Abstract
Background
Acute liver injury is liver cell injury that occurs rapidly in a short period of time. Caffeine has been shown to maintain hepatoprotective effect with an unclear mechanism. Endoplasmic reticulum stress (ERS) has significant effects in acute liver injury. Induction of GRP78 is a hallmark of ERS. Whether or not caffeine’s function is related to GRP78 remains to be explored.
Methods
Acute liver injury model was established by LPS-treated L02 cells and in vivo administration of LPS/D-Gal in mice. Caffeine was pre-treated in L02 cells or mice. Gene levels was determined by real-time PCR and western blot. Cell viability was tested by CCK-8 assay and cell apoptosis was tested by flow cytometry. The interaction of GRP78 and NEDD4L was determined by Pull-down and co-immunoprecipitation (Co-IP) assay. The ubiquitination by NEDD4L on GRP78 was validated by in vitro ubiquitination assay.
Results
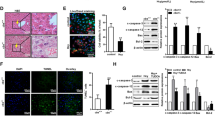
Caffeine protected liver cells against acute injury induced cell apoptosis and ERS both in vitro and in vivo. Suppression of GRP78 could block the LPS-induced cell apoptosis and ERS. NEDD4L was found to interact with GRP78 and ubiquitinate its lysine of 324 site directly. Caffeine treatment induced the expression of NEDD4L, resulting in the ubiquitination and inhibition of GRP78.
Conclusion
Caffeine mitigated the acute liver injury by stimulating NEDD4L expression, which inhibited GRP78 expression via ubiquitination at its K324 site. Low dose of caffeine could be a promising therapeutic treatment for acute liver injury.







Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this article. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ER:
-
Endoplasmic reticulum
- ERS:
-
Endoplasmic reticulum stress
- GRP78:
-
Glucose-regulated protein 78
- PERK:
-
Protein kinase R-like ER kinase
- IRE-1α:
-
Inositol-requiring enzyme 1 alpha
- ATF6:
-
Activating transcription factor 6
- ATF4:
-
Activating transcription factor 6
- CHOP:
-
C/EBP homologous protein
- CAFF:
-
Caffeine
- H and E:
-
Hematoxylin–eosin
- FBS:
-
Fetal bovine serum
- qRT-PCR:
-
Quantitative real-time PCR
- CCK8:
-
Cell counting kit-8
- LPS:
-
Lipopolysaccharide
- D-gal:
-
D-galactosamine
- Co-IP:
-
Co-immunoprecipitation
References
Ding W, Fan YY, Zhang C, Fu L, Chen X, Xu DX. Obeticholic acid differentially regulates hepatic injury and inflammation at different stages of D-galactosamine/lipopolysaccharide-evoked acute liver failure. Eur J Pharm. 2019;850:150–7.
Huang ZB, Zheng YX, Li N, Cai SL, Huang Y, Wang J, et al. Protective effects of specific cannabinoid receptor 2 agonist GW405833 on concanavalin A-induced acute liver injury in mice. Acta Pharmacol Sin. 2019;40:1404–11.
Alpert L, Hart J. The pathology of alcoholic liver disease. Clin Liver Dis. 2016;20:473–89.
Clark VC, Marek G, Liu C, Collinsworth A, Shuster J, Kurtz T, et al. Clinical and histologic features of adults with alpha-1 antitrypsin deficiency in a non-cirrhotic cohort. J Hepatol. 2018;69:1357–64.
Kroemer G, Galluzzi L, Vandenabeele P, Abrams J, Alnemri ES, Baehrecke EH, et al. Classification of cell death: recommendations of the nomenclature committee on cell death 2009. Cell Death Differ. 2009;16:3–11.
Mahdi AA, Rizvi SH, Parveen A. Role of endoplasmic reticulum stress and unfolded protein responses in health and diseases. Indian J Clin Biochem. 2016;31:127–37.
Iurlaro R, Munoz-Pinedo C. Cell death induced by endoplasmic reticulum stress. FEBS J. 2016;283:2640–52.
Liu G, Sun Y, Li Z, Song T, Wang H, Zhang Y, et al. Apoptosis induced by endoplasmic reticulum stress involved in diabetic kidney disease. Biochem Biophys Res Commun. 2008;370:651–6.
Bernales S, Papa FR, Walter P. Intracellular signaling by the unfolded protein response. Annu Rev Cell Dev Biol. 2006;22:487–508.
Lien YC, Kung HN, Lu KS, Jeng CJ, Chau YP. Involvement of endoplasmic reticulum stress and activation of MAP kinases in beta-lapachone-induced human prostate cancer cell apoptosis. Histol Histopathol. 2008;23:1299–308.
Harada N, Okuyama M, Yoshikatsu A, Yamamoto H, Ishiwata S, Hamada C, et al. Endoplasmic reticulum stress in mice increases hepatic expression of genes carrying a premature termination codon via a nutritional status-independent GRP78-dependent mechanism. J Cell Biochem. 2017;118:3810–24.
Wang X, Wu X, Wang Q, Zhang Y, Wang C, Chen J. NLRP6 suppresses gastric cancer growth via GRP78 ubiquitination. Exp Cell Res. 2020;395: 112177.
Hughes A, Oxford AE, Tawara K, Jorcyk CL, Oxford JT. Endoplasmic reticulum stress and unfolded protein response in cartilage pathophysiology; contributing factors to apoptosis and osteoarthritis. Int J Mol Sci. 2017;18:665.
Meng X, Zhu Y, Tao L, Zhao S, Qiu S. Periostin has a protective role in melatonin-induced cell apoptosis by inhibiting the eIF2alphaATF4 pathway in human osteoblasts. Int J Mol Med. 2018;41:1003–12.
Tian RD, Chen YQ, He YH, Tang YJ, Chen GM, Yang FW, et al. Phosphorylation of eIF2alpha mitigates endoplasmic reticulum stress and hepatocyte necroptosis in acute liver injury. Ann Hepatol. 2020;19:79–87.
Ruhl CE, Everhart JE. Coffee and tea consumption are associated with a lower incidence of chronic liver disease in the United States. Gastroenterology. 2005;129:1928–36.
Azam S, Hadi N, Khan NU, Hadi SM. Antioxidant and prooxidant properties of caffeine, theobromine and xanthine. Med Sci Monit. 2003;9:BR325–30.
Cachon AU, Quintal-Novelo C, Medina-Escobedo G, Castro-Aguilar G, Moo-Puc RE. Hepatoprotective effect of low doses of caffeine on CCl4-induced liver damage in rats. J Diet Suppl. 2017;14:158–72.
Yang B, Kumar S. Nedd4 and Nedd4-2: closely related ubiquitin-protein ligases with distinct physiological functions. Cell Death Differ. 2010;17:68–77.
Kwak YD, Wang B, Li JJ, Wang R, Deng Q, Diao S, et al. Upregulation of the E3 ligase NEDD4-1 by oxidative stress degrades IGF-1 receptor protein in neurodegeneration. J Neurosci. 2012;32:10971–81.
Wang H, Sun RQ, Camera D, Zeng XY, Jo E, Chan SM, et al. Endoplasmic reticulum stress up-regulates Nedd4-2 to induce autophagy. FASEB J. 2016;30:2549–56.
Zhou YP, Xia Q. Inhibition of miR-103a-3p suppresses lipopolysaccharide-induced sepsis and liver injury by regulating FBXW7 expression. Cell Biol Int. 2020;44:1798–810.
Li Y, Chen Y, Huang H, Shi M, Yang W, Kuang J, et al. Autophagy mediated by endoplasmic reticulum stress enhances the caffeine-induced apoptosis of hepatic stellate cells. Int J Mol Med. 2017;40:1405–14.
Hu J, Zhu Z, Ying H, Yao J, Ma H, Li L, et al. Oleoylethanolamide protects against acute liver injury by regulating Nrf-2/HO-1 and NLRP3 pathways in mice. Front Pharm. 2020;11: 605065.
Li S, Jin S, Chen W, Yu J, Fang P, Zhou G, et al. Mangiferin alleviates endoplasmic reticulum stress in acute liver injury by regulating the miR-20a/miR-101a-Nrf2 axis. J Biochem. 2020;168:365–74.
Pan Y, Long X, Yi R, Zhao X. Polyphenols in Liu Bao tea can prevent CCl(4)-induced hepatic damage in mice through its antioxidant capacities. Nutrients. 2018;10:1280.
Shi T, Song W, Xu R. Autophagy and ER stress in LPS/GalN induced acute liver injury. Mol Med Rep. 2017;16:7001–5.
Tai M, Zhang J, Song S, Miao R, Liu S, Pang Q, et al. Protective effects of luteolin against acetaminophen-induced acute liver failure in mouse. Int Immunopharmacol. 2015;27:164–70.
Clarke R, Cook KL, Hu R, Facey CO, Tavassoly I, Schwartz JL, et al. Endoplasmic reticulum stress, the unfolded protein response, autophagy, and the integrated regulation of breast cancer cell fate. Cancer Res. 2012;72:1321–31.
Clarke R, Shajahan AN, Wang Y, Tyson JJ, Riggins RB, Weiner LM, et al. Endoplasmic reticulum stress, the unfolded protein response, and gene network modeling in antiestrogen resistant breast cancer. Horm Mol Biol Clin Investig. 2011;5:35–44.
Hu R, Warri A, Jin L, Zwart A, Riggins RB, Fang HB, et al. NF-kappaB signaling is required for XBP1 (unspliced and spliced)-mediated effects on antiestrogen responsiveness and cell fate decisions in breast cancer. Mol Cell Biol. 2015;35:379–90.
Cook KL, Soto-Pantoja DR, Clarke PA, Cruz MI, Zwart A, Warri A, et al. Endoplasmic reticulum stress protein GRP78 modulates lipid metabolism to control drug sensitivity and antitumor immunity in breast cancer. Cancer Res. 2016;76:5657–70.
Chang YJ, Huang YP, Li ZL, Chen CH. GRP78 knockdown enhances apoptosis via the down-regulation of oxidative stress and Akt pathway after epirubicin treatment in colon cancer DLD-1 cells. PLoS ONE. 2012;7: e35123.
Samanta S, Yang S, Debnath B, Xue D, Kuang Y, Ramkumar K, et al. The Hydroxyquinoline analogue YUM70 inhibits GRP78 to induce ER stress-mediated apoptosis in pancreatic cancer. Cancer Res. 2021;81:1883–95.
Wu J, Wu Y, Lian X. Targeted inhibition of GRP78 by HA15 promotes apoptosis of lung cancer cells accompanied by ER stress and autophagy. Biol Open. 2020;9:bio053298.
Mukwevho E, Kohn TA, Lang D, Nyatia E, Smith J, Ojuka EO. Caffeine induces hyperacetylation of histones at the MEF2 site on the Glut4 promoter and increases MEF2A binding to the site via a CaMK-dependent mechanism. Am J Physiol Endocrinol Metab. 2008;294:E582–8.
Ping J, Wang JF, Liu L, Yan YE, Liu F, Lei YY, et al. Prenatal caffeine ingestion induces aberrant DNA methylation and histone acetylation of steroidogenic factor 1 and inhibits fetal adrenal steroidogenesis. Toxicology. 2014;321:53–61.
Yu H, Yang T, Gao P, Wei X, Zhang H, Xiong S, et al. Caffeine intake antagonizes salt sensitive hypertension through improvement of renal sodium handling. Sci Rep. 2016;6:25746.
Kawaratani H, Tsujimoto T, Douhara A, Takaya H, Moriya K, Namisaki T, et al. The effect of inflammatory cytokines in alcoholic liver disease. Mediators Inflamm. 2013;2013: 495156.
Su GL. Lipopolysaccharides in liver injury: molecular mechanisms of kupffer cell activation. Am J Physiol Gastrointest Liver Physiol. 2002;283:G256–65.
Acknowledgements
We would like to give our sincere gratitude to the reviewers for their constructive comments.
Funding
This work was supported by grant from National Natural Sciences Foundation of China (No.81970523), and Natural Sciences Foundation of Hunan province (2020JJ4877).
Author information
Authors and Affiliations
Contributions
XWH: conceptualization, writing–original draft, and methodology; XML: formal analysis; AMW: supervision; YMF: validation; FJZ: data curation; FZ: resources; LPC: investigation; HL: software; YHX: visualization; JX: project administration; JL: funding acquisition, writing–review and editing. All authors have read and approved the final version of this manuscript to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Not applicable. This article does not contain any studies with human participants or animals performed by any of the authors.
Consent for publication
Not applicable. This article does not contain any studies with human participants performed by any of the authors.
Additional information
Responsible Editor: John Di Battista.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11_2022_1603_MOESM1_ESM.tif
Supplementary file1 NEDD4L inhibited liver cell apoptosis via regulating GRP78. A mRNA and B protein levels of NEDD4L and GRP78 in L02 cells. C Flow cytometry and statistic result for apoptosis detection of L02 cells. *P < 0.05, **P < 0.01, ***P < 0.001, one-way ANOVA followed by Tukey’s multiple comparison test, N = 3. (TIF 1122 KB)
11_2022_1603_MOESM2_ESM.tif
Supplementary file2 The effect of caffeine treatment on LPS level. Relative level of LPS in the culture medium of L02 cells after treated with or without caffeine (TIF 100 KB)
Rights and permissions
About this article
Cite this article
Hu, XW., Li, XM., Wang, AM. et al. Caffeine alleviates acute liver injury by inducing the expression of NEDD4L and deceasing GRP78 level via ubiquitination. Inflamm. Res. 71, 1213–1227 (2022). https://doi.org/10.1007/s00011-022-01603-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-022-01603-0