Abstract
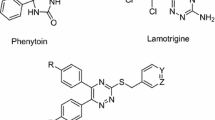
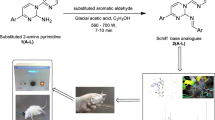
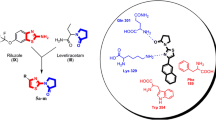
Novel Schiff bases of 1-(2-Aminoethyl)piperidine-3-carboxylic acid were synthesized, characterized and screened for anticonvulsant activity. Compounds were evaluated for in vitro blood–brain barrier (BBB) permeability by parallel artificial membrane permeability BBB assay (PAMPA-BBB). Compounds 5d, 5f, 5j, 5l, 5m, 5n, 5w, 5x and 5y elicited considerable in vitro permeability across BBB and further screened for in vivo anticonvulsant activity by sc-PTZ and DMCM-induced seizure models. The outcome of the in vivo models suggested that 5d, 5w, and 5y were most potent amongst the synthesized compounds. The neurotoxicity evaluation of 5d, 5w, and 5y by rotarod indicates no impairment of muscle coordination in comparison to standard diazepam. The MTT assay revealed that the test compounds (5d, 5w, and 5y) were not found to alter the cell viability considerably. In silico molecular docking and dynamics simulations were carried out on the homology modeled protein of human GABA transporter 1 (GAT1), which exhibited complementary interactions of compound 5w within the active binding pocket.








Similar content being viewed by others
References
Ali FE, Bondinell WE, Dandridge PA, Frazee JS, Garvey E, Girard GR, Kaiser C, Ku TW, Lafferty JJ, Moonsammy GI (1985) Orally active and potent inhibitors of gamma-aminobutyric acid uptake. J Med Chem 28:653–660
Aly MM, Mohamed YA, El-Bayouki KA, Basyouni WM, Abbas SY (2010) Synthesis of some new 4 (3H)-quinazolinone-2-carboxaldehyde thiosemicarbazones and their metal complexes and a study on their anticonvulsant, analgesic, cytotoxic and antimicrobial activities–Part-1. Eur J Med Chem 45:3365–3373
Ambrósio AF, Silva AP, Araújo I, Malva JO, Soares-da-Silva Pc, Carvalho AP, Carvalho CM (2000) Neurotoxic/neuroprotective profile of carbamazepine, oxcarbazepine and two new putative antiepileptic drugs, BIA 2-093 and BIA 2-024. Eur J Pharmacol 406:191–201
Andersen KE, Lau J, Lundt BF, Petersen H, Huusfeldt PO, Suzdak PD, Swedberg MD (2001a) Synthesis of novel GABA uptake inhibitors. Part 6: preparation and evaluation of N-Ω asymmetrically substituted nipecotic acid derivatives. Bioorg Med Chem 9:2773–2785
Andersen KE, Sørensen JL, Lau J, Lundt BF, Petersen H, Huusfeldt PO, Suzdak PD, Swedberg MD (2001b) Synthesis of novel γ-aminobutyric acid (GABA) uptake inhibitors. 5. 1 Preparation and structure− activity studies of tricyclic analogues of known GABA uptake inhibitors. J Med Chem 44:2152–2163
Araújo IM, Ambrósio AF, Leal EC, Verdasca MJ, Malva JO, Soares‐da‐Silva P, Carvalho AP, Carvalho CM (2004) Neurotoxicity induced by antiepileptic drugs in cultured hippocampal neurons: a comparative study between carbamazepine, oxcarbazepine, and two new putative antiepileptic drugs, BIA 2‐024 and BIA 2‐093. Epilepsia 45:1498–1505
Banerjee AG, Das N, Shengule SA, Sharma PA, Srivastava RS, Shrivastava SK (2016) Design, synthesis, evaluation and molecular modelling studies of some novel 5, 6-diphenyl-1, 2, 4-triazin-3 (2H)-ones bearing five-member heterocyclic moieties as potential COX-2 inhibitors: a hybrid pharmacophore approach. Bioorg Chem 69:102–120
Bhat MA, Al-Omar MA (2011) Synthesis, characterization and in vivo anticonvulsant and neurotoxicity screening of Schiff bases of phthalimide. Acta Pol Pharm 68:375–380
Bjorge S, Black A, Bockbrader H, Chang T, Gregor VE, Lobbestael SJ, Nugiel D, Pavia MR, Radulovic L, Woolf T (1990) Synthesis and metabolic profile of Cl‐966: a potent, orally‐active inhibitor of GABA uptake. Drug Dev Res 21:189–193
Braestrup C, Nielsen EB, Sonnewald U, Knutsen LJ, Andersen KE, Jansen JA, Frederiksen K, Andersen PH, Mortensen A, Suzdak PD (1990) R)‐N‐[4, 4‐Bis (3‐Methyl‐2‐Thienyl) but‐3‐en‐1‐yl] nipecotic acid binds with high affinity to the brain γ‐aminobutyric acid uptake carrier. J Neurochem 54:639–647
Chen N-H, Reith ME, Quick MW (2004) Synaptic uptake and beyond: the sodium-and chloride-dependent neurotransmitter transporter family SLC6. Pflüg Arch 447:519–531
Das N, Garabadu D, Banerjee AG, Krishnamurthy S, Shrivastava SK (2014) Synthesis and pharmacological evaluation of some N3-aryl/heteroaryl-substituted 2-(2-chlorostyryl)-6, 7-dimethoxy-quinazolin-4 (3H)-ones as potential anticonvulsant agents. Med Chem Res 23:4167–4176
Dhar TM, Borden LA, Tyagarajan S, Smith KE, Branchek TA, Weinshank RL, Gluchowski C (1994) Design, synthesis and evaluation of substituted triarylnipecotic acid derivatives as GABA uptake inhibitors: identification of a ligand with moderate affinity and selectivity for the cloned human GABA transporter GAT-3. J Med Chem 37:2334–2342
Di L, Kerns EH, Fan K, McConnell OJ, Carter GT (2003) High throughput artificial membrane permeability assay for blood–brain barrier. Eur J Med Chem 38:223–232
El-Helby AGA, Ayyad RR, Sakr HM, Abdelrahim AS, El-Adl K, Sherbiny FS, Eissa IH, Khalifa MM (2017) Design, synthesis, molecular modeling and biological evaluation of novel 2, 3-dihydrophthalazine-1, 4-dione derivatives as potential anticonvulsant agents. J Mol Struct 1130:333–351
Falch E, Krogsgaard-Larsen P (1989) GABA uptake inhibitors containing mono-and diarylmethoxyalkyl N-substituents. Drug Des Deliv 4:205–215
Gao X-M, Chuang D-M (1992) Carbamazepine-induced neurotoxicity and its prevention by NMDA in cultured cerebellar granule cells. Neurosci Lett 135:159–162
Gao X-M, Margolis RL, Leeds P, Hough C, Post RM, Chuang D-M (1995) Carbamazepine induction of apoptosis in cultured cerebellar neurons: effects ofN-methyl-d-aspartate, aurintricarboxylic acid and cycloheximide. Brain Res 703:63–71
Ghadimi S, Latif Mousavi S, Javani Z (2008) Synthesis, lipophilicity study and in vitro evaluation of some rodenticides as acetylcholinesterase reversible inhibitors. J Enzyme Inhib Med Chem 23:213–217
Ghareb N, Daim MMA, El-Sayed NM, Elgawish MS (2017) Synthesis, molecular modelling, and preliminary anticonvulsant activity evaluation of novel naphthalen-2-yl acetate and 1, 6-dithia-4, 9-diazaspiro [4.4] nonane-3, 8-dione derivatives. Bioorg Chem 71:110–119
Holmes GL (1995) Role of glutamate and GABA in the pathophysiology of epilepsy. Dev Disabil Res Rev 1:208–219
Jurik A, Reicherstorfer R, Zdrazil B, Ecker GF (2013) Classification of high‐activity tiagabine analogs by binary QSAR modeling. Mol Inform 32:415–419
Jurik A, Zdrazil B, Holy M, Stockner T, Sitte HH, Ecker GF (2015) A binding mode hypothesis of tiagabine confirms liothyronine effect on γ-aminobutyric acid transporter 1 (GAT1). J Med Chem 58:2149–2158
Kälviäinen R (2001) Long‐term safety of Tiagabine. Epilepsia 42:46–48
Kim M, Cho AE (2016) Incorporating QM and solvation into docking for applications to GPCR targets. Phys Chem Chem Phys 18:28281–28289
Kowalczyk P, Sałat K, Höfner GC, Mucha M, Rapacz A, Podkowa A, Filipek B, Wanner KT, Kulig K (2014) Synthesis, biological evaluation and structure–activity relationship of new GABA uptake inhibitors, derivatives of 4-aminobutanamides. Eur J Med Chem 83:256–273
Krogsgaard-Larsen P (1980) Inhibitors of the GABA uptake systems. Mol Cell Biochem 31:105–121
Krogsgaard‐Larsen P, Johnston G (1975) Inhibition of GABA uptake in rat brain slices by nipecotic acid, various isoxazoles and related compounds. J Neurochem 25:797–802
Kulkarni A, Wankhede S, Dhawale N, Yadav P, Deore V, Gonjari I (2017) Synthesis, characterization and biological behavior of some Schiff’s and Mannich base derivatives of Lamotrigine. Arab J Chem 10:S184–S189
Lim S-W, Loh H-S, Ting K-N, Bradshaw TD, Allaudin ZN (2015) Reduction of MTT to purple formazan by vitamin E isomers in the absence of cells. Trop Life Sci Res 26:111
Liu Y, Wang X-y, Li D, Yang L, Huang S-p (2015) Short-term use of antiepileptic drugs is neurotoxic to the immature brain. Neural Regen Res 10:599
Löscher W (1985) Anticonvulsant action in the epileptic gerbil of novel inhibitors of GABA uptake. Eur J Pharmacol 110:103–108
Meena P, Nemaysh V, Khatri M, Manral A, Luthra PM, Tiwari M (2015) Synthesis, biological evaluation and molecular docking study of novel piperidine and piperazine derivatives as multi-targeted agents to treat Alzheimer’s disease. Bioorg Med Chem 23:1135–1148
Murali Dhar T, Nagarathnam D, Marzabadi MR, Lagu B, Wong WC, Chiu G, Tyagarajan S, Miao SW, Zhang F, Sun W (1999) Design and synthesis of novel α1a adrenoceptor-selective antagonists. 2. Approaches to eliminate opioid agonist metabolites via modification of linker and 4-methoxycarbonyl-4-phenylpiperidine moiety. J Med Chem 42:4778–4793
Nielsen EB, Suzdak PD, Andersen KE, Knutsen LJ, Sonnewald U, Braestrup C (1991) Characterization of tiagabine (NO-328), a new potent and selective GABA uptake inhibitor. Eur J Pharmacol 196:257–266
Nonaka S, Katsube N, Chuang D-M (1998) Lithium protects rat cerebellar granule cells against apoptosis induced by anticonvulsants, phenytoin and carbamazepine. J Pharmacol Exp Ther 286:539–547
Ortinski P, Meador KJ (2004) Cognitive side effects of antiepileptic drugs. Epilepsy Behav 5:60–65
Pandeya SN, Rajput N (2012) Synthesis and anticonvulsant activity of various Mannich and Schiff bases of 1,5-benzodiazepines. Int J Med Chem 2012:1–10
Pavia MR, Lobbestael SJ, Nugiel D, Mayhugh DR, Gregor VE, Taylor CP, Schwarz RD, Brahce L, Vartanian MG (1992) Structure-activity studies on benzhydrol-containing nipecotic acid and guvacine derivatives as potent, orally-active inhibitors of GABA uptake. J Med Chem 35:4238–4248
Petersen EN (1983) DMCM: a potent convulsive benzodiazepine receptor ligand. Eur J Pharmacol 94:117–124
Petrera M, Wein T, Allmendinger L, Sindelar M, Pabel J, Höfner G, Wanner KT (2016) Development of highly potent GAT1 inhibitors: synthesis of nipecotic acid derivatives by Suzuki–Miyaura cross‐coupling reactions. ChemMedChem 11:519–538
Petroff OA, Rothman DL, Behar KL, Mattson RH (1996) Low brain GABA level is associated with poor seizure control. Ann Neurol 40:908–911
Prescott L (1997) Tiagabine offers effective seizure control. Inpharma Wkly 1085:15–16
Quandt G, Höfner G, Wanner KT (2013) Synthesis and evaluation of N-substituted nipecotic acid derivatives with an unsymmetrical bis-aromatic residue attached to a vinyl ether spacer as potential GABA uptake inhibitors. Bioorg Med Chem 21:3363–3378
Regulska M, Pomierny B, Basta-Kaim A, Starek A, Filip M, Lasoń W, Budziszewska B (2010) Effects of ethylene glycol ethers on cell viability in the human neuroblastoma SH-SY5Y cell line. Pharmacol Rep 62:1243–1249
Shidore M, Machhi J, Shingala K, Murumkar P, Sharma MK, Agrawal N, Tripathi A, Parikh Z, Pillai P, Yadav MR (2016) Benzylpiperidine-linked Diarylthiazoles as potential anti-Alzheimer’s agents: synthesis and biological evaluation. J Med Chem 59:5823–5846
Siddiqui N, Alam MS, Sahu M, Naim MJ, Yar MS, Alam O (2017) Design, synthesis, anticonvulsant evaluation and docking study of 2-[(6-substituted benzo [d] thiazol-2-ylcarbamoyl) methyl]-1-(4-substituted phenyl) isothioureas. Bioorg Chem 71:230–243
Singh RB, Singh GK, Chaturvedi K, Kumar D, Singh SK, Zaman MK (2017) Design, synthesis, characterization, and molecular modeling studies of novel oxadiazole derivatives of nipecotic acid as potential anticonvulsant and antidepressant agents. Med Chem Res 27:137–152
Skovstrup S, Taboureau O, Bräuner‐Osborne H, Jørgensen FS (2010) Homology modelling of the GABA transporter and analysis of tiagabine binding. ChemMedChem 5:986–1000
Suzdak PD, Jansen JA (1995) A review of the preclinical pharmacology of tiagabine: a potent and selective anticonvulsant GABA uptake inhibitor. Epilepsia 36:612–626
Sveinbjornsdottir S, Sander J, Patsalos P, Upton D, Thompson P, Duncan J (1994) Neuropsychological effects of tiagabine, a potential new antiepileptic drug. Seizure 3:29–35
Swathi K, Sarangapani M (2015) Synthesis and antiepileptic activity of Schiff’s bases of dialkylamino alkoxy isatin derivatives. GeNeDis 2014. Springer International Publishing, Swizerland
Taylor CP, Vartanian MG, Schwarz RD, Rock DM, Callahan MJ, Davis MD (1990) Pharmacology of Cl‐966: A potent GABA uptake inhibitor, in vitro and in experimental animals. Drug Dev Res 21:195–215
Thompson SM, Gahwiler B (1992) Effects of the GABA uptake inhibitor tiagabine on inhibitory synaptic potentials in rat hippocampal slice cultures. J Neurophysiol 67:1698–1701
Villalba ML, Enrique AV, Higgs J, Castaño RA, Goicoechea S, Taborda FD, Gavernet L, Lick ID, Marder M, Blanch LEB (2016) Novel sulfamides and sulfamates derived from amino esters: synthetic studies and anticonvulsant activity. Eur J Pharmacol 774:55–63
Wang KH, Penmatsa A, Gouaux E (2015) Neurotransmitter and psychostimulant recognition by the dopamine transporter. Nature 521:322–327
Yunger L, Fowler P, Zarevics P, Setler P (1984) Novel inhibitors of gamma-aminobutyric acid (GABA) uptake: anticonvulsant actions in rats and mice. J Pharmacol Exp Ther 228:109–115
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Seth, A., Sharma, P.A., Tripathi, A. et al. Design, synthesis, evaluation and molecular modeling studies of some novel N-substituted piperidine-3-carboxylic acid derivatives as potential anticonvulsants. Med Chem Res 27, 1206–1225 (2018). https://doi.org/10.1007/s00044-018-2141-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-018-2141-9