Abstract
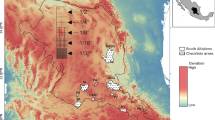
Flow regulations, human activities and drying events have been shown to drive diversity patterns of stream fish communities globally. Along with alpha-diversity distributions across space and time, study of beta-diversity patterns provides a deeper understanding of the mechanisms and processes of overall diversity distributions. It has been shown that water flow conditions can determine the beta-diversity patterns in stream fish communities: in general, perennial habitats are more similar, while intermittent and regulated conditions tend to increase dissimilarities among sites. However, it is not clear whether these patterns result from changes in abundance replacement or from differences in species abundance. Here, we investigated beta-diversity patterns in tropical fish communities of central India and their relation to habitat structural properties and water conditions. We performed our analysis for the overall region (18 sites) and also across three distinct flow conditions (6 sites for each flow regime). We used a partitioning framework to uncover the contribution of abundance replacement and abundance difference to beta-diversity patterns for the overall region and for three flow conditions separately. Our results suggest that at a regional scale all the sites show an equal contribution of replacement and abundance difference components, while seasonal samples were homogeneous. Our results confirmed that intermittent and regulated sites are more heterogeneous than perennial sites. The observed changes in beta-diversity in intermittent and regulated sites were related to both abundance difference and replacement components. Dissimilarities between sites were explained by physicochemical (temperature, pH, dissolved oxygen) parameters but not by habitat structural (stream width, depth) parameters.







Similar content being viewed by others
References
Alexandre CM, Ferreira TF, Almeida PR (2013) Fish assemblages in non-regulated and regulated rivers from permanent and temporary Iberian systems. River Res Appl 29:1042–1058. https://doi.org/10.1002/rra.2591
Alexandre CM, Ferreira MT, Almeida PR (2015) Life history of a cyprinid species in non-regulated and regulated rivers from permanent and temporary Mediterranean basins. Ecohydrology 8:1137–1153. https://doi.org/10.1002/eco.1572
Altermatt F (2013) Diversity in riverine metacommunities: a network perspective. Aquat Ecol 47:365–377
Altermatt F, Seymour M, Martinez N (2013) River network properties shape α-diversity and community similarity patterns of aquatic insect communities across major drainage basins. J Biogeogr 40:2249–2260. https://doi.org/10.1111/jbi.12178
Anderson MJ (2006) Distance-based tests for homogeneity of multivariate dispersions. Biometrics 62:245–253. https://doi.org/10.1111/j.1541-0420.2005.00440.x
Anderson MJ, Crist TO, Chase JM et al (2011) Navigating the multiple meanings of β diversity: a roadmap for the practicing ecologist. Ecol Lett 14:19–28. https://doi.org/10.1111/j.1461-0248.2010.01552.x
Astorga A, Oksanen J, Luoto M et al (2012) Distance decay of similarity in freshwater communities: do macro- and microorganisms follow the same rules? Glob Ecol Biogeogr 21:365–375. https://doi.org/10.1111/j.1466-8238.2011.00681.x
Bain MB, Finn JT, Booke HE (1988) Streamflow regulation and fish community structure. Ecology 69:382–392. https://doi.org/10.2307/1940436
Balcombe SR, Arthington AH (2009) Temporal changes in fish abundance in response to hydrological variability in a dryland floodplain river. Mar Freshw Res 60:146. https://doi.org/10.1071/MF08118
Bhat A (2004) Patterns in the distribution of freshwater fishes in rivers of Central Western Ghats, India and their associations with environmental gradients. Hydrobiologia 529:83–97. https://doi.org/10.1007/s10750-004-4949-1
Boschilia SM, de Oliveira EF, Schwarzbold A (2016) Partitioning beta diversity of aquatic macrophyte assemblages in a large subtropical reservoir: prevalence of turnover or nestedness? Aquat Sci 78:615–625. https://doi.org/10.1007/s00027-015-0450-3
Bunn SE, Arthington AH (2002) Basic principles and ecological consequences of altered flow regimes for aquatic biodiversity. Environ Manage 30:492–507. https://doi.org/10.1007/s00267-002-2737-0
Butler MJ, Stein AR (1985) An analysis of the mechanisms governing species replacement in crayfish. Oecologia 66:168–177
Cañedo-Argüelles M, Boersma KS, Bogan MT et al (2015) Dispersal strength determines meta-community structure in a dendritic riverine network. J Biogeogr 42:778–790. https://doi.org/10.1111/jbi.12457
Daniels RJR (2002) Freshwater fishes of peninsular India. University Press, India
Datry T, Larned ST, Fritz KM et al (2014a) Broad-scale patterns of invertebrate richness and community composition in temporary rivers: effects of flow intermittence. Ecography 37:94–104. https://doi.org/10.1111/j.1600-0587.2013.00287.x
Datry T, Larned ST, Tockner K (2014b) Intermittent rivers: a challenge for freshwater ecology. Bioscience 64:229–235. https://doi.org/10.1093/biosci/bit027
Datry T, Moya N, Zubieta J, Oberdorff T (2016a) Determinants of local and regional communities in intermittent and perennial headwaters of the Bolivian Amazon. Freshw Biol 61:1335–1349. https://doi.org/10.1111/fwb.12706
Datry T, Melo AS, Moya N et al (2016b) Metacommunity patterns across three Neotropical catchments with varying environmental harshness. Freshw Biol 61:277–292. https://doi.org/10.1111/fwb.12702
Davey AJH, Kelly DJ (2007) Fish community responses to drying disturbances in an intermittent stream: a landscape perspective. Freshw Biol 52:1719–1733. https://doi.org/10.1111/j.1365-2427.2007.01800.x
Dudgeon D (2008) Tropical streams ecology. Elsevier, USA
Dufrêne M, Legendre P (1997) Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecol Monogr 67:345–366. https://doi.org/10.2307/2963459
Fischer S, Kummer H (2000) Effects of residual flow and habitat fragmentation on distribution and movement of bullhead (Cottus gobio L.) in an alpine stream. Hydrobiologia 422/423:305–317. https://doi.org/10.1023/A:1017083714513
Freeman MC, Bowen ZH, Bovee KD, Irwin ER (2001) Flow and habitat effects on juvenile fish abundance in natural and altered flow regimes. Ecol Appl 11:179–190. https://doi.org/10.1890/1051-0761(2001)011[0179:FAHEOJ]2.0.CO;2
Gehrke PC, Brown P, Schiller CB et al (1995) River regulation and fish communities in the Murray-Darling River system, Australia. Regul Rivers Res Manag 11:363–375
Guégan J-F, Lek S, Oberdorff T (1998) Energy availability and habitat heterogeneity predict global riverine fish diversity. Nature 391:382–384. https://doi.org/10.1038/34899
Heino J (2013) The importance of metacommunity ecology for environmental assessment research in the freshwater realm. Biol Rev 88:166–178. https://doi.org/10.1111/j.1469-185X.2012.00244.x
Heino J, Melo AS, Bini LM, Altermatt F et al (2015) A comparative analysis reveals weak relationships between ecological factors and beta diversity of stream insect metacommunities at two spatial levels. Ecol Evol 5(6):1235–1248
Hill AM, Lodge DM (1994) Diel changes in resource demand: competition and predation in species replacement among Crayfishes. Ecology 75:2118–2126. https://doi.org/10.2307/1941615
Hoeinghaus DJ, Layman CA, Arrington DA, Winemiller KO (2003) Spatiotemporal variation in fish assemblage structure in tropical floodplain creeks. Environ Biol Fishes 67:379–387. https://doi.org/10.1023/A:1025818721158
Horwitz RJ (1978) Temporal variability patterns and the distributional patterns of stream fishes. Ecol Monogr 48:307–321. https://doi.org/10.2307/2937233
Jackson DA, Peres-Neto PR, Olden JD (2001) What controls who is where in freshwater fish communities; the roles of biotic, abiotic, and spatial factors. Can J Fish Aquat Sci 58:157–170. https://doi.org/10.1139/f00-239
Jayaram KC (2012) The freshwater fishes of the indian region, second. Narendra Publishing House, New Delhi
Kornis MS, Weidel BC, Powers SM et al (2015) Fish community dynamics following dam removal in a fragmented agricultural stream. Aquat Sci 77:465–480. https://doi.org/10.1007/s00027-014-0391-2
Kougioumoutzis K, Simaiakis SM, Tiniakou A (2014) Network biogeographical analysis of the central Aegean archipelago. J Biogeogr 41:1848–1858. https://doi.org/10.1111/jbi.12342
Labbe TR, Fausch KD (2000) Dynamics of intermittent stream habitat regulate persistence of a threatened fish at multiple scales. Ecol Appl 10:1774–1791. https://doi.org/10.1890/1051-0761(2000)010[1774:DOISHR]2.0.CO;2
Lake PS (2003) Ecological effects of perturbation by drought in flowing waters. Freshw Biol 48:1161–1172. https://doi.org/10.1046/j.1365-2427.2003.01086.x
Legendre P (2014) Interpreting the replacement and richness difference components of beta diversity. Glob Ecol Biogeogr 23:1324–1334. https://doi.org/10.1111/geb.12207
Legendre P, Legendre L (1998) Numerical ecology, second. Elsevier Science BV, Amsterdam
Leprieur F, Tedesco PA, Hugueny B et al (2011) Partitioning global patterns of freshwater fish beta diversity reveals contrasting signatures of past climate changes. Ecol Lett 14:325–334. https://doi.org/10.1111/j.1461-0248.2011.01589.x
MacArthur RH, Wilson EO (1967) The theory of island biogeography. Princeton University Press, Princeton
Magurran AE, Dornelas M, Moyes F et al (2015) Rapid biotic homogenization of marine fish assemblages. Nat Commun 6:8405. https://doi.org/10.1038/ncomms9405
Marshall JC, Menke N, Crook DA et al (2016) Go with the flow: the movement behaviour of fish from isolated waterhole refugia during connecting flow events in an intermittent dryland river. Freshw Biol 61:1242–1258. https://doi.org/10.1111/fwb.12707
Matthews WJ, Marsh-Matthews E (2003) Effects of drought on fish across axes of space, time and ecological complexity. Freshw Biol 48:1232–1253. https://doi.org/10.1046/j.1365-2427.2003.01087.x
Matthews WJ, Hough DJ, Robison HW (1992) Similarities in fish distribution and water quality patterns in streams of Arkansas: congruence of multivariate analyses. Copeia 1992:296. https://doi.org/10.2307/1446191
McClure MM, McIntyre PB, McCune AR (2006) Notes on the natural diet and habitat of eight danionin fishes, including the zebrafish Danio rerio. J Fish Biol 69:553–570. https://doi.org/10.1111/j.1095-8649.2006.01125.x
Mims MC, Olden JD (2012) Life history theory predicts fish assemblage response to hydrologic regimes. Ecology 93:35–45. https://doi.org/10.1890/11-0370.1
Miyazono S, Taylor CM (2013) Effects of habitat size and isolation on species immigration-extinction dynamics and community nestedness in a desert river system. Freshw Biol 58:1303–1312. https://doi.org/10.1111/fwb.12127
Muneepeerakul R, Bertuzzo E, Lynch HJ et al (2008) Neutral metacommunity models predict fish diversity patterns in Mississippi-Missouri basin. Nature 453:220–222
Oksanen J, Blanchet FG, Friendly M et al (2015) vegan: community ecology package. R package version 2.2-1
Perkin JS, Gido KB, Cooper AR et al (2015) Fragmentation and dewatering transform Great Plains stream fish communities. Ecol Monogr 85:73–92. https://doi.org/10.1890/14-0121.1
Podani J, Schmera D (2011) A new conceptual and methodological framework for exploring and explaining pattern in presence - absence data. Oikos 120:1625–1638. https://doi.org/10.1111/j.1600-0706.2011.19451.x
Podani J, Ricotta C, Schmera D (2013) A general framework for analyzing beta diversity, nestedness and related community-level phenomena based on abundance data. Ecol Complex 15:52–61. https://doi.org/10.1016/j.ecocom.2013.03.002
Poff NL, Zimmerman JKH (2010) Ecological responses to altered flow regimes: a literature review to inform the science and management of environmental flows. Freshw Biol 55:194–205. https://doi.org/10.1111/j.1365-2427.2009.02272.x
Power ME, Stewart AJ (1987) Disturbance and recovery of an algal assemblage following flooding in an Oklahoma Stream. Am Midl Nat 117:333. https://doi.org/10.2307/2425975
QGIS Development Team (2016) QGIS geographic information system. Open source geospatial foundation. http://qgis.osgeo.org. Accessed 19 Sept 2017
R Core Team (2015). R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rodriguez MA (2010) A modeling framework for assessing long-distance dispersal and loss of connectivity in stream fish. In: Gido KB, Jackson DA (eds) Community ecology of stream fishes: concepts, approaches, and techniques. American Fisheries Society Symposium, Bethesda
Schlosser IJ (1991) Stream fish ecology: a landscape perspective. Bioscience 41:704–712. https://doi.org/10.2307/1311765
Schlosser IJ (1995) Critical landscape attributes that influence fish population dynamics in headwater streams. Hydrobiologia 303:71–81. https://doi.org/10.1007/BF00034045
Shukla R, Bhat A (2017a) Environmental drivers of α-diversity patterns in monsoonal tropical stream fish assemblages: a case study from tributaries of Narmada basin, India. Environ Biol Fish 7:749–761
Shukla R, Bhat A (2017b) Morphological divergences and ecological correlates among wild populations of zebrafish (Danio rerio). Environ Biol Fish 1 100(3):251–264
Socolar JB, Gilroy JJ, Kunin WE, Edwards DP (2016) How should beta-diversity inform biodiversity conservation? Trends Ecol Evol 31:67–80. https://doi.org/10.1016/j.tree.2015.11.005
Strayer D, Dudgeon D (2010) Freshwater biodiversity conservation: recent progress and future challenges. J N Am Benthol Soc 29:344–358. https://doi.org/10.1899/08-171.1
Suriyampola PS, Shelton DS, Shukla R et al (2016) Zebrafish social behavior in the wild. Zebrafish 13:1–8. https://doi.org/10.1089/zeb.2015.1159
Taylor CM (1997) Fish species richness and incidence patterns in isolated and connected stream pools: effects of pool volume and spatial position. Oecologia 110:560–566. https://doi.org/10.1007/s004420050196
Taylor CM, Holder TL, Fiorillo RA et al (2006) Distribution, abundance, and diversity of stream fishes under variable environmental conditions. Can J Fish Aquat Sci 63:43–54. https://doi.org/10.1139/f05-203
Tyers MB (2017) riverdist: river network distance computation and applications. R package version 0.15.0
Unni KS (1996) Ecology of river Narmada. APH, Ontario
Webb JA, Miller KA, King EL et al (2013) Squeezing the most out of existing literature: a systematic re-analysis of published evidence on ecological responses to altered flows. Freshw Biol 58:2439–2451. https://doi.org/10.1111/fwb.12234
Weihs C, Ligges U, Luebke K, Raabe N (2005) klaR analyzing German business cycles. In: Baier D, Decker R, Schmidt-Thieme L (eds) Data analysis and decision support. Springer, Berlin, pp 335–343
Whittaker RH (1952) A study of summer foliage insect communities in the Great Smoky Mountains. Ecol Monogr 22:1–44. https://doi.org/10.2307/1948527
Wikramanayake ED (1990) Ecomorphology and biogeography of a tropical stream fish assemblage: evolution of assemblage structure. Ecology 71:1756. https://doi.org/10.2307/1937583
Winemiller KO (1989) Patterns of variation in life history among South American fishes in seasonal environments. Oecologia 81:225–241. https://doi.org/10.1007/BF00379810
Winemiller KO, Jepsen DB (1998) Effects of seasonality and fish movement on tropical river food webs. J Fish Biol 53:267–296. https://doi.org/10.1111/j.1095-8649.1998.tb01032.x
Acknowledgements
The authors thank Durlabh Shukla for field assistance and local fish collectors for help with collections of fish and data on environmental parameters at the study sites. The authors also wish to thank the Indian Institute of Science Education and Research Kolkata (IISER-Kolkata, India) for financial support during this study. RS was supported through Institutional Junior and Senior Research Fellowships provided by IISER Kolkata. We thank Dr. Florian Altermatt for very helpful and constructive suggestions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Ethical approval
No animals were harmed during this study and all individuals were returned to their natural habitats in the field. The study protocol carried out were in accordance with the ethical standards of the institution (Institutional Animal Ethics Committee’s (IAEC)), IISER Kolkata and adhered to the guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Government of India.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shukla, R., Bhat, A. Beta-diversity partitioning and drivers of variations in tropical fish community structure in central India. Aquat Sci 80, 18 (2018). https://doi.org/10.1007/s00027-018-0568-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00027-018-0568-1