, not just RNA: results for a competition dialysis study. J Am Chem Soc 125:10148–10149
Balouiri M, Sadiki M, Ibnsouda SK (2016) Methods for in vitro evaluating antimicrobial activity: a review. J Pharm Anal 6:71–79
Blount KF, Tor Y (2006) A tale of two targets: differential RNA selectivity of nucleobase-aminoglycoside conjugates. ChemBioChem 7:1612–1621
Degtyareva NN, Gong C, Story S, Levinson NS, Oyelere AK, Green KD, Garneau-Tsodikova S, Arya DP (2017) Antimicrobial activity, AME resistance, and A-site binding studies of anthraquinone-neomycin conjugates. ACS Infect Dis 3:206–215
Edward Lindsell W, Murray C, Preston PN, Woodman TAJ (2000) Synthesis of 1,3-diynes in the purine, pyrimidine, 1,3,5-triazine and acridine series. Tetrahedron 56:1233–1245
Fourmy D, Recht MI, Puglisi JD (1998) Binding of neomycin-class aminoglycoside antibiotics to the a-site of 16 s rrna. J Mol Biol 277:347–362
Fridman M, Belakhov V, Yaron S, Baasov T (2003) A new class of branched aminoglycosides: pseudo-pentasaccharide derivatives of neomycin b. Org Lett 5:3575–3578
Grau-Campistany A, Massaguer A, Carrion-Salip D, Barragan F, Artigas G, Lopez-Senin P, Moreno V, Marchan V (2013) Conjugation of a ru(ii) arene complex to neomycin or to guanidinoneomycin leads to compounds with differential cytotoxicities and accumulation between cancer and normal cells. Mol Pharm 10:1964–1976
Green KD, Chen W, Houghton JL, Fridman M, Garneau-Tsodikova S (2010) Exploring the substrate promiscuity of drug-modifying enzymes for the chemoenzymatic generation of n-acylated aminoglycosides. ChemBioChem 11:119–126
Guckian KM, Schweitzer BA, Ren RX, Sheils CJ, Tahmassebi DC, Kool ET (2000) Factors contributing to aromatic stacking in water: evaluation in the context of DNA. J Am Chem Soc 122:2213–2222
Guthrie OW (2008) Aminoglycoside induced ototoxicity. Toxicology 249:91–96
Houghton JL, Green KD, Chen W, Garneau-Tsodikova S (2010) The future of aminoglycosides: the end or renaissance? ChemBioChem 11:880–902
Kirk SR, Luedtke NW, Tor Y (2000) Neomycin−acridine conjugate: a potent inhibitor of rev-rre binding. J Am Chem Soc 122:980–981
Liu M, Haddad J, Azucena E, Kotra LP, Kirzhner M, Mobashery S (2000) Tethered bisubstrate derivatives as probes for mechanism and as inhibitors of aminoglycoside 3‘-phosphotransferases. J Org Chem 65:7422–7431
Liu W, Minier MA, Franz AH, Curtis M, Xue L (2011) Synthesis of nucleobase-calix[4]arenes via click chemistry and evaluation of their complexation with alkali metal ions and molecular assembly. Supramol Chem 23:806–818
Liu W, Wang S, Dotsenko IA, Samoshin VV, Xue L (2017) Arylsulfanyl groups-suitable side chains for 5-substituted 1,10-phenanthroline and nickel complexes as g4 ligands and telomerase inhibitors. J Inorg Biochem 173:12–20
Lu W, Sengupta S, Petersen JL, Akhmedov NG, Shi X (2007) Mitsunobu coupling of nucleobases and alcohols: An efficient, practical synthesis for novel nonsugar carbon nucleosides. J Org Chem 72:5012–5015
Oliveira JF, Silva CA, Barbieri CD, Oliveira GM, Zanetta DM, Burdmann EA (2009) Prevalence and risk factors for aminoglycoside nephrotoxicity in intensive care units. Antimicrob Agents Chemother 53:2887–2891
Ramirez MS, Tolmasky ME (2010) Aminoglycoside modifying enzymes. Drug Resist Updat 13:151–171
Vakulenko SB, Mobashery S (2003) Versatility of aminoglycosides and prospects for their future. Clin Microbiol Rev 16:430–450
Wellenzohn B, Flader W, Winger RH, Hallbrucker A, Mayer E, Liedl KR (2001) Exocyclic groups in the minor groove influence the backbone conformation of DNA. Nucleic Acids Res 29:5036–5043
Wong C-H, Hendrix M, Scott Priestley E, Greenberg WA (1998) Specificity of aminoglycoside antibiotics for the A-site of the decoding region of ribosomal RNA. Chem Biol 5:397–406
Xue L, Charles I, Arya DP (2002) Pyrene–neomycin conjugate: dual recognition of a DNA triple helix. Chem Commun 70–71
Xue L, Ling M, Wang S (2017) A reliable method to measure the melting temperatures of DNA oligonucleotide triplexes. J Undergrad Chem Res 16:19–23
Xue L, Ranjan N, Arya DP (2011) Synthesis and spectroscopic studies of the aminoglycoside (neomycin)--perylene conjugate binding to human telomeric DNA. Biochemistry 50:2838–2849
Zhang J, Keller K, Takemoto JY, Bensaci M, Litke A, Czyryca PG, Chang CW (2009) Synthesis and combinational antibacterial study of 5”-modified neomycin. J Antibiot 62:539–544
Acknowledgements
This work was supported by the University of the Pacific. We also thank Dr. William K. Chan for providing MCF-7 and HeLa cells.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
These authors contributed equally: Siwen Wang, Mandeep Singh.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Wang, S., Singh, M., Ling, M. et al. Synthesis of nucleobase-neomycin conjugates and evaluation of their DNA binding, cytotoxicities, and antibacterial properties. Med Chem Res 27, 1517–1527 (2018). https://doi.org/10.1007/s00044-018-2169-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-018-2169-x